Abstract
Background
Regular physical activity is associated with improved symptom control in patients with breast cancer but its association with chemotherapy completion or response is unclear.
Methods
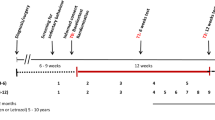
Using a prospective design, 1075 breast cancer patients receiving neoadjuvant chemotherapy between March 2012 and February 2017 were studied. Physical activity was assessed using the Global Physical Activity Questionnaire [GPAQ-16], quantified in standardised MET-h/wk. Chemotherapy completion was defined as the proportion of patients completing planned treatment course, requiring dose reduction, or requiring dose delay. Response was evaluated by pathologic complete response (pCR). Associations between physical activity and primary outcomes were assessed using multivariable logistic regression models.
Results
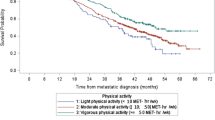
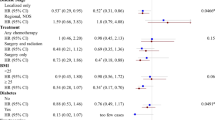
There was no differences between any chemotherapy completion outcome on the basis of physical activity classification. The percent of patients not completing planned treatment was 5.7% for ≦0.33 MET-h/wk, compared with 6.8% for 0.34–16.65 MET-h/wk, and 4.6% for ≥16.6 MET-h/wk (p = 0.52). No significant relationships were observed between physical activity dose classification and pCR for the overall cohort or upon stratification by clinical subtype.
Conclusion
Future studies are required to further investigate the relationship between pre-treatment levels of physical activity and function on treatment completion and response in breast and other cancer populations.
Clinical trial registration
NCT01993498.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Data availability
Data will be made available upon reasonable request.
References
Budman DR, Berry DA, Cirrincione CT, Henderson IC, Wood WC, Weiss RB, et al. Dose and dose intensity as determinants of outcome in the adjuvant treatment of breast cancer. The Cancer and Leukemia Group B. J Natl Cancer Inst. 1998;90:1205–11.
Yuan JQ, Wang SM, Tang LL, Mao J, Wu YH, Hai J, et al. Relative dose intensity and therapy efficacy in different breast cancer molecular subtypes: a retrospective study of early stage breast cancer patients treated with neoadjuvant chemotherapy. Breast Cancer Res Treat. 2015;151:405–13.
Bonadonna G, Valagussa P, Moliterni A, Zambetti M, Brambilla C. Adjuvant cyclophosphamide, methotrexate, and fluorouracil in node-positive breast cancer: the results of 20 years of follow-up. N Engl J Med. 1995;332:901–6.
Lyman GH, Dale DC, Crawford J. Incidence and predictors of low dose-intensity in adjuvant breast cancer chemotherapy: a nationwide study of community practices. J Clin Oncol. 2003;21:4524–31.
Bland KA, Zadravec K, Landry T, Weller S, Meyers L, Campbell KL. Impact of exercise on chemotherapy completion rate: a systematic review of the evidence and recommendations for future exercise oncology research. Crit Rev Oncol Hematol. 2019;136:79–85.
Groen WG, Naaktgeboren WR, van Harten WH, van Vulpen JK, Kool N, Sonke GS, et al. Physical fitness and chemotherapy tolerance in patients with early-stage breast cancer. Med Sci Sports Exerc. 2021;54:537–42.
An KY, Arthuso FZ, Kang DW, Morielli AR, Ntoukas SM, Friedenreich CM, et al. Exercise and health-related fitness predictors of chemotherapy completion in breast cancer patients: pooled analysis of two multicenter trials. Breast Cancer Res Treat. 2021;188:399–407.
Scott JM, Stene G, Edvardsen E, Jones LW. Performance status in cancer: not broken, but time for an upgrade? J Clin Oncol. 2020;38:2824–9.
Kokkinos P, Myers J. Exercise and physical activity: clinical outcomes and applications. Circulation. 2010;122:1637–48.
Koelwyn GJ, Khouri M, Mackey JR, Douglas PS, Jones LW. Running on empty: cardiovascular reserve capacity and late effects of therapy in cancer survivorship. J Clin Oncol. 2012;30:4458–61.
Betof AS, Lascola CD, Weitzel D, Landon C, Scarbrough PM, Devi GR, et al. Modulation of murine breast tumor vascularity, hypoxia and chemotherapeutic response by exercise. J Natl Cancer Inst. 2015;107:djv040.
Vaz-Luis I, Cottu P, Mesleard C, Martin AL, Dumas A, Dauchy S, et al. UNICANCER: French prospective cohort study of treatment-related chronic toxicity in women with localised breast cancer (CANTO). ESMO Open. 2019;4:e000562.
WHO. WHO STEPS Surveillance Manual: the WHO STEPwise approach to chronic disease risk factor surveillance. Geneva: Wrold Health Organization; 2005.
WHO. Global recommendations on physical activity for health. Geneva: World Health Organization; 2010.
Bull FC, Maslin TS, Armstrong T. Global physical activity questionnaire (GPAQ): nine country reliability and validity study. J Phys Act Health. 2009;6:790–804.
von Minckwitz G, Untch M, Blohmer JU, Costa SD, Eidtmann H, Fasching PA, et al. Definition and impact of pathologic complete response on prognosis after neoadjuvant chemotherapy in various intrinsic breast cancer subtypes. J Clin Oncol. 2012;30:1796–804.
Usiskin I, Li F, Irwin ML, Cartmel B, Sanft T. Association between pre-diagnosis BMI, physical activity, pathologic complete response, and chemotherapy completion in women treated with neoadjuvant chemotherapy for breast cancer. Breast Cancer. 2019;26:719–28.
Courneya KS, Segal RJ, Mackey JR, Gelmon K, Reid RD, Friedenreich CM, et al. Effects of aerobic and resistance exercise in breast cancer patients receiving adjuvant chemotherapy: a multicenter randomized controlled trial. J Clin Oncol. 2007;25:4396–404.
van Waart H, Stuiver MM, van Harten WH, Geleijn E, Kieffer JM, Buffart LM, et al. Effect of low-intensity physical activity and moderate- to high-intensity physical exercise during adjuvant chemotherapy on physical fitness, fatigue, and chemotherapy completion rates: results of the PACES randomized clinical trial. J Clin Oncol. 2015;33:1918–27.
Van Blarigan EL, Gerstenberger JP, Kenfield SA, Giovannucci EL, Stampfer MJ, Jones LW, et al. Physical activity and prostate tumor vessel morphology: data from the health professionals follow-up study. Cancer Prev Res (Philos). 2015;8:962–7.
Nia HT, Munn LL, Jain RK. Physical traits of cancer. Science. 2020;370:eaaz0868.
Yang L, Morielli AR, Heer E, Kirkham AA, Cheung WY, Usmani N, et al. Effects of exercise on cancer treatment efficacy: a systematic review of preclinical and clinical studies. Cancer Res. 2021;81:4889–95.
Friedenreich CM, Stone CR, Cheung WY, Hayes SC. Physical activity and mortality in cancer survivors: a systematic review and meta-analysis. JNCI Cancer Spectr. 2020;4:pkz080.
Wennerberg E, Lhuillier C, Rybstein MD, Dannenberg K, Rudqvist NP, Koelwyn GJ, et al. Exercise reduces immune suppression and breast cancer progression in a preclinical model. Oncotarget. 2020;11:452–61.
Koelwyn GJ, Quail DF, Zhang X, White RM, Jones LW. Exercise-dependent regulation of the tumour microenvironment. Nat Rev Cancer. 2017;17:620–32.
Acknowledgements
An interim analysis from this study was presented at the San Antonio Breast Cancer Symposium 2018, Dec 4–8, San Antonio, TX.
Funding
CANTO is funded by Agence Nationale de la Recherche (ANR-10-COHO-0004). IV-L received support from Odyssea, Foundation Gustave Roussy, and CCR17483507 Career Catalyst Research grant. ADM received support from Susan G. Komen, Clinical Research Fellowship from ESMO. LWJ is supported in part by funding from the National Cancer Institute, AKTIV Against Cancer, the KavliTrust, and the Memorial Sloan Kettering Cancer Center Support Grant/Core Grant (P30 CA008748).
Author information
Authors and Affiliations
Contributions
JLB: Wrote the paper. ADM, ASG, MEM: Conceived and designed the analysis and performed the analysis. NMI: Conceived and designed the analysis. SM, PC, FL, CC, AL, OT, LV, CJ, IH, SE, A-LM, PA, AF: Collected the data. IV-L, LWJ: Conceived and designed the analysis and wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
LWJ owns stock in Pacylex, Inc. and Illumisonics, Inc. IV-L reports personal fees from AstraZeneca, Kephren, Amgen, and Novartis. ADM reports personal fees from Thermo Fisher and grants from European Society for Medical Oncology (ESMO), fellowship support, outside the submitted work.
Ethics approval and consent to participate
Informed consent was obtained from all patients prior to study participation at each institution.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Baker, J.L., Di Meglio, A., Gbenou, A.S. et al. Association between physical activity and neoadjuvant chemotherapy completion and pathologic complete response in primary breast cancer: the CANTO study. Br J Cancer 127, 886–891 (2022). https://doi.org/10.1038/s41416-022-01870-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41416-022-01870-y