Abstract
Introduction The medical and cosmetic use of botulinum toxin (BTX) is now widespread. With an increased number of clinicians adopting the use of BTX in the management of temporomandibular disorders (TMD) and/or bruxism, as either a standalone treatment or as an adjunct, affirmation is required in regards to whether it has a clinically justifiable position among the current spectrum of available treatment modalities.
Objectives To establish the usefulness of BTX when treating patients with TMD and/or bruxism, and thereby determine whether there may be an appropriate purpose for the prescription of BTX in the management of these patients.
Data sources and data selection A systematic review of the relevant literature was conducted. The literature search was carried out by applying key terms to appropriate data sources (Medline, Embase, Pubmed, Cochrane Central Register of Controlled Trials, and OpenSIGLE). The resultant papers were subjected to inclusion and exclusion criteria, which were then assessed for bias using a framework outlined in the Cochrane Handbook.
Results A total of 11 trials met the inclusion criteria. The primary outcome measure was changes in pain experience in groups that had been treated with BTX, relative to an appropriate control group. Secondary outcomes included changes in the frequency of bruxism events, changes in maximum mouth opening, changes in occlusal force and changes in electromyography (EMG) readings of muscles of mastication.
Conclusion The evidence to support the use of BTX in the management of TMD and/or bruxism is not entirely unequivocal. A number of studies that have met the inclusion criteria have shown promising results and thereby justify further investigation. Given the current evidence, BTX should certainly be considered but due to financial implications and possible side effects, it seems appropriate that conservative options, such as self-management with explanation and physical therapies, should be exhausted first.
Similar content being viewed by others
Key points
-
Describes how the use of botulinum toxin in the management of TMD and bruxism is becoming more widespread.
-
Suggests the current evidence is positive and indicates there is potentially a place for botulinum toxin in the management of these conditions.
-
Highlights primary conservative options such as self-management with explanation should be exhausted first, before botulinum toxin is considered.
Introduction
Temporomandibular disorders (TMD) have been described by the American Association for Dental Research (AADR) as 'a group of musculoskeletal and neuromuscular conditions that involve the temporomandibular joints (TMJ), the masticatory muscles, and all associated tissues'.1 It is believed to be the most common cause of chronic pain in the orofacial region, and third overall after headache and backache.2 TMD encompasses a broad range of disorders and, as there are various classifications in use, it is difficult to assess its prevalence accurately, although it is thought to be in the region of 10-30%.3,4
The aetiology is multifactorial, and it is considered that biopsychosocial factors, including genetics and psychological characteristics, as well as parafunction, occlusion and trauma, have possible roles.2 Of the parafunctions, bruxism is the most common5 and the relationship between bruxism and TMD, despite in some cases being weak, has been widely described.5,6,7,8 Bruxism is described as:
'A repetitive jaw-muscle activity characterised by the clenching or grinding of the teeth and/or by bracing or thrusting of the mandible. Bruxism has two clear circadian manifestations: it can occur during wakefulness (indicated as awake bruxism) or during sleep (indicated as sleep bruxism)'.9
It is estimated that 24% and 16% of the adult population suffer from awake and nocturnal bruxism, respectively.10 It is thought by the World Health Organization (WHO) that there is a large psychogenic component in the aetiology of bruxism,5 with increased muscles of mastication activity and possible associated pain being common.11 Similarly, patients with TMD may present with myogenous clinical manifestations12 and this plays a significant role in Axis I, one of the most commonly used classifications for TMD, the research diagnostic criteria for TMD (RDC/TMD).5,13 Despite the pathophysiological and aetiological complexities involved in TMD and bruxism, a commonality between the two conditions is their potential myogenic involvement resulting in the presentation of clinical signs and symptoms.
Methods of management for TMD and bruxism
Over the years there has been a shift in consensual views regarding the management approach for treatment of TMD and/or bruxism. Previously, there was a tendency to consider invasive dental procedures or surgery, whereas now a greater emphasis is placed on dealing with psychosocial/social factors.14,15 When dental intervention is required, it is kept as conservative as possible and can often involve the use of a full coverage stabilisation splint (Box 1).
A Cochrane review, analysing trials up until June 2001, showed weak evidence to suggest that stabilisation splints may be beneficial in reducing pain in the TMJ against minimal or no treatment.16 In addition, with removable appliances the matter of compliance becomes a factor that can make treatment outcomes unpredictable.17 A Swedish study in 2012 attempted to demonstrate patient adherence to hard acrylic interocclusal appliance treatment. A response from their questionnaire sent to 457 patients showed 73% and 54% used their appliance 1-1.5 years after they had received them from general practice and specialist practice, respectively.18 One of the most common reasons for not using the appliance was shown to be comfort related. Non-compliance has been estimated to be between 30-60% for non-therapeutic regimens,19 and a study by Wig et al. (2004) to assess patient compliance with temporomandibular disorder treatment recommendations over two weeks, showed a median rate of only 54.8%.20
Current uses of botulinum toxin
Botulinum toxin is a safe primary treatment choice for cervical dystonia and also an option for various other muscle-related disorders, including blepharospasm and hemifacial spasm.21 The cosmetic use of BTX is becoming ever more common in the treatment of facial rhytides, where there is strong evidence to show its effectiveness against a placebo, without severe complications.22 BTX is injected intramuscularly and acts on presynaptic cholinergic nerve terminals by blocking the release of acetylcholine, resulting in relaxation of the muscle until the sprouting of new synaptic connections occurs.23 In addition, BTX is shown to block the release of inflammatory mediators, such as substance P and glutamate, creating an antinociceptive effect.24,25 These muscle relaxing and analgesic properties, as well as a reduction in issues relating to compliance, have seen an increase in the number of clinicians using BTX as a treatment modality for myogenous TMD and/or bruxism.
Objective
This systematic review concentrates on determining the usefulness of BTX when treating patients with TMD and/or bruxism to establish whether there may be an appropriate rationale for the prescription of BTX in the management of these patients.
Method
A literature search was carried out by applying key terms, including and relating to botulinum toxin, to appropriate data sources (Medline [MeSH] via OVID, Embase via OVID, Pubmed, Cochrane Central Register of Controlled Trials, and OpenSIGLE). These terms included ('Botulinum toxin' [MeSH] or 'Botox' or 'Botulinum') with relevance to: terms including and relating to temporomandibular disorders ('TMD' or 'Temporomandibular Disorder' or 'Temporomandibular Joint Disorders' [MeSH] or 'Temporomandibular pain dysfunction syndrome' or 'Myofacial pain' or 'Temporomandibular' or 'Temporomandibular Joint Dysfunction Syndrome' [MeSH] or 'Facial Pain' [MeSH] or 'Face Pain') and/or terms including and relating to bruxism ('Bruxism' [MeSH] or 'parafunction' or 'tooth grinding' or 'tooth clenching' or 'bruxist'). These search terms were applied to search dates selected from inception to 15 July 2018. The results were then subjected to the eligibility criteria (Box 2).
Results
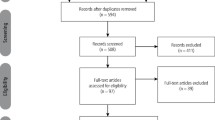
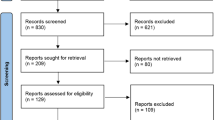
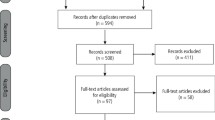
After duplicates were removed, a total of 306 results were yielded which were subsequently subjected to the eligibility criteria (Box 2). This left 11 results that were deemed appropriate for risk of bias analysis (Table 1) and for data collection (Table 2).
Results relating to primary outcome measures
Eight out of the eleven studies used visual analogue scores (VAS) or a pain scale to assess pain.27,28,29,31,32,34,36,37 The test groups were injected with BTX and, in most cases, the controls were injected with saline.28,29,31,32,37 Guarda-Nardini et al.,27 Carli et al.34 and Chaurand et al.36 being the exceptions, with the controls being subjected to a facial manipulation technique, low-level laser and conservative treatments, respectively. For the test groups (those injected with BTX) there was a reduction in scores for all of these studies from baseline to intervals up to and including three months. Guarda-Nardini et al.,27 Ernberg et al.,28 Lindern et al.,29 Nixdorf et al.,31 Guarda-Nardini et al.,32 Carli et al.,34 and Chaurand et al.36 showed a reduction of VAS from 7.3 to 4.8 (baseline to three months on a 0-10 scale), -11% (baseline to one month), 3.2 reduction (baseline to four weeks on a 0-10 scale), 19 mm reduction (baseline to eight weeks on a 0-100 mm scale), 6.2 to 3.6 (baseline to one month on a 0-10 scale), to 3.5 (baseline to 30 days on a 0-10 scale) and 19.2% reduction (baseline to one month), respectively. Patel et al.37 showed a 4.5 point reduction from 5.4 in a pain scale from baseline to one month. Aside from the study by Guarda-Nardini et al.,27 these reductions in VAS were all greater than the relative control groups (Table 2). Kurtoglu et al.30 used a biobehavioural questionnaire relating to RDC/TMD Axis II to assess reduction in pain, which also showed a mean reduction of scores in the test group (56.1 to 43.9 from baseline to 28 days). Again this was a larger difference than the control group (58.9 to 41.4 from baseline to 28 days).
Results relating to secondary outcome measures
There were more variable results regarding maximum mouth opening with Guarda-Nardini et al.27 and Ernberg et al.28 noting improvements, and Nixdorf et al.,31 Guarda-Nardini et al.,32 and Carli et al.34 showing reductions in mouth opening from baseline to their respective intervals (Table 2).
In the study by Lee et al.,33 there was a reduction in the number of EMG bruxism events per hour (using the 20% MVC criterion) in the test group from baseline to 12 weeks from 2.77 ± 1.86 to 0.26 ± 0.24; compared to the control group, which stayed relatively constant in this same time interval 2.77 ± 1.86 to 0.26 ± 0.24. The mean EMG readings by Kurtoglu et al.30 of the anterior temporal muscles and masseters bilaterally at rest and maximal clenching, reduced from 206.3 mV and 296.0 mV to 165.0 mV and 199.0 mV, respectively, after 14 days in the test group; with more variable results in the control group with an increase from 200.0 mV to 252.5 mV at rest and from 529.3 mV to 498.8 mV at maximal clenching for the same time interval. Zhang et al.35 noted a 48.17 kg reduction in mean maximum biting forces in the group injected with BTX after three months, relative to a 13.33 kg and 8.63 kg reduction with the group injected with saline and the one provided with no treatments, respectively.
Discussion
The results from this systematic review seem to indicate that BTX helps to lessen pain levels in those suffering from TMD. Ernberg et al.,28 Lindern et al.,29 Nixdorf et al.,31 Guarda-Nardini et al.,32 Carli et al.,34 Kurtoglu et al.,30 Chaurand et al.36 and Patel et al.,37 all demonstrated reduction in pain in the groups treated with BTX relative to the control. Despite decreases in pain with BTX, the results published by Guarda-Nardini et al.27 showed that fascial manipulation provided greater improvements. A number of the trials that did not meet the inclusion criteria also demonstrated encouraging results for the effectiveness of BTX when treating patients with TMD.38,39,40 The paper by Guarda-Nardini et al.32 was the only study to look at pain as a variable in individuals with both bruxism and TMD. They showed that there was enough positive data to suggest that BTX reduced pain when chewing and at rest.
There was a general lack of trials that met the inclusion criteria when looking at the use of BTX on patients with bruxism. A plausible reason for this is that bruxism does not always result in pain and so, when looking at pain as a variable, many of the patients will fall under the umbrella of TMD. Molina et al. found that the prevalence of bruxism was higher in subjects with TMD (57%) than in controls (37%).41 The results from the papers that did meet the inclusion criteria were favourable.32,33 Lee et al.33 noted that the number of bruxism events reduced with the use of BTX and Kurtoglu et al.30 showed that the EMG of the temporalis and masseter muscles both showed greater reduction during clenching, having been administered with BTX in relation to the control group, implicating force reduction by these muscle groups in the test subjects. Zhang et al.35 looked at participants with both TMD and bruxism; investigating the maximal occlusal forces that can be generated as a variable. The results were clear to show that these forces were significantly lower after treatment with BTX, relative to no treatment or treatment with saline. With these studies demonstrating a reduction in the number of bruxism events and also the level of force, which for bruxists can often be significantly greater than normal functional forces, with validation from further research, BTX could certainly provide a greater level of dental protection against non-treatment in those that brux.
Other research looking at the use of BTX in the management of bruxism, that did not meet the inclusion criteria, also presented some encouraging results. Shim et al. showed that the use of BTX resulted in reduction in the intensity of the contractions in jaw-closing muscles42 and trials by Sener et al.43 and Bolayir et al.44 both resulted in significant reduction in pain. Other studies where bruxism was the result of other conditions, such as brain injuries and autism, also exhibited promising outcomes when treated with BTX.45,46,47,48,49,50,51
Limitations
Despite a number of positive outcomes with the use of BTX, there were certain noteworthy limitations that were evident.
Number of participants
Many of the studies had very low number of subjects with nine of the 11 having less than 31 participants.27,28,30,31,32,33,35,36 With such diminutive numbers of partakers, concerns relating to the reliability of the results could justifiably be raised.
Diagnostic criteria
The RDC/TMD was the most commonly used diagnostic tool for TMD.27,28,30,31,36 However, von Lindern et al.29 used their own criteria. This leads to more inconsistencies in diagnoses between studies, and ultimately makes them harder to compare. If well-recognised criteria, such as the RDC/TMD, were consistently used in future research, this would lead to a greater level of standardisation. The similar principle is applicable to bruxism, as Guarda-Nardini et al.32 used their own criteria and Lee et al.33 used participants who self-reported the issue.
Failed previous treatment
In some of the studies, participants were provided with BTX treatment, where previously other conservative treatments had clearly failed to address the TMD.28,29,30 Therefore it could be argued that these groups of participants had TMDs that were more challenging to treat. By showing how BTX could be effective when other approaches were not, it would therefore be fair to hypothesise that if an average patient with TMD of myogenous origin, with no previous treatment, were to have BTX as a first line option, the results could potentially be even more favourable.
Visual analogue scores
The majority of the studies used VAS to evaluate pain.27,28,29,31,32,34 There are mixed thoughts about the use of VAS when evaluating pain. VAS is a 'single-point measurement in time based on recall of patient to represent their pain. Patient recollection and single measurements are shown to vary considerably and so is not the most reliable or accurate method of evaluation'.52 Others accept the limitations when using VAS to assess pain and appreciate it may be more reliable than other methods. Conti et al. looked at a behaviour rating scale and compared it to the reliability of VAS (a numeric scale) and determined that the best approach, with the greatest validity, to score reproducible pain was via a numeric scale.53
Risk of bias
The risk of bias was assessed using guidance from the Cochrane Handbook (Table 1).54 With not a single paper showing low risk for every domain, this illustrates the necessity for further investigations where these areas of potential bias are addressed to help improve the reliability of the results. Chaurand et al.36 used the same participants for the control and test, with no washout period leading to high levels of bias (Table 1).
Muscles injected
All the studies injected both masseters and temporalis muscle groups aside from three studies in which the masseters alone were injected.28,33,35 By injecting both muscles groups there is potential for a more pronounced difference in outcomes in relation to pre-operative measurements.
Other factors to consider before treatment with BTX
Side effects
There were various side effects reported. All were temporary and some were experienced by participants in both the test and control groups (headaches, tiredness, jaw pains and influenza type symptoms).28 Other side effects experienced by only the groups administered with BTX included minor discomfort when chewing,27 one case of dry mouth,28 and one patient with swallowing difficulty and temporary paralysis of facial expressions.29,30,31,32,33 Patients also experienced temporary zygomaticus major paralysis in the study by Nixdorf et al.31
Financial costs
Unlike other treatment options for TMD and/or bruxism (for example, oral splints and exercises), administration of BTX has the benefit of not requiring daily compliance. However, BTX is an expensive treatment option. The product itself is expensive and maximum effect is usually reached at approximately two weeks, and effectiveness for approximately three to four months; at which point new nerve endings sprout from previously blocked presynaptic cholinergic nerve terminals.55 Repeat treatments would be required at these intervals, resulting in reoccurring and, therefore, cumulative costs.
Relevant aesthetic benefits
Patients who have parafunctional habits, such as bruxism, may also develop hypertrophy of the masseters. This may have aesthetic implications in the form of squaring of the jaw (wide lower third of the face with prominent mandibular angles). This is thought to be an unfeminine characteristic, unlike triangular and heart-shaped faces. The use of BTX helps reduce this effect and can therefore lead to what some may deem as an improved facial aesthetic outcome.56
Conclusion
Although the evidence to support the use of BTX in the treatment of TMD of myogenous origin and bruxism is not entirely unequivocal, there is certainly enough evidence to justify further research into these areas. There are a sufficient number of studies, despite their limitations, showing promising results and degrees of effectiveness. It will be unlikely that participants who have not had previous treatment or no previous treatment failures could be recruited for such studies, but larger study sizes would be more reasonable. Financial viability is an issue with BTX, however unlike the use of oral splints to manage TMD and bruxism, BTX does not pose as much of a compliance issue and can provide potential aesthetic benefit. Considering highly targeted treatment for unilateral/localised myofascial pain/spasms with BTX is likely to be less of a financial burden but again, is likely to incur difficulties in formulating studies with large number of participants.
The multifaceted nature of TMD and bruxism and their actual aetiology cannot be emphasised enough. TMD 'arise[s] from multiple sources and involve[s] complex interactions between psychosocial and biological variables'.57 The WHO recognises teeth grinding (bruxism) in its ICD-10 and describes it as a 'somatoform disorder' under the mental and behavioural disorders sub-classification.5 It is, therefore, important to establish that BTX does not have a role in tackling the underlying aetiology of TMD or bruxism but only in the potential outcomes (for example, pain and bruxism events). Primary conservative options, such as self-management with explanation, should clearly be exhausted first before BTX is considered.
References
American Association for Dental Research. Temporomandibular Disorders (TMD). 2015. Available at http://www.iadr.org/AADR/About-Us/Policy-Statements/Science-Policy/Temporomandibular-Disorders-TMD (accessed April 2019).
Durham J, Aggarwal V, Davies S J et al. Temporomandibular Disorders (TMDs): an update and management guidance for primary care from the UK Specialist Interest Group in Orofacial Pain and TMDs (USOT). 2013. Available at https://www.rcseng.ac.uk/-/media/files/rcs/fds/publications/temporomandibular-disorders-2013.pdf (accessed April 2019).
Dworkin S F, Huggins K H, LeResche L et al. Epidemiology of signs and symptoms in temporomandibular disorders: clinical signs in cases and controls. J Am Dent Assoc 1990; 120: 273-281.
Goulet J P, Lavigne G J, Lund J P. Jaw pain prevalence among Frenchspeaking Canadians in Québec and related symptoms of temporomandibular disorders. J Dent Res 1995; 74: 1738-1744.
Kelleher M, Ray-Chaudhuri A, Khawaja N. Patients' Priorities and Attitudes Towards Their Temporo-Mandibular Disorders. Prim Dent J 2015; 4: 17-21.
Huang G J, LeResche L, Critchlow C W, Martin M D, Drangsholt M T. Risk factors for diagnostic subgroups of painful temporomandibular disorders (TMD). J Dent Res 2002; 81: 284-288.
Macfarlane T V, Grey R J, Kincey J, Worthington H V. Factors associated with the temporomandibular disorder, pain dysfunction syndrome (PDS): Manchester case-control study. Oral Dis 2001; 7: 321-330.
Velly A M, Gornitsky M, Philippe P. Contributing factors to chronic myofascial pain: a case-control study. Pain 2003; 104: 491-499.
Lobbezoo F, Ahlberg J, Glaros A G et al. Bruxism defined and graded: an international consensus. J Oral Rehabil 2013; 40: 2-4.
Lobbezoo F, Ahlberg D, Manfredini D, Winocur E. Review article: are bruxism and the bite causally related? J Oral Rehabil 2012; 39: 489-501.
Guarda-Nardini L, Manfredini D, Salamone M, Salmaso L, Tonello S, Ferronato G. Efficacy of botulinum toxin in treating myofascial pain in bruxers: a controlled placebo pilot study. Cranio 2008; 26: 126-135.
Lyons M F. Current practice in the management of temporomandibular disorders. Dent Update 2008; 35: 314-316: 318.
Schiffman E, Ohrbach R, Truelove E et al. Diagnostic Criteria for Temporomandibular Disorders (DC/TMD) for Clinical and Research Applications: Recommendations of the International RDC/TMD Consortium Network and Orofacial Pain Special Interest Group†. J Oral Facial Pain Headache 2014; 28: 6-27.
Dimitroulis G. The role of surgery in the management of the TMJ: a critical review of the literature. Part 1. Int J Oral Maxillofac Surg 2005; 34: 107-113.
Dimitroulis G. The role of surgery in the management of the TMJ: a critical review of the literature. Part 2. Int J Oral Maxillofac Surg 2005; 34: 231-237.
Al-Ani M Z, Davies S J, Grey R J, Sloan P, Glenny A M. Stabilisation splint therapy for temporomandibular pain dysfunction syndrome. Cochrane Database Syst Rev 2004; CD002778.
Kacer K A, Valiathan M, Narendran S, Hans M G. Retainer wear and compliance in the first 2 years after active orthodontic treatment. Am J Orthod Dentofacial Orthop 2010; 138: 592-598.
Lindfors E, Helkimo M, Magnusson T. Patients' adherence to hard acrylic interocclusal appliance treatment in general dental practice in Sweden. Swed Dent J 2011; 35: 133-142.
Roter D L, Hall J A, Merisca R, Nordstrom B, Cretin D, Svarstad B. Effectiveness of interventions to improve patient compliance: a meta-analysis. Med Care 1998; 36: 1138-1161.
Wig A D, Aaron L A Turner J A, Huggins K H, Truelove E. Short-term clinical outcomes and patient compliance with temporomandibular disorder treatment recommendations. J Orofac Pain 2004; 18: 203-213.
Naumann M, So Y, Argoff C E et al. Assessment: botulinum neurotoxin in the treatment of autonomic disorders and pain (an evidence-based review): report of the Therapeutics and Technology Assessment Subcommittee of the American Academy of Neurology. Neurology 2008; 70: 1707-1714.
Gadhia K, Walmsley A D. Facial aesthetics: is botulinum toxin treatment effective and safe? A systematic review of randomised controlled trials. Br Dent J 2009; 207: E9.
Sellin L C, Thesleff S. Preand post-synaptic actions of botulinum toxin at the rat neuromuscular junction. J Physiol 1981; 317: 487-495.
Aoki K R. Review of a proposed mechanism for the antinociceptive action of botulinum toxin type A. Neurotoxicology 2005; 26: 785-793.
Purkiss J, Welch M, Doward S, Foster K. Capsaicin-stimulated release of substance P from cultured dorsal root ganglion neurons: involvement of two distinct mechanisms. Biochem Pharmacol 2000; 59: 1403-1406.
Scottish Intercollegiate Guidelines Network. Algorithm for classifying study design for questions of effectiveness. Available at https://www.sign.ac.uk/assets/study_design.pdf (accessed April 2019).
Guarda-Nardini L, Stecco A, Stecco C, Masiero S, Manfredini D. Myofascial pain of the jaw muscles: comparison of short-term effectiveness of botulinum toxin injections and fascial manipulation technique. Cranio 2012; 30: 95-102.
Ernberg M, Hedenberg-Magnusson B, List T, Svensson P. Efficacy of botulinum toxin typeA for treatment of persistent myofascial TMD pain: A randomized, controlled, double-blind multicentre study. Pain 2011; 152: 1988-1996.
von Lindern J J, Niederhagen B, Berge S, Appel T. Type A botulinum toxin in the treatment of chronic facial pain associated with masticatory hyperactivity. J Oral Maxillofac Surg 2003; 61: 774-778.
Kurtoglu C, Gur O H, Kurkcu M, Sertdemir Y, Guler-Uysal F, Uysal H. Effect of botulinum toxinA in myofascial pain patients with or without functional disc displacement. J Oral Maxillofac Surg 2008; 66: 1644-1651.
Nixdorf D R, Heo G, Major P W. Randomized controlled trial of botulinum toxin A for chronic myogenous orofacial pain. Pain 2002; 99: 465-473.
Guarda-Nardini L, Manfredini D, Salamone M, Salmaso L, Tonello S, Ferronato G. Efficacy of botulinum toxin in treating myofascial pain in bruxers: a controlled placebo pilot study. Cranio 2008; 26: 126-135.
Lee S J, McCall W D Jr, Kim Y K, Chung S C, Chung J W. Effect of botulinum toxin injection on nocturnal bruxism: a randomized controlled trial. Am J Phys Med Rehabil 2010; 89: 16-23.
De Carli B M, Magro A K, Souza-Silva B N et al. The effect of laser and botulinum toxin in the treatment of myofascial pain and mouth opening: A randomized clinical trial. J Photochem Photobiol B 2016; 159: 120-123.
Zhang L D, Liu Q, Zou D R, Yu L F. Occlusal force characteristics of masseteric muscles after intramuscular injection of botulinum toxin A (BTX A) for treatment of temporomandibular disorder. Br J Oral Maxillofac Surg 2016; 54: 736-740.
Chaurand J, Pacheco-Ruíz L, Orozco-Saldívar H, López-Valdés J Efficacy of botulinum toxin therapy in treatment of myofascial pain. J Oral Sci 2017; 59: 351-356.
Patel A A, Lerner M Z, Blitzer A. IncobotulinumtoxinA Injection for Temporomandibular Joint Disorder: A Randomized Controlled Pilot Study. Ann Otol Rhinol Laryngol 2017; 126: 328-333.
von Lindern J J. Type A Botulinum toxin in the treatment of chronic facial pain associated with temporo-mandibular dysfunction. Acta Neurol Belg 2001; 101: 39-41.
Sidebottom A J, Patel A A, Amin J. Botulinum injection for the management of myofascial pain in the masticatory muscles. A prospective outcome study. Br J Oral Maxillofac Surg 2013; 51: 199-205.
Lee K M, Chow J, Hui E, Li W. Botulinum Toxin Type A Injection for the Management of Myofascial Temporomandibular Pain Disorder. Asian J Oral Maxillofac Surg 2005; 17: 100-103.
Molina O F, dos Santos J Jr, Nelson S J, Grossman E. Prevalence of modalities of headaches and bruxism among patients with craniomandibular disorder. Cranio 1997; 15: 314-325.
Shim Y J, Lee M K, Kato T, Park H U, Heo K, Kim S T. Effects of botulinum toxin on jaw motor events during sleep in sleep bruxism patients: a polysomnographic evaluation. J Clin Sleep Med 2014; 10: 291-298.
Sener H O, Oral N, Keyf F. Intramasseteric botulinum toxin injection is as effective as oral overnight splint in nocturnal bruxism. Cephalalgia 2007; 27: 1191.
Bolayir G, Bolayir E, Coskun A, Özdemir A K, Topaktaş S. Botulinum toxin typeA practice in bruxism cases. Neurol Psychiat Br 2005; 12: 43-45.
Ivanhoe C B, Lai J M, Francisco G E. Bruxism after brain injury: Successful treatment with botulinum toxinA. Arch Phys Med Rehabil 1997; 78: 1272-1273.
Watts M W, Tan E K, Jankovic J: Bruxism and cranialcervical dystonia: Is there a relationship? Cranio 1999; 17: 196-201.
Tan E K, Jankovic J. Treating severe bruxism with botulinum toxin. J Am Dent Assoc 2000; 131: 211-216.
Pidcock F S, Wise J M, Christensen J R. Treatment of severe post-traumatic bruxism with botulinum toxinA: case report. J Oral Maxillofac Surg 2002; 60: 115-117.
See S J, Tan E K. Severe amphetamine-induced bruxism: Treatment with botulinum toxin. Acta Neurol Scand 2003; 107: 161-163.
Nash M C, Ferrell R B, Lombardo M A, Williams R B. Treatment of bruxism in Huntington's disease with botulinum toxin. J Neuropsychiatry Clin Neurosci 2004; 16: 381-382.
Monroy P G, da Fonseca M A. The use of botulinum toxinA in the treatment of severe bruxism in a patient with autism: a case report. Spec Care Dentist 2006; 26: 37-39.
Jensen M P. Validity of self-report and observational measures. In Jensen T S, Turner J A, Wiesenfeld-Hallin Z (eds) Proceedings of the 8th World Congress on Pain. pp 637-661.Seattle, WA: IASP Press, 1997.
Conti P C, de Azevedo L R, de Souza N V, Ferreira F V. Pain measurement in TMD patients: evaluation of precision and sensitivity of different scales. J Oral Rehabil 2001; 28: 534-539.
Higgins J P T, Green S (eds). Cochrane Handbook for Systematic Reviews of Interventions. Version 5.1.0. London: The Cochrane Collaboration, 2011.
Gerwin R. Botulinum toxin treatment of myofascial pain: a critical review of the literature. Curr Pain Headache Rep 2012; 16: 413-422.
Klein F H, Brenner F M, Sato M S, Robert F M, Helmer K A Lower facial remodeling with botulinum toxin type A for the treatment of masseter hypertrophy. An Bras Dermatol 2014; 89: 878-884.
Rollman G B, Gillespie J M. The role of psychosocial factors in temporomandibular disorders. Curr Rev Pain 2000; 4: 71-81.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Patel, J., Cardoso, J. & Mehta, S. A systematic review of botulinum toxin in the management of patients with temporomandibular disorders and bruxism. Br Dent J 226, 667–672 (2019). https://doi.org/10.1038/s41415-019-0257-z
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41415-019-0257-z
This article is cited by
-
Botulinum toxin-A effects on pain, somatosensory and psychosocial features of patients with refractory masticatory myofascial pain: a randomized double-blind clinical trial
Scientific Reports (2024)
-
Effects of different interventions on bruxism: an overview of systematic reviews
Sleep and Breathing (2024)
-
The relationship between myodural bridge, atrophy and hyperplasia of the suboccipital musculature, and cerebrospinal fluid dynamics
Scientific Reports (2023)
-
The dental demolition derby: bruxism and its impact - part 2: early management of bruxism
British Dental Journal (2022)
-
Botulinum toxin A in dentistry and orofacial surgery: an evidence-based review - part 1: therapeutic applications
Evidence-Based Dentistry (2022)