Abstract
Background
Epidemiological studies and meta-analyses show an association between statin use and a reduced incidence of colorectal cancer (CRC). We have shown that statins act on CRC through bone morphogenetic protein (BMP) signalling, but the exact cellular targets and underlying mechanism of statin action remain elusive. In this study, we set out to assess the influence of statins on global cancer cell signalling by performing an array-based kinase assay using immobilised kinase substrates spanning the entire human kinome.
Methods
CRC cells with or without Lovastatin treatment were used for kinome analysis. Findings on kinome arrays were further confirmed by immunoblotting with activity-specific antibodies. Experiments in different CRC cell lines using immunoblotting, siRNA-mediated knockdown and treatment with specific BMP inhibitor Noggin were performed. The relevance of in vitro findings was confirmed in xenografts and in CRC patients treated with Simvastatin.
Results
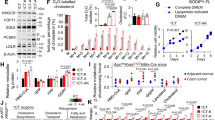
Kinome analysis can distinguish between non-specific, toxic effects caused by 10 µM of Lovastatin and specific effects on cell signalling caused by 2 µM Lovastatin. Statins induce upregulation of PTEN activity leading to downregulation of the PI3K/Akt/mTOR signalling. Treatment of cells with the specific BMP inhibitor Noggin as well as PTEN knockdown and transfection of cells with a constitutively active form of AKT abolishes the effect of Lovastatin on mTOR phosphorylation. Experiments in xenografts and in patients treated with Simvastatin confirm statin-mediated BMP pathway activation, activation of PTEN and downregulation of mTOR signalling.
Conclusions
Statins induce BMP-specific activation of PTEN and inhibition of PI3K/Akt/mTOR signalling in CRC.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Change history
10 May 2021
A Correction to this paper has been published: https://doi.org/10.1038/s41416-021-01426-6
References
Bardou, M., Barkun, A. & Martel, M. Effect of statin therapy on colorectal cancer. Gut 59, 1572–1585 (2010).
Segnan, N., Senore, C., Andreoni, B., Arrigoni, A., Bisanti, L., Cardelli, A. et al. Randomized trial of different screening strategies for colorectal cancer: patient response and detection rates. J. Natl Cancer Inst. 97, 347–357 (2005).
U. S. Preventive Services Task Force, Bibbins-Domingo, K., Grossman, D. C., Curry, S. J., Davidson, K. W., Epling, J. W. et al. Screening for colorectal cancer: US Preventive Services Task Force Recommendation Statement. JAMA 315, 2564–2575 (2016).
Doubeni, C. A., Corley, D. A., Quinn, V. P., Jensen, C. D., Zauber, A. G., Goodman, M. et al. Effectiveness of screening colonoscopy in reducing the risk of death from right and left colon cancer: a large community-based study. Gut 67, 291–298 (2018).
Jacobs, R. J., Kodach, L. L. & Hardwick, J. C. H. The potential of statins for individualized colorectal cancer chemoprevention. Curr. Drug Targets 12, 1903–1908 (2011).
Arber, N. & Levin, B. Chemoprevention of colorectal neoplasia: the potential for personalized medicine. Gastroenterology 134, 1224–1237 (2008).
Poynter, J. N., Gruber, S. B., Higgins, P. D. R., Almog, R., Bonner, J. D., Rennert, H. S. et al. Statins and the risk of colorectal cancer. N. Engl. J. Med. 352, 2184–2192 (2005).
Graaf, M. R., Beiderbeck, A. B., Egberts, A. C. G., Richel, D. J. & Guchelaar, H.-J. The risk of cancer in users of statins. J. Clin. Oncol. 22, 2388–2394 (2004).
Mamtani, R., Lewis, J. D., Scott, F. I., Ahmad, T., Goldberg, D. S., Datta, J. et al. Disentangling the association between statins, cholesterol, and colorectal cancer: a nested case-control study. PLoS Med. 13, e1002007 (2016).
Lytras, T., Nikolopoulos, G. & Bonovas, S. Statins and the risk of colorectal cancer: an updated systematic review and meta-analysis of 40 studies. World J. Gastroenterol. 20, 1858–1870 (2014).
Nissen, S. E., Tuzcu, E. M., Schoenhagen, P., Crowe, T., Sasiela, W. J., Tsai, J. et al. Statin therapy, LDL cholesterol, C-reactive protein, and coronary artery disease. N. Engl. J. Med. 352, 29–38 (2005).
Downs, J. R., Clearfield, M., Weis, S., Whitney, E., Shapiro, D. R., Beere, P. A. et al. Primary prevention of acute coronary events with lovastatin in men and women with average cholesterol levels: results of AFCAPS/TexCAPS. Air Force/Texas Coronary Atherosclerosis Prevention Study. JAMA 279, 1615–1622 (1998).
Egan, A. & Colman, E. Weighing the benefits of high-dose simvastatin against the risk of myopathy. N. Engl. J. Med. 365, 285–287 (2011).
Gazzerro, P., Proto, M. C., Gangemi, G., Malfitano, A. M., Ciaglia, E., Pisanti, S. et al. Pharmacological actions of statins: a critical appraisal in the management of cancer. Pharmacol. Rev. 64, 102–146 (2012).
Kodach, L. L., Bleuming, S. A., Peppelenbosch, M. P., Hommes, D. W., van den Brink, G. R. & Hardwick, J. C. H. The effect of statins in colorectal cancer is mediated through the bone morphogenetic protein pathway. Gastroenterology 133, 1272–1281 (2007).
Kodach, L. L., Jacobs, R. J., Voorneveld, P. W., Wildenberg, M. E., Verspaget, H. W., van Wezel, T. et al. Statins augment the chemosensitivity of colorectal cancer cells inducing epigenetic reprogramming and reducing colorectal cancer cell “stemness” via the bone morphogenetic protein pathway. Gut 60, 1544–1553 (2011).
Voorneveld, P. W., Reimers, M. S., Bastiaannet, E., Jacobs, R. J., van Eijk, R., Zanders, M. M. J. et al. Statin use after diagnosis of colon cancer and patient survival. Gastroenterology 153, 470.e4–479.e4 (2017).
Demierre, M.-F., Higgins, P. D. R., Gruber, S. B., Hawk, E. & Lippman, S. M. Statins and cancer prevention. Nat. Rev. Cancer 5, 930–942 (2005).
Guruswamy, S. & Rao, C. V. Multi-target approaches in colon cancer chemoprevention based on systems biology of tumor cell-signaling. Gene Regul. Syst. Biol. 2, 163–176 (2008).
Johnson, S. A. & Hunter, T. Kinomics: methods for deciphering the kinome. Nat. Methods 2, 17–25 (2005).
Hazen, A. L., Diks, S. H., Wahle, J. A., Fuhler, G. M., Peppelenbosch, M. P. & Kerr, W. G. Major remodelling of the murine stem cell kinome following differentiation in the hematopoietic compartment. J. Proteome Res. 10, 3542–3550 (2011).
Diks, S. H., Kok, K., O’Toole, T., Hommes, D. W., van Dijken, P., Joore, J. et al. Kinome profiling for studying lipopolysaccharide signal transduction in human peripheral blood mononuclear cells. J. Biol. Chem. 279, 49206–49213 (2004).
Wu, D., Sylvester, J. E., Parker, L. L., Zhou, G. & Kron, S. J. Peptide reporters of kinase activity in whole cell lysates. Biopolymers 94, 475–486 (2010).
Löwenberg, M., Tuynman, J., Scheffer, M., Verhaar, A., Vermeulen, L., van Deventer, S. et al. Kinome analysis reveals nongenomic glucocorticoid receptor-dependent inhibition of insulin signaling. Endocrinology 147, 3555–3562 (2006).
Johnson, S. C. Hierarchical clustering schemes. Psychometrika 32, 241–254 (1967).
Abramoff, M., Magalhaes, P. & Ram, S. Image processing with ImageJ. Biophotonics Int. 11, 36–42 (2004).
Agarwal, B., Halmos, B., Feoktistov, A. S., Protiva, P., Ramey, W. G., Chen, M. et al. Mechanism of lovastatin-induced apoptosis in intestinal epithelial cells. Carcinogenesis 23, 521–528 (2002).
Savas, S., Azorsa, D. O., Jarjanazi, H., Ibrahim-Zada, I., Gonzales, I. M., Arora, S. et al. NCI60 cancer cell line panel data and RNAi analysis help identify EAF2 as a modulator of simvastatin and lovastatin response in HCT-116 cells. PLoS ONE 6, e18306 (2011).
Cho, S.-J., Kim, J. S., Kim, J. M., Lee, J. Y., Jung, H. C. & Song, I. S. Simvastatin induces apoptosis in human colon cancer cells and in tumor xenografts, and attenuates colitis-associated colon cancer in mice. Int. J. Cancer 123, 951–957 (2008).
Pan, H. Y., DeVault, A. R., Wang-Iverson, D., Ivashkiv, E., Swanson, B. N. & Sugerman, A. A. Comparative pharmacokinetics and pharmacodynamics of pravastatin and lovastatin. J. Clin. Pharmacol. 30, 1128–1135 (1990).
Vazquez, F., Ramaswamy, S., Nakamura, N. & Sellers, W. R. Phosphorylation of the PTEN tail regulates protein stability and function. Mol. Cell Biol. 20, 5010–5018 (2000).
Rahdar, M., Inoue, T., Meyer, T., Zhang, J., Vazquez, F. & Devreotes, P. N. A phosphorylation-dependent intramolecular interaction regulates the membrane association and activity of the tumor suppressor PTEN. Proc. Natl Acad. Sci. USA 106, 480–485 (2009).
Ross, A. H. & Gericke, A. Phosphorylation keeps PTEN phosphatase closed for business. Proc. Natl Acad. Sci. USA 106, 1297–1298 (2009).
Burgering, B. M. & Coffer, P. J. Protein kinase B (c-Akt) in phosphatidylinositol-3-OH kinase signal transduction. Nature 376, 599–602 (1995).
Howe, J. R., Bair, J. L., Sayed, M. G., Anderson, M. E., Mitros, F. A., Petersen, G. M. et al. Germline mutations of the gene encoding bone morphogenetic protein receptor 1A in juvenile polyposis. Nat. Genet. 28, 184–187 (2001).
Tenesa, A. & Dunlop, M. G. New insights into the aetiology of colorectal cancer from genome-wide association studies. Nat. Rev. Genet. 10, 353–358 (2009).
Zhou, X. P., Woodford-Richens, K., Lehtonen, R., Kurose, K., Aldred, M., Hampel, H. et al. Germline mutations in BMPR1A/ALK3 cause a subset of cases of juvenile polyposis syndrome and of Cowden and Bannayan-Riley-Ruvalcaba syndromes. Am. J. Hum. Genet. 69, 704–711 (2001).
Waite, K. A. & Eng, C. BMP2 exposure results in decreased PTEN protein degradation and increased PTEN levels. Hum. Mol. Genet. 12, 679–684 (2003).
Zimmerman, L. B., De Jesús-Escobar, J. M. & Harland, R. M. The Spemann organizer signal noggin binds and inactivates bone morphogenetic protein 4. Cell 86, 599–606 (1996).
Kobielak, K., Stokes, N., de la Cruz, J., Polak, L. & Fuchs, E. Loss of a quiescent niche but not follicle stem cells in the absence of bone morphogenetic protein signaling. Proc. Natl Acad. Sci. USA 104, 10063–10068 (2007).
He, X. C., Zhang, J., Tong, W.-G., Tawfik, O., Ross, J., Scoville, D. H. et al. BMP signaling inhibits intestinal stem cell self-renewal through suppression of Wnt-beta-catenin signaling. Nat. Genet. 36, 1117–1121 (2004).
Stratton, M. R., Campbell, P. J. & Futreal, P. A. The cancer genome. Nature 458, 719–724 (2009).
Chan, A. T., Ogino, S. & Fuchs, C. S. Aspirin and the risk of colorectal cancer in relation to the expression of COX-2. N. Engl. J. Med. 356, 2131–2142 (2007).
Liao, X., Morikawa, T., Lochhead, P., Imamura, Y., Kuchiba, A., Yamauchi, M. et al. Prognostic role of PIK3CA mutation in colorectal cancer: cohort study and literature review. Clin. Cancer Res. 18, 2257–2268 (2012).
Din, F. V. N., Valanciute, A., Houde, V. P., Zibrova, D., Green, K. A., Sakamoto, K. et al. Aspirin inhibits mTOR signaling, activates AMP-activated protein kinase, and induces autophagy in colorectal cancer cells. Gastroenterology 142, 1504.e3–1515.e3 (2012).
Engelman, J. A., Luo, J. & Cantley, L. C. The evolution of phosphatidylinositol 3-kinases as regulators of growth and metabolism. Nat. Rev. Genet. 7, 606–619 (2006).
Roudier, E., Mistafa, O. & Stenius, U. Statins induce mammalian target of rapamycin (mTOR)-mediated inhibition of Akt signaling and sensitize p53-deficient cells to cytostatic drugs. Mol. Cancer Ther. 5, 2706–2715 (2006).
Holzer, T. R., Fulford, A. D., Arkins, A. M., Grondin, J. M., Mundy, C. W., Nasir, A. et al. Ischemic time impacts biological integrity of phospho-proteins in PI3K/Akt, Erk/MAPK, and p38 MAPK signaling networks. Anticancer Res. 31, 2073–2081 (2011).
Author information
Authors and Affiliations
Contributions
S.O.: designed and performed experiments, analysed data, wrote manuscript. M.P.P., S.D., G.M.F.: analysed kinome array. R.J.J., J.H., M.E.W., G.M.F.: performed experiments. L.J.A.C.H.: advised and facilitated. L.L.K.: designed experiments, analysed data, and supervised the project. P.W.V.: designed and performed experiments, analysed data, and wrote the manuscript. J.C.H.H.: designed experiments, analysed data, wrote manuscript, and supervised the project.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The study was performed in accordance with the Declaration of Helsinki. The patient study was approved by the LUMC Medical Ethics Committee. Patients provided their informed consent for participation. The animal study was approved by the AMC Animal Experiments Committee.
Data availability
Data are available upon request from the corresponding author (p.w.voorneveld@lumc.nl).
Competing interests
The authors declare no competing interests.
Funding information
R.J.J., L.L.K. and J.H. are funded by the Dutch Cancer Society (KWF). P.W.V. and J.H. are funded by the Netherlands Digestive Diseases foundation.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised: Lucas J. A. C. Hawinkels should be Lukas J. A. C. Hawinkels.
Supplementary information
Rights and permissions
About this article
Cite this article
Ouahoud, S., Jacobs, R.J., Peppelenbosch, M.P. et al. Kinome-wide analysis of the effect of statins in colorectal cancer. Br J Cancer 124, 1978–1987 (2021). https://doi.org/10.1038/s41416-021-01318-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41416-021-01318-9