Abstract
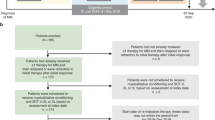
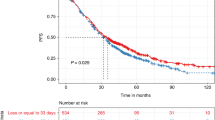
The role of maintenance therapy after high-dose chemotherapy and first autologous transplantation in multiple myeloma (MM) is well established. We explored the effect of maintenance therapy on outcomes after salvage second autologous hematopoietic cell transplant (AHCT2) using the Center for International Blood and Marrow Transplant Research registry. Outcomes of interest included non-relapse mortality (NRM), relapse/progression (REL), progression-free and overall survival (PFS, OS). Of 522 patients who underwent AHCT2 between 2010 and 2018, 342 received maintenance therapy and 180 did not. Maintenance regimens included lenalidomide (42%), pomalidomide (13%), and bortezomib (13%). Median follow up was 58 months in the maintenance group and 61.5 months in the no-maintenance group. Univariate analysis showed superior outcomes at 5 years in maintenance compared to the no-maintenance group: NRM 2 (0.7–3.9)% vs 9.9 (5.9–14.9)%, (p < 0.01), REL 70.2 (64.4–75.8)% vs 80.3 (73.6–86.3)% (p < 0.01), PFS 27.8 (22.4–33.5)% vs. 9.8 (5.5–15.2)% (p < 0.01), and OS 54 (47.5–60.5)% vs 30.9 (23.2–39.2)% (p < 0.01), respectively. Use of maintenance therapy retained its association with improved outcomes in multivariate analysis. There was no difference in second cancers in the two groups (p = 0.39). We conclude that maintenance after AHCT2 is associated with improved 5-year outcomes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
Data use statement
CIBMTR supports accessibility of research in accord with the National Institutes of Health (NIH) Data Sharing Policy and the National Cancer Institute (NCI) Cancer Moonshot Public Access and Data Sharing Policy. The CIBMTR only releases de-identified datasets that comply with all relevant global regulations regarding privacy and confidentiality.
References
Sengsayadeth S, Malard F, Savani BN, Garderet L, Mohty M. Posttransplant maintenance therapy in multiple myeloma: the changing landscape. Blood Cancer J. 2017;7:e545 https://doi.org/10.1038/bcj.2017.23. e-pub ahead of print 2017/03/25
Palumbo A, Cavallo F, Gay F, Di Raimondo F, Ben Yehuda D, Petrucci MT, et al. Autologous transplantation and maintenance therapy in multiple myeloma. N Engl J Med. 2014;371:895–905. https://doi.org/10.1056/NEJMoa1402888. e-pub ahead of print 2014/09/04
Wang Y, Yang F, Shen Y, Zhang W, Wang J, Chang VT, et al. Maintenance therapy with immunomodulatory drugs in multiple myeloma: a meta-analysis and systematic review. J Natl Cancer Inst. 2016;108. https://doi.org/10.1093/jnci/djv342
McCarthy PL, Holstein SA, Petrucci MT, Richardson PG, Hulin C, Tosi P, et al. Lenalidomide maintenance after autologous stem-cell transplantation in newly diagnosed multiple myeloma: a meta-analysis. J Clin Oncol. 2017;35:3279–89. https://doi.org/10.1200/JCO.2017.72.6679. e-pub ahead of print 2017/07/26
Sonneveld P, Schmidt-Wolf IG, van der Holt B, El Jarari L, Bertsch U, Salwender H, et al. Bortezomib induction and maintenance treatment in patients with newly diagnosed multiple myeloma: results of the randomized phase III HOVON-65/ GMMG-HD4 trial. J Clin Oncol: Off J Am Soc Clin Oncol. 2012;30:2946–55. https://doi.org/10.1200/JCO.2011.39.6820
Nooka AK, Kaufman JL, Muppidi S, Langston A, Heffner LT, Gleason C, et al. Consolidation and maintenance therapy with lenalidomide, bortezomib and dexamethasone (RVD) in high-risk myeloma patients. Leukemia. 2014;28:690–3. https://doi.org/10.1038/leu.2013.335. e-pub ahead of print 2013/11/14
Gossi U, Jeker B, Mansouri Taleghani B, Bacher U, Novak U, Betticher D, et al. Prolonged survival after second autologous transplantation and lenalidomide maintenance for salvage treatment of myeloma patients at first relapse after prior autograft. Hematol Oncol. 2018;36:436–44. https://doi.org/10.1002/hon.2490. e-pub ahead of print 2018/01/25
Modi D, Chi J, Kim S, Ayash L, Alavi A, Kin A, et al. Lenalidomide maintenance after second autologous stem cell transplant improves overall survival in multiple myeloma. Leuk Lymphoma. 2020;61:1877–84. https://doi.org/10.1080/10428194.2020.1749603. e-pub ahead of print 2020/04/10
Mohyuddin GR, Okoniewski M, Diab O, Ganguly S, Shune L, Abdallah AO, et al. Maintenance regimens after a second autologous transplant for multiple myeloma. Leuk Lymphoma. 2020:1–3. e-pub ahead of print 2020/11/05; https://doi.org/10.1080/10428194.2020.1842402
Kumar S, Paiva B, Anderson KC, Durie B, Landgren O, Moreau P, et al. International Myeloma Working Group consensus criteria for response and minimal residual disease assessment in multiple myeloma. Lancet Oncol. 2016;17:e328–e346. https://doi.org/10.1016/S1470-2045(16)30206-6. e-pub ahead of print 2016/08/12
Rajkumar SV. Multiple myeloma: 2020 update on diagnosis, risk-stratification and management. Am J Hematol. 2020;95:548–67. https://doi.org/10.1002/ajh.25791. e-pub ahead of print 2020/03/27
Chim CS, Kumar SK, Orlowski RZ, Cook G, Richardson PG, Gertz MA, et al. Management of relapsed and refractory multiple myeloma: novel agents, antibodies, immunotherapies and beyond. Leukemia. 2018;32:252–62. https://doi.org/10.1038/leu.2017.329. e-pub ahead of print 2017/12/20
Fenk R, Liese V, Neubauer F, Bruns I, Kondakci M, Balleisen S, et al. Predictive factors for successful salvage high-dose therapy in patients with multiple myeloma relapsing after autologous blood stem cell transplantation. Leuk Lymphoma. 2011;52:1455–62. https://doi.org/10.3109/10428194.2011.575967. e-pub ahead of print 2011/06/11
Lemieux E, Hulin C, Caillot D, Tardy S, Dorvaux V, Michel J, et al. Autologous stem cell transplantation: an effective salvage therapy in multiple myeloma. Biol Blood Marrow Transpl. 2013;19:445–9. https://doi.org/10.1016/j.bbmt.2012.11.013. e-pub ahead of print 2012/11/29
Dhakal B, D’Souza A, Kleman A, Chhabra S, Mohan M, Hari P. Salvage second transplantation in relapsed multiple myeloma. Leukemia. 2020. e-pub ahead of print 2020/08/05; https://doi.org/10.1038/s41375-020-1005-8
Maclachlan K, Diamond B, Maura F, Hillengass J, Turesson I, Landgren CO, et al. Second malignancies in multiple myeloma; emerging patterns and future directions. Best Pract Res Clin Haematol. 2020;33: 101144. e-pub ahead of print 2020/03/07; https://doi.org/10.1016/j.beha.2020.101144
McCarthy PL, Owzar K, Hofmeister CC, Hurd DD, Hassoun H, Richardson PG, et al. Lenalidomide after stem-cell transplantation for multiple myeloma. N Engl J Med. 2012;366:1770–81. https://doi.org/10.1056/NEJMoa1114083
Huang J, Phillips S, Byrne M, Chinratanalab W, Engelhardt BG, Goodman SA, et al. Lenalidomide vs bortezomib maintenance choice post-autologous hematopoietic cell transplantation for multiple myeloma. Bone Marrow Transpl. 2018;53:701–7. https://doi.org/10.1038/s41409-018-0177-6. e-pub ahead of print 2018/04/29
Acknowledgements
The CIBMTR is supported primarily by Public Health Service U24CA076518 from the National Cancer Institute (NCI), the National Heart, Lung and Blood Institute (NHLBI), and the National Institute of Allergy and Infectious Diseases (NIAID); HHSH250201700006C from the Health Resources and Services Administration (HRSA); and N00014-20-1-2705 and N00014-20-1-2832 from the Office of Naval Research; Support is also provided by Be the Match Foundation, the Medical College of Wisconsin, the National Marrow Donor Program, and from the following commercial entities: AbbVie; Accenture; Actinium Pharmaceuticals, Inc.; Adaptive Biotechnologies Corporation; Adienne SA; Allovir, Inc.; Amgen, Inc.; Astellas Pharma US; bluebird bio, inc.; Bristol Myers Squibb Co.; CareDx; CSL Behring; CytoSen Therapeutics, Inc.; Daiichi Sankyo Co., Ltd.; Eurofins Viracor; ExcellThera; Fate Therapeutics; Gamida-Cell, Ltd.; Genentech Inc; Gilead; GlaxoSmithKline; Incyte Corporation; Janssen/Johnson & Johnson; Jasper Therapeutics; Jazz Pharmaceuticals, Inc.; Karyopharm Therapeutics; Kiadis Pharma; Kite, a Gilead Company; Kyowa Kirin; Magenta Therapeutics; Medac GmbH; Merck & Co.; Millennium, the Takeda Oncology Co.; Miltenyi Biotec, Inc.; MorphoSys; Novartis Pharmaceuticals Corporation; Omeros Corporation; Oncopeptides, Inc.; Orca Biosystems, Inc.; Pfizer, Inc.; Pharmacyclics, LLC; Sanofi Genzyme; Seagen, Inc.; Stemcyte; Takeda Pharmaceuticals; Tscan; Vertex; Vor Biopharma; Xenikos BV. Anita D’Souza is supported by K23 HL141445 grant from the NHLBI.
Author information
Authors and Affiliations
Contributions
Protocol development—OP, MY, AD. Reviewed protocol: MY, UR, LS, AA, RB, NB, RG, PH, LH, NH, HL, CL, HM, KM, SN, AN, TN, RP, SP, MS, SU, TW, BW, NE, RF, SK, NS, MQ. Data cleanup: NE, AD Data analysis: NE, RF, AD. Results review: OP, NE, RF, SK, NS, MQ, AD. Wrote the first draft: OP, AD Reviewed and edited paper: MY, UR, LS, AA, RB, NB, RG, PH, LH, NH, HL, CL, HM, KM, SN, AN, TN, RP, SP, MS, SU, TW, BW, NE, RF, SK, NS, MQ. All authors approved the final submitted draft.
Corresponding author
Ethics declarations
Competing interests
Anita D’Souza reports institutional research funding from Sanofi, TeneoBio and Takeda; Consulting fees from Janssen; 2/2020-current. Rahul Banerjee reports grants or contracts from Pack Health to institution; consulting fees from SparkCures to self; and leadership or fiduciary role in other board, society, committee or advocacy group, paid or unpaid to ASTCT Content Committee. Robert Peter Gale reports consulting fees with Kite Pharms, Inc., Fusion Pharma, LLC, C Stone Pharmaceuticals; and stock or stock options with Celgene Corporation. Leona Holmberg reports grants to self from Seattle Genetics, Sanofi, Millennium-Takada, Bristol Myers Squibb, Merck, Janssen; and royalties to Up-To-Date. Hillard M. Lazarus reports participation on a Data Safety Monitoring Board or Advisory Board with Bristol Myers Squibb/Celgene DSMB for CAR-T cell therapy in myeloma. Taiga Nishihori reports clinical trial support by Novartis to the institution; and drug supply by Karyopharm to the institution. Mark A. Schroeder reports consulting fees with Equillium Inc 2021; served on the speakers bureau and received honorary as a consultant for this work from Abbvie (May 2019), Merck (May 2019), Takeda (May 2019) and all speakers bureau service terminated in August 2019; and served on advisory boards and received honorary or consultant fees from Amgen (2017), Dova pharmaceuticals (May 2019), Dova pharmaceuticals (May 2019), FlatIron Inc (June 2019), GSK (October 2019), Gilead Sciences Inc., (~2017), Incyte (May 2018), Janssen (December 2020), Novo Nordisk (2018), Partner Therapeutics (November 2018), Pfizer (July 2018), and Sanofi Genzyme (December 2020). Saad Z. Usmani reports grants and/or personal fees from Amgen, Abbvie, BMS, Celgene, MundiPharma, Pharmacyclics, Sanofi, Seattle Genetics, Janssen, Takeda, SkylineDX, grants Merck, GSK, outside the submitted work. Shaji Kumar reports research support for clinical trials paid to institution form Abbvie, Celgene, Janssen, Takeda, Adaptive, KITE, Medimmune/Astra Zeneca, Merck, Novartis, Roche, Sanofi; research support for clincal trials paid to institution for past 36 months from Abbvie, Celgene, Janseen, Takeda, Adaptive, KITE, Medimmune/Astra Zeneca, Merck, Novartis, Roche and Sanofi; advisory board participation, paid to institution from Abbvie, Celgene, Janssen, Takeda, Adaptive, KITE, Medimmune/Astra Zeneca; and independent review committee from Oncopeptides. Muzaffar H. Qazilbash reports participation on advisory board from Oncopeptides and data safety monitoring board for Autolus. Nina Shah reports compensation for research funding from Celgene/BMS, Janssen, Bluebird Bio, Sutro Biopharma, Teneobio, Poseida, Nektar; Advisory role for GSK, Amgen, Indapta Therapeutics, Sanofi, CareDx, Kite, Karyopharm, Oncopeptides; and payments for research funding from Celgene/BMS, Janssen, Bluebird Bio, Sutro Biopharma, Teneobio, Poseida, Nektar.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Pasvolsky, O., Yeshurun, M., Fraser, R. et al. Maintenance therapy after second autologous hematopoietic cell transplantation for multiple myeloma. A CIBMTR analysis. Bone Marrow Transplant 57, 31–37 (2022). https://doi.org/10.1038/s41409-021-01455-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41409-021-01455-y