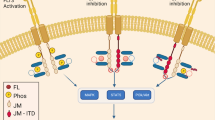
Abstract
Background
Activating mutations in the Fms-like tyrosine kinase 3 (FLT3) are among the most prevalent oncogenic mutations in acute myeloid leukaemia. Inhibitors selectively targeting FLT3 kinase have shown promising clinical activity; their success in the clinic, however, has been limited due to the emergence of acquired resistance.
Methods
CCT245718 was identified and characterised as a dual Aurora A/FLT3 inhibitor through cell-based and biochemical assays. The ability of CCT245718 to overcome TKD-mediated resistance was evaluated in a cell line-based model of drug resistance to FLT3 inhibitors.
Results
CCT245718 exhibits potent antiproliferative activity towards FLT3-ITD + AML cell lines and strongly binds to FLT3-ITD and TKD (D835Y) mutants in vitro. Activities of both FLT3-ITD and Aurora A are also inhibited in cells. Inhibition of FLT3 results in reduced phosphorylation of STAT5, downregulation of survivin and induction of apoptotic cell death. Moreover, CCT245718 overcomes TKD-mediated resistance in a MOLM-13-derived cell line containing FLT3 with both ITD and D835Y mutations. It also inhibits FLT3 signalling in both parental and resistant cell lines compared to FLT3-specific inhibitor MLN518, which is only active in the parental cell line.
Conclusions
Our results demonstrate that CCT245718 is a potent dual FLT3/Aurora A inhibitor that can overcome TKD-mediated acquired resistance.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Peterson LF, Boyapati A, Ahn E-Y, Biggs JR, Okumura AJ, Lo M-C, et al. Acute myeloid leukemia with the 8q22; 21q22 translocation: secondary mutational events and alternative t (8; 21) transcripts. Blood. J Am Soc Hematol. 2007;110:799–805.
Grafone T, Palmisano M, Nicci C, Storti S. An overview on the role of FLT3-tyrosine kinase receptor in acute myeloid leukemia: biology and treatment. Oncology Rev. 2012;6:64–74.
Gilliland DG, Griffin JD. The roles of FLT3 in hematopoiesis and leukemia. Blood J Am Soc Hematol. 2002;100:1532–42.
Levis M. FLT3 mutations in acute myeloid leukemia: what is the best approach in 2013? Hematology. 2013;2013:220–6.
Larrosa-Garcia M, Baer MR. FLT3 inhibitors in acute myeloid leukemia: current status and future directions. Mol Cancer Therapeut. 2017;16:991–1001.
Sidaway P. Gilteritinib improves outcomes in AML. Nat Rev Clin Oncol. 2020;17:69.
Levis M. Midostaurin approved for FLT3-mutated AML. Blood. 2017;129:3403–6.
Assi R, Ravandi F. FLT3 inhibitors in acute myeloid leukemia: choosing the best when the optimal does not exist. Am J Hematol. 2018;93:553–63.
Smith CC, Wang Q, Chin C-S, Salerno S, Damon LE, Levis MJ, et al. Validation of ITD mutations in FLT3 as a therapeutic target in human acute myeloid leukaemia. Nature. 2012;485:260–3.
Li C, Liu L, Liang L, Xia Z, Li Z, Wang X, et al. AMG 925 is a dual FLT3/CDK4 inhibitor with the potential to overcome FLT3 inhibitor resistance in acute myeloid leukemia. Mol Cancer Therapeut. 2015;14:375–83.
Minson KA, Smith CC, DeRyckere D, Libbrecht C, Lee-Sherick AB, Huey MG, et al. The MERTK/FLT3 inhibitor MRX-2843 overcomes resistance-conferring FLT3 mutations in acute myeloid leukemia. JCI Insight. 2016;1(3):e85630
Zhang W, Borthakur G, Gao C, Chen Y, Mu H, Ruvolo VR, et al. The dual MEK/FLT3 inhibitor E6201 exerts cytotoxic activity against acute myeloid leukemia cells harboring resistance-conferring FLT3 mutations. Cancer Res. 2016;76:1528–37.
Moore AS, Faisal A, De Castro DG, Bavetsias V, Sun C, Atrash B, et al. Selective FLT3 inhibition of FLT3-ITD+ acute myeloid leukaemia resulting in secondary D835Y mutation: a model for emerging clinical resistance patterns. Leukemia. 2012;26:1462–70.
Malik HS, Bilal A, Ullah R, Iqbal M, Khan S, Ahmed I, et al. Natural and semisynthetic chalcones as dual FLT3 and microtubule polymerization inhibitors. J Nat Prod. 2020;83:3111–21.
Andrews PD. Aurora kinases: shining lights on the therapeutic horizon? Oncogene. 2005;24:5005–15.
Vader G, Lens SM. The Aurora kinase family in cell division and cancer. Biochimica et Biophysica Acta (BBA)-Rev Cancer. 2008;1786:60–72.
Gautschi O, Heighway J, Mack PC, Purnell PR, Lara PN, Gandara DR. Aurora kinases as anticancer drug targets. Clin Cancer Res. 2008;14:1639–48.
Pollard JR, Mortimore M. Discovery and development of aurora kinase inhibitors as anticancer agents. J Med. Chem. 2009;52:2629–51.
Bavetsias V, Linardopoulos S. Aurora kinase inhibitors: current status and outlook. Front Oncol. 2015;5:278.
Goldberg SL, Fenaux P, Craig MD, Gyan E, Lister J, Kassis J, et al. An exploratory phase 2 study of investigational Aurora A kinase inhibitor alisertib (MLN8237) in acute myelogenous leukemia and myelodysplastic syndromes. Leuk Res Rep. 2014;3:58–61.
Löwenberg B, Muus P, Ossenkoppele G, Rousselot P, Cahn J-Y, Ifrah N, et al. Phase 1/2 study to assess the safety, efficacy, and pharmacokinetics of barasertib (AZD1152) in patients with advanced acute myeloid leukemia. Blood. 2011;118:6030–6.
Kantarjian HM, Sekeres MA, Ribrag V, Rousselot P, Garcia-Manero G, Jabbour EJ, et al. Phase I study assessing the safety and tolerability of barasertib (AZD1152) with low-dose cytosine arabinoside in elderly patients with AML. Clin Lymphoma Myeloma Leuk. 2013;13:559–67.
Grundy M, Seedhouse C, Shang S, Richardson J, Russell N, Pallis M. The FLT3 internal tandem duplication mutation is a secondary target of the aurora B kinase inhibitor AZD1152-HQPA in acute myelogenous leukemia cells. Mol Cancer Therapeut. 2010;9:661–72.
Moore AS, Faisal A, Mak GWY, Miraki-Moud F, Bavetsias V, Valenti M, et al. Quizartinib-resistant FLT3-ITD acute myeloid leukemia cells are sensitive to the FLT3-Aurora kinase inhibitor CCT241736. Blood Adv. 2020;4:1478–91.
Bavetsias V, Faisal A, Crumpler S, Brown N, Kosmopoulou M, Joshi A, et al. Aurora isoform selectivity: design and synthesis of imidazo [4, 5-b] pyridine derivatives as highly selective inhibitors of Aurora-A kinase in cells. J Med Chem. 2013;56:9122–35.
Schrodinger. Ligprep V. 2.3. Schrodinger, LLC, New York; 2009.
Zorn JA, Wang Q, Fujimura E, Barros T, Kuriyan J. Crystal structure of the FLT3 kinase domain bound to the inhibitor quizartinib (AC220). PLoS ONE. 2015;10:e0121177.
Friesner RA, Banks JL, Murphy RB, Halgren TA, Klicic JJ, Mainz DT, et al. Glide: a new approach for rapid, accurate docking and scoring. 1. Method and assessment of docking accuracy. J Med Chem. 2004;47:1739–49.
LeRoy PJ, Hunter JJ, Hoar KM, Burke KE, Shinde V, Ruan J, et al. Localization of human TACC3 to mitotic spindles is mediated by phosphorylation on Ser558 by Aurora A: a novel pharmacodynamic method for measuring Aurora A activity. Cancer Res. 2007;67:5362–70.
Manfredi MG, Ecsedy JA, Meetze KA, Balani SK, Burenkova O, Chen W, et al. Antitumor activity of MLN8054, an orally active small-molecule inhibitor of Aurora A kinase. Proc Natl Acad Sci USA. 2007;104:4106–11.
Weisberg E, Boulton C, Kelly LM, Manley P, Fabbro D, Meyer T, et al. Inhibition of mutant FLT3 receptors in leukemia cells by the small molecule tyrosine kinase inhibitor PKC412. Cancer Cell. 2002;1:433–43.
Burnett A, Wetzler M, Lowenberg B. Therapeutic advances in acute myeloid leukemia. J Clin Oncol. 2011;29:487–94.
Kiyoi H, Naoe T, Nakano Y, Yokota S, Minami S, Miyawaki S, et al. Prognostic implication of FLT3 and N-RAS gene mutations in acute myeloid leukemia. Blood, J Am Soc Hematol. 1999;93:3074–80.
Komanduri KV, Levine RL. Diagnosis and therapy of acute myeloid leukemia in the era of molecular risk stratification. Annu Rev Med. 2016;67:59–72.
Thiede C, Steudel C, Mohr B, Schaich M, Schäkel U, Platzbecker U, et al. Analysis of FLT3-activating mutations in 979 patients with acute myelogenous leukemia: association with FAB subtypes and identification of subgroups with poor prognosis. Blood 2002;99:4326–35. Presented in part at the 42nd Annual Meeting of the American Society of Hematology, December 1–5, 2000, San Francisco, CA (abstract 2334).
Abu‐Duhier F, Goodeve A, Wilson G, Care R, Peake I, Reilly J. Identification of novel FLT‐3 Asp835 mutations in adult acute myeloid leukaemia. Br J Haematol. 2001;113:983–8.
Moore A, Blagg J, Linardopoulos S, Pearson A. Aurora kinase inhibitors: novel small molecules with promising activity in acute myeloid and Philadelphia-positive leukemias. Leukemia. 2010;24:671–8.
Fukuda S, Singh P, Moh A, Abe M, Conway EM, Boswell HS, et al. Survivin mediates aberrant hematopoietic progenitor cell proliferation and acute leukemia in mice induced by internal tandem duplication of Flt3. Blood, J Am Soc Hematol. 2009;114:394–403.
Cheung CHA, Sarvagalla S, Lee JY-C, Huang Y-C, Coumar MS. Aurora kinase inhibitor patents and agents in clinical testing: an update (2011–2013). Expert Opin Therapeutic Pat. 2014;24:1021–38.
Chang Hsu Y, Ke YY, Shiao HY, Lee CC, Lin WH, Chen CH, et al. Facile identification of dual FLT3–Aurora A inhibitors: a computer‐guided drug design approach. ChemMedChem. 2014;9:953–61.
Quentmeier H, Reinhardt J, Zaborski M, Drexler H. FLT3 mutations in acute myeloid leukemia cell lines. Leukemia. 2003;17:120–4.
Weisberg EL, Puissant A, Stone R, Sattler M, Buhrlage SJ, Yang J, et al. Characterization of midostaurin as a dual inhibitor of FLT3 and SYK and potentiation of FLT3 inhibition against FLT3-ITD-driven leukemia harboring activated SYK kinase. Oncotarget. 2017;8:52026–44.
Altieri DC. Survivin, versatile modulation of cell division and apoptosis in cancer. Oncogene. 2003;22:8581–9.
Williams NS, Gaynor RB, Scoggin S, Verma U, Gokaslan T, Simmang C, et al. Identification and validation of genes involved in the pathogenesis of colorectal cancer using cDNA microarrays and RNA interference. Clin Cancer Res. 2003;9:931–46.
Grossman D, Kim PJ, Schechner JS, Altieri DC. Inhibition of melanoma tumor growth in vivo by survivin targeting. Proc Natl Acad Sci USA. 2001;98:635–40.
Yoshida A, Ookura M, Zokumasu K, Ueda T. Gö6976, a FLT3 kinase inhibitor, exerts potent cytotoxic activity against acute leukemia via inhibition of survivin and MCL-1. Biochemical Pharmacol. 2014;90:16–24.
Kamran M, Long Z, Xu D, Lv S, Liu B, Wang C, et al. Aurora kinase A regulates survivin stability through targeting FBXL7 in gastric cancer drug resistance and prognosis. Oncogenesis. 2017;6:e298.
Shin S, Sung B-J, Cho Y-S, Kim H-J, Ha N-C, Hwang J-I, et al. An anti-apoptotic protein human survivin is a direct inhibitor of caspase-3 and-7. Biochemistry. 2001;40:1117–23.
Smith CC, Lin K, Stecula A, Sali A, Shah NP. FLT3 D835 mutations confer differential resistance to type II FLT3 inhibitors. Leukemia. 2015;29:2390–2.
Alvarado Y, Kantarjian HM, Luthra R, Ravandi F, Borthakur G, Garcia‐Manero G, et al. Treatment with FLT3 inhibitor in patients with FLT3‐mutated acute myeloid leukemia is associated with development of secondary FLT3–tyrosine kinase domain mutations. Cancer. 2014;120:2142–9.
Acknowledgements
We thank Dr. Safee Ullah Chaudhary and Dr. Ahmad Jawad Afzal for carefully reading the manuscript and providing useful insights.
Funding
This study was supported by generous grants from Lahore University of Management Sciences to Amir Faisal (Faculty Initiative Fund-253 and Startup Grant STG-064).
Author information
Authors and Affiliations
Contributions
AF, VB and SL conceived and designed the study with MUT providing additional input. AF, MUT, MF, HP, RU and MM performed all the experiments. MUT, HP and AF wrote the manuscript. RSZS, VB and SL helped in writing and reviewing the manuscript. MF helped in the revision of the manuscript. AF supervised the whole study. All the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Peripheral blood mononuclear cells (PBMCs) were isolated from blood taken from consenting healthy volunteers after approval from Institutional Review Board.
Consent to publish
Not applicable.
Competing interests
Some of the authors are current (VB) or former (SL and AF) employees of The Institute of Cancer Research which operates a rewards to inventors scheme applicable to all current and former employees. For all the other authors, there is no financial or commercial conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Tariq, M.U., Furqan, M., Parveen, H. et al. CCT245718, a dual FLT3/Aurora A inhibitor overcomes D835Y-mediated resistance to FLT3 inhibitors in acute myeloid leukaemia cells. Br J Cancer 125, 966–974 (2021). https://doi.org/10.1038/s41416-021-01527-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41416-021-01527-2