Abstract
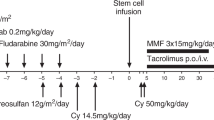
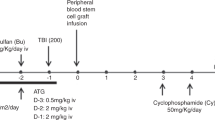
T-cell depletion of an HLA-haploidentical (haplo) graft is often used to reduce the risk of graft-versus-host disease (GVHD), but the lack of donor T cells in the infused product may lead to graft failure, slow T-cell reconstitution, infections, and relapse. More selective T-cell depletion targeting CD45RA can effectively deplete naive T cells but preserve large numbers of memory T cells leading to robust engraftment of diverse T-cell populations and reduction of viremia in the early posttransplant period. Herein, we report the outcome of 143 pediatric and young adult hematologic malignancy patients receiving a first allogeneic hematopoietic cell transplantation (HCT) on six consecutive ex vivo T-cell depleted haploHCT protocols over the past 15 years at a single institution—including the first 50 patients on an active CD45RA-depleted haploHCT study in which patients also received NK-cells and pharmacological GvHD prophylaxis post transplant. Our data demonstrated an increase in the 3-year overall survival and event-free survival in nonchemorefractory recipients receiving CD45RA-depleted grafts (78.9% and 77.7%, respectively) compared with historic T-cell depleted haploHCT cohorts (46.7% and 42.7%, respectively, p = 0.004, and 0.003). This improvement was primarily due to a reduction in transplant related mortality without significant increase in the rates of GVHD.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Oliansky DM, Camitta B, Gaynon P, Nieder ML, Parsons SK, Pulsipher MA, et al. The role of cytotoxic therapy with hematopoietic stem cell transplantation in the treatment of pediatric acute lymphoblastic leukemia: update of the 2005 evidence-based review. ASBMT Position Statement. Biol Blood Marrow Transplant. 2012;18:979–81. https://doi.org/10.1016/j.bbmt.2012.03.011.
Oliansky DM, Rizzo JD, Aplan PD, Arceci RJ, Leone L, Ravindranath Y, et al. The role of cytotoxic therapy with hematopoietic stem cell transplantation in the therapy of acute myeloid leukemia in children: an evidence-based review. Biol Blood Marrow Transplant. 2007;13:1–25. https://doi.org/10.1016/j.bbmt.2006.10.024.
Brissot E, Rialland F, Cahu X, Strullu M, Corradini N, Thomas C, et al. Improvement of overall survival after allogeneic hematopoietic stem cell transplantation for children and adolescents: a three-decade experience of a single institution. Bone Marrow Transplant. 2016;51:267–72. https://doi.org/10.1038/bmt.2015.250. e-pub ahead of print 2015/12/08
Gratwohl A, Brand R, Frassoni F, Rocha V, Niederwieser D, Reusser P, et al. Cause of death after allogeneic haematopoietic stem cell transplantation (HSCT) in early leukaemias: an EBMT analysis of lethal infectious complications and changes over calendar time. Bone Marrow Transplant. 2005;36:757–69. https://doi.org/10.1038/sj.bmt.1705140.
Holtan SG, DeFor TE, Lazaryan A, Bejanyan N, Arora M, Brunstein CG, et al. Composite end point of graft-versus-host disease-free, relapse-free survival after allogeneic hematopoietic cell transplantation. Blood. 2015;125:1333–8. https://doi.org/10.1182/blood-2014-10-609032.
Solh M, Zhang X, Connor K, Brown S, Solomon SR, Morris LE, et al. Factors predicting graft-versus-host disease-free, relapse-free survival after allogeneic hematopoietic cell transplantation: multivariable analysis from a single center. Biol Blood Marrow Transplant. 2016;22:1403–9. https://doi.org/10.1016/j.bbmt.2016.04.006.
Kuhlen M, Willasch AM, Dalle JH, Wachowiak J, Yaniv I, Ifversen M, et al. Outcome of relapse after allogeneic HSCT in children with ALL enrolled in the ALL-SCT 2003/2007 trial. Br J Haematol. 2018;180:82–89. https://doi.org/10.1111/bjh.14965.
Avigan D, Hari P, Battiwalla M, Bishop MR, Giralt SA, Hardy NM, et al. Proceedings from the National Cancer Institute’s Second International Workshop on the biology, prevention, and treatment of relapse after hematopoietic stem cell transplantation: part II. Autologous Transplantation-novel agents and immunomodulatory strategies. Biol Blood Marrow Transplant. 2013;19:1661–9. https://doi.org/10.1016/j.bbmt.2013.08.011.
Gragert L, Eapen M, Williams E, Freeman J, Spellman S, Baitty R, et al. HLA match likelihoods for hematopoietic stem-cell grafts in the U.S. registry. N Engl J Med. 2014;371:339–48. https://doi.org/10.1056/NEJMsa1311707
Lang P, Handgretinger R. Haploidentical SCT in children: an update and future perspectives. Bone Marrow Transpl. 2008;42:S54–59. https://doi.org/10.1038/bmt.2008.285.
Luznik L, O’Donnell PV, Symons HJ, Chen AR, Leffell MS, Zahurak M, et al. HLA-haploidentical bone marrow transplantation for hematologic malignancies using nonmyeloablative conditioning and high-dose, posttransplantation cyclophosphamide. Biol Blood Marrow Transplant. 2008;14:641–50. https://doi.org/10.1016/j.bbmt.2008.03.005.
Leung W, Campana D, Yang J, Pei D, Coustan-Smith E, Gan K, et al. High success rate of hematopoietic cell transplantation regardless of donor source in children with very high-risk leukemia. Blood. 2011;118:223–30. https://doi.org/10.1182/blood-2011-01-333070.
Klingebiel T, Cornish J, Labopin M, Locatelli F, Darbyshire P, Handgretinger R, et al. Results and factors influencing outcome after fully haploidentical hematopoietic stem cell transplantation in children with very high-risk acute lymphoblastic leukemia: impact of center size: an analysis on behalf of the Acute Leukemia and Pediatric Disease Working Parties of the European Blood and Marrow Transplant group. Blood. 2010;115:3437–46. https://doi.org/10.1182/blood-2009-03-207001.
Yanir AD, Martinez CA, Sasa G, Leung K, Gottschalk S, Omer B, et al. Current allogeneic hematopoietic stem cell transplantation for pediatric acute lymphocytic leukemia: success, failure and future perspectives-a single-center experience, 2008 to 2016. Biol Blood Marrow Transplant. 2018;24:1424–31. https://doi.org/10.1016/j.bbmt.2018.03.001.
Federmann B, Bornhauser M, Meisner C, Kordelas L, Beelen DW, Stuhler G, et al. Haploidentical allogeneic hematopoietic cell transplantation in adults using CD3/CD19 depletion and reduced intensity conditioning: a phase II study. Haematologica. 2012;97:1523–31. https://doi.org/10.3324/haematol.2011.059378.
Vadakekolathu J, Rutella S. T-cell manipulation strategies to prevent graft-versus-host disease in haploidentical stem cell transplantation. Biomedicines 2017;5. https://doi.org/10.3390/biomedicines5020033.
Bertaina A, Zecca M, Buldini B, Sacchi N, Algeri M, Saglio F, et al. Unrelated donor vs HLA-haploidentical alpha/beta T-cell- and B-cell-depleted HSCT in children with acute leukemia. Blood. 2018;132:2594–607. https://doi.org/10.1182/blood-2018-07-861575.
Huang W, Chao NJ. Memory T cells: A helpful guard for allogeneic hematopoietic stem cell transplantation without causing graft-versus-host disease. Hematol Oncol Stem Cell Ther. 2017;10:211–9. https://doi.org/10.1016/j.hemonc.2017.05.006.
Teschner D, Distler E, Wehler D, Frey M, Marandiuc D, Langeveld K, et al. Depletion of naive T cells using clinical grade magnetic CD45RA beads: a new approach for GVHD prophylaxis. Bone Marrow Transpl. 2014;49:138–44. https://doi.org/10.1038/bmt.2013.114
Bleakley M, Heimfeld S, Loeb KR, Jones LA, Chaney C, Seropian S, et al. Outcomes of acute leukemia patients transplanted with naive T cell-depleted stem cell grafts. J Clin Investig. 2015. https://doi.org/10.1172/JCI81229.
Sisinni L, Gasior M, de Paz R, Querol S, Bueno D, Fernandez L, et al. Unexpected High Incidence of Human Herpesvirus-6 Encephalitis after Naive T Cell-Depleted Graft of Haploidentical Stem Cell Transplantation in Pediatric Patients. Biol Blood Marrow Transplant. 2018;24:2316–23. https://doi.org/10.1016/j.bbmt.2018.07.016
Shook DR, Triplett BM, Eldridge PW, Kang G, Srinivasan A, Leung W. Haploidentical stem cell transplantation augmented by CD45RA negative lymphocytes provides rapid engraftment and excellent tolerability. Pediatr Blood Cancer. 2015;62:666–73. https://doi.org/10.1002/pbc.25352.
Triplett BM, Muller B, Kang G, Li Y, Cross SJ, Moen J, et al. Selective T-cell depletion targeting CD45RA reduces viremia and enhances early T-cell recovery compared with CD3-targeted T-cell depletion. Transpl Infect Dis. 2018; 20. https://doi.org/10.1111/tid.12823.
Triplett BM, Shook DR, Eldridge P, Li Y, Kang G, Dallas M, et al. Rapid memory T-cell reconstitution recapitulating CD45RA-depleted haploidentical transplant graft content in patients with hematologic malignancies. Bone Marrow Transpl. 2015;50:1012 https://doi.org/10.1038/bmt.2015.139.
Perez-Martinez A, Leung W, Munoz E, Iyengar R, Ramirez M, Vicario JL, et al. KIR-HLA receptor-ligand mismatch associated with a graft-versus-tumor effect in haploidentical stem cell transplantation for pediatric metastatic solid tumors. Pediatr Blood Cancer. 2009;53:120–4. https://doi.org/10.1002/pbc.21955.
Prentice RL, Kalbfleisch JD, Peterson AV Jr., Flournoy N, Farewell VT, Breslow NE. The analysis of failure times in the presence of competing risks. Biometrics. 1978;34:541–54.
Gray RJ. A class of K-sample tests for comparing the cumulative incidence of a competing risk. Ann Stat. 1988;16:1141–54.
Ciurea SO, Zhang MJ, Bacigalupo AA, Bashey A, Appelbaum FR, Aljitawi OS, et al. Haploidentical transplant with posttransplant cyclophosphamide vs matched unrelated donor transplant for acute myeloid leukemia. Blood. 2015;126:1033–40. https://doi.org/10.1182/blood-2015-04-639831.
Yu X, Liu L, Xie Z, Dong C, Zhao L, Zhang J, et al. Bone marrow versus peripheral blood as a graft source for haploidentical donor transplantation in adults using post-transplant cyclophosphamide—a systematic review and meta-analysis. Crit Rev Oncol Hematol. 2019;133:120–8. https://doi.org/10.1016/j.critrevonc.2018.05.017.
Wang Y, Chang YJ, Chen L, Xu LP, Bian ZL, Zhang XH, et al. Low-dose post-transplant cyclophosphamide can mitigate GVHD and enhance the G-CSF/ATG induced GVHD protective activity and improve haploidentical transplant outcomes. Oncoimmunology. 2017;6:e1356152. https://doi.org/10.1080/2162402X.2017.1356152.
Klein OR, Buddenbaum J, Tucker N, Chen AR, Gamper CJ, Loeb D, et al. Nonmyeloablative haploidentical bone marrow transplantation with post-transplantation cyclophosphamide for pediatric and young adult patients with high-risk hematologic malignancies. Biol Blood Marrow Transpl. 2017;23:325–32. https://doi.org/10.1016/j.bbmt.2016.11.016.
Copelan OR, Sanikommu SR, Trivedi JS, Butler C, Ai J, Ragon BK, et al. Higher incidence of hemorrhagic cystitis following haploidentical related donor transplantation compared with matched related donor transplantation. Biol Blood Marrow Transplant. 2018. https://doi.org/10.1016/j.bbmt.2018.12.142.
Mohyuddin GR, Roller J, Shune L, Lin T, Dias A, Ganguly S, et al. Epstein-Barr viremia and post-transplant lymphoproliferative disorders in patients undergoing haploidentical stem cell transplantation with post-transplant cyclophosphamide. Hematol Oncol Stem Cell Ther. 2018. https://doi.org/10.1016/j.hemonc.2018.11.002.
Raiola AM, Dominietto A, di Grazia C, Lamparelli T, Gualandi F, Ibatici A, et al. Unmanipulated haploidentical transplants compared with other alternative donors and matched sibling grafts. Biol Blood Marrow Transpl. 2014;20:1573–9. https://doi.org/10.1016/j.bbmt.2014.05.029
Or-Geva N, Reisner Y. The evolution of T-cell depletion in haploidentical stem-cell transplantation. Br J Haematol. 2016;172:667–84. https://doi.org/10.1111/bjh.13868
Aversa F, Terenzi A, Tabilio A, Falzetti F, Carotti A, Ballanti S, et al. Full haplotype-mismatched hematopoietic stem-cell transplantation: a phase II study in patients with acute leukemia at high risk of relapse. J Clin Oncol. 2005;23:3447–54. https://doi.org/JCO.2005.09.117.
Roux C, Harbi S, Devillier R, Legrand F, Furst S, Granata A, et al. Donor lymphocyte infusion after haploidentical transplantation with post-transplant cyclophosphamide. Blood. 2016;128:5863.
Martelli MF, Ianni MD, Ruggeri L, Falzetti F, Carotti A, Reisner Y, et al. Next generation HLA-haploidentical HSCT. Bone Marrow Transplant. 2015;50:S63–S66. https://doi.org/10.1038/bmt.2015.98
Rujkijyanont P, Morris C, Kang G, Gan K, Hartford C, Triplett B, et al. Risk-adapted donor lymphocyte infusion based on chimerism and donor source in pediatric leukemia. Blood Cancer J. 2013;3:e137. https://doi.org/10.1038/bcj.2013.39.
Locatelli F, Merli P, Li Pira G, Bertaina V, Lucarelli B, Brescia LP, et al. Clinical outcome after adoptive infusion of BPX-501 cells (donor T cells transduced with iC9 suicide gene) in children given alpha/beta T-cell depleted HLA-haploidentical hematopoietic stem cell transplantation (haplo-HSCT): preliminary results of a phase I–II Trial. Blood. 2015;126:1931. https://doi.org/10.1182/blood.V126.23.1931.1931
Di Ianni M, Falzetti F, Carotti A, Terenzi A, Castellino F, Bonifacio E, et al. Tregs prevent GVHD and promote immune reconstitution in HLA-haploidentical transplantation. Blood. 2011;117:3921–8. https://doi.org/10.1182/blood-2010-10-311894
Lang P, Feuchtinger T, Teltschik HM, Schwinger W, Schlegel P, Pfeiffer M, et al. Improved immune recovery after transplantation of TCRalphabeta/CD19-depleted allografts from haploidentical donors in pediatric patients. Bone Marrow Transplant. 2015;50:S6–10. https://doi.org/10.1038/bmt.2015.87
Maschan M, Shelikhova L, Ilushina M, Kurnikova E, Boyakova E, Balashov D, et al. TCR-alpha/beta and CD19 depletion and treosulfan-based conditioning regimen in unrelated and haploidentical transplantation in children with acute myeloid leukemia. Bone Marrow Transplant. 2016;51:668–74. https://doi.org/10.1038/bmt.2015.343.
Zvyagin IV, Mamedov IZ, Tatarinova OV, Komech EA, Kurnikova EE, Boyakova EV, et al. Tracking T-cell immune reconstitution after TCRalphabeta/CD19-depleted hematopoietic cells transplantation in children. Leukemia. 2017;31:1145–53. https://doi.org/10.1038/leu.2016.321.
Locatelli F, Merli P, Pagliara D, Li Pira G, Falco M, Pende D, et al. Outcome of children with acute leukemia given HLA-haploidentical HSCT after alphabeta T-cell and B-cell depletion. Blood. 2017;130:677–85. https://doi.org/10.1182/blood-2017-04-779769
Upadhyayula S, Michaels MG. Ganciclovir, Foscarnet, and Cidofovir: Antiviral Drugs Not Just for Cytomegalovirus. J Pediatr Infect Dis Soc. 2013;2:286–90. https://doi.org/10.1093/jpids/pit048.
Symeonidis N, Jakubowski A, Pierre-Louis S, Jaffe D, Pamer E, Sepkowitz K, et al. Invasive adenoviral infections in T-cell-depleted allogeneic hematopoietic stem cell transplantation: high mortality in the era of cidofovir. Transpl Infect Dis. 2007;9:108–13. https://doi.org/10.1111/j.1399-3062.2006.00184.x.
Chan WK, Suwannasaen D, Throm RE, Li Y, Eldridge PW, Houston J, et al. Chimeric antigen receptor-redirected CD45RA-negative T cells have potent antileukemia and pathogen memory response without graft-versus-host activity. Leukemia. 2015;29:387–95. https://doi.org/10.1038/leu.2014.174.
Acknowledgements
The authors would like to thank our colleagues for data collection and clinical management. Our thanks also go out to the many patients and families who participated in the transplantation and cellular therapy research program. We would like to recognize additional Principle Investigators of the historic T-cell depleted haploidentical donor therapeutic transplant trials initiated at this institution: Rupert Handgretinger, Eli Benaim, and Greg Hale. This work is supported in part by the National Institutes of Health Cancer Center Support (CORE) grant P30 CA021765, and the American Lebanese Syrian Associated Charities (ALSAC).
Author information
Authors and Affiliations
Contributions
Contribution: BMT and WL designed the therapeutic trial. BMT and SG analyzed and interpreted data, BMT and EM wrote the paper; EM, RM, AQ, ASr, AT, ASh, ASul, GM, WL, SG, and BMT provided patient information and data for analysis, contributed to interpretation of the data; GK and ASun provided statistical analysis; and all authors contributed to the revisions of the draft and approval of the final paper.
Corresponding author
Ethics declarations
Conflict of interest
WL is currently an employee of Miltenyi Biotech, all efforts contributing to this work except for final paper review occurred prior to this employment. BMT received travel support from Miltenyi Biotech to present previously published work at EBMT annual meeting 2018. The other authors have no financial relationships or other conflicts of interest to disclose relevant to this paper. No author received an honorarium, grant, or other form of payment to produce the paper.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Mamcarz, E., Madden, R., Qudeimat, A. et al. Improved survival rate in T-cell depleted haploidentical hematopoietic cell transplantation over the last 15 years at a single institution. Bone Marrow Transplant 55, 929–938 (2020). https://doi.org/10.1038/s41409-019-0750-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41409-019-0750-7
This article is cited by
-
Development of a cGMP-compliant process to manufacture donor-derived, CD45RA-depleted memory CD19-CAR T cells
Gene Therapy (2023)
-
Contemporary haploidentical stem cell transplant strategies in children with hematological malignancies
Bone Marrow Transplantation (2021)
-
Association of persistent tachycardia with early myocardial dysfunction in children undergoing allogeneic hematopoietic cell transplantation
Bone Marrow Transplantation (2021)
-
Selective T-cell depletion targeting CD45RA as a novel approach for HLA-mismatched hematopoietic stem cell transplantation in pediatric nonmalignant hematological diseases
International Journal of Hematology (2021)