Abstract
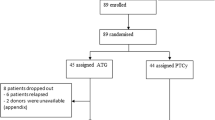
In this multicenter Phase 2 single arm study, we substituted low dose total body irradiation (TBI) for antithymocyte globulin (ATG) in a reduced intensity conditioning regimen with the intent to lower the risk for viral infections after double umbilical cord blood (UCB) transplantation. The conditioning regimen consisted of fludarabine (30 mg/m2/day, Day −7 to −2), melphalan (100 mg/m2/day, Day −1), and TBI (200cGy, Day 0). Graft-versus-host disease prophylaxis was sirolimus and tacrolimus. Thirty-one patients were treated on the protocol. The median time of follow-up for survivors was 24 months (range, 3.3–55.1). Nineteen patients experienced a total of 24 clinically significant viral reactivations or infections, with 1-year cumulative incidence rate of first significant viral event as 64% (95% CI, 43–79%), compared with our historical control of 53%. Within the context of these 24 clinically significant viral reactivations, there were a total of 10 infections with organ involvement. Nonrelapse mortality was 28% (95% CI 13–45%) at 2 years. The 2-year overall and progression-free survivals were 53% (95% CI 33–69%) and 47% (95% CI 28–64%), respectively. In conclusion, the substitution of low dose TBI for ATG did not decrease the incidence of significant viral events after UCB transplantation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Gragert L, Eapen M, Williams E, Freeman J, Spellman S, Baitty R, et al. HLA match likelihoods for hematopoietic stem-cell grafts in the U.S. registry. New Engl J Med. 2014;371:339–48.
Ballen KK, Gluckman E, Broxmeyer HE. Umbilical cord blood transplantation: the first 25 years and beyond. Blood. 2013;122:491–8.
Stevens CE, Carrier C, Carpenter C, Sung D, Scaradavou A. HLA mismatch direction in cord blood transplantation: impact on outcome and implications for cord blood unit selection. Blood. 2011;118:3969–78.
Ballen KK, Spitzer TR, Yeap BY, McAfee S, Dey BR, Attar E, et al. Double unrelated reduced-intensity umbilical cord blood transplantation in adults. Biol Blood Marrow Transplant. 2007;13:82–89.
Cutler C, Stevenson K, Kim HT, Brown J, McDonough S, Herrera M, et al. Double umbilical cord blood transplantation with reduced intensity conditioning and sirolimus-based GVHD prophylaxis. Bone Marrow Transplant. 2011;46:659–67.
Lindemans CA, Chiesa R, Amrolia PJ, Rao K, Nikolajeva O, de Wildt A, et al. Impact of thymoglobulin prior to pediatric unrelated umbilical cord blood transplantation on immune reconstitution and clinical outcome. Blood. 2014;123:126–32.
Pascal L, Tucunduva L, Ruggeri A, Blaise D, Ceballos P, Chevallier P, et al. Impact of ATG-containing reduced-intensity conditioning after single- or double-unit allogeneic cord blood transplantation. Blood. 2015;126:1027–32.
Rashidi A, Ebadi M, Said B, Cao Q, Shanley R, Curtsinger J et al. Absence of early HHV-6 reactivation after cord blood allograft predicts powerful graft-versus-tumor effect. Am J Hematol. 2018;93:1014–9.
Brunstein CG, Weisdorf DJ, DeFor T, Barker JN, Tolar J, van Burik JA, et al. Marked increased risk of Epstein–Barr virus-related complications with the addition of antithymocyte globulin to a nonmyeloablative conditioning prior to unrelated umbilical cord blood transplantation. Blood. 2006;108:2874–80.
Brunstein CG, Barker JN, Weisdorf DJ, DeFor TE, Miller JS, Blazar BR, et al. Umbilical cord blood transplantation after nonmyeloablative conditioning: impact on transplantation outcomes in 110 adults with hematologic disease. Blood. 2007;110:3064–70.
Chen YB, Aldridge J, Kim HT, Ballen KK, Cutler C, Kao G, et al. Reduced-intensity conditioning stem cell transplantation: comparison of double umbilical cord blood and unrelated donor grafts. Biol Blood Marrow Transplant. 2012;18:805–12.
Ruggeri A, Labopin M, Sanz G, Piemontese S, Arcese W, Bacigalupo A, et al. Comparison of outcomes after unrelated cord blood and unmanipulated haploidentical stem cell transplantation in adults with acute leukemia. Leukemia. 2015;29:1891–1900.
Admiraal R, van Kesteren C, Jol-van der Zijde CM, Lankester AC, Bierings MB, Egberts TC, et al. Association between anti-thymocyte globulin exposure and CD4+ immune reconstitution in paediatric haemopoietic cell transplantation: a multicentre, retrospective pharmacodynamic cohort analysis. Lancet Haematol. 2015;2:e194–203.
Montesinos P, Sanz J, Cantero S, Lorenzo I, Martin G, Saavedra S, et al. Incidence, risk factors, and outcome of cytomegalovirus infection and disease in patients receiving prophylaxis with oral valganciclovir or intravenous ganciclovir after umbilical cord blood transplantation. Biol Blood Marrow Transplant. 2009;15:730–40.
Scheurer ME, Pritchett JC, Amirian ES, Zemke NR, Lusso P, Ljungman P. HHV-6 encephalitis in umbilical cord blood transplantation: a systematic review and meta-analysis. Bone Marrow Transplantant. 2013;48:574–80.
Bejanyan N, Rogosheske J, DeFor TE, Lazaryan A, Arora M, Holtan SG, et al. Sirolimus and mycophenolate mofetil as calcineurin inhibitor-free graft-versus-host disease prophylaxis for reduced-intensity conditioning umbilical cord blood transplantation. Biol Blood Marrow Transplant. 2016;22:2025–30.
Jacobson CA, Turki AT, McDonough SM, Stevenson KE, Kim HT, Kao G, et al. Immune reconstitution after double umbilical cord blood stem cell transplantation: comparison with unrelated peripheral blood stem cell transplantation. Biol Blood Marrow Transplant. 2012;18:565–74.
Kennedy VE, Chen H, Savani BN, Greer J, Kassim AA, Engelhardt BG, et al. Optimizing antithymocyte globulin dosing for unrelated donor allogeneic hematopoietic cell transplantation based on recipient absolute lymphocyte count. Biol Blood Marrow Transplant. 2018;24:150–5.
Funding
Amgen Inc. provided clinical trial funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
DeFilipp, Z., Li, S., Avigan, D. et al. A phase II study of reduced intensity double umbilical cord blood transplantation using fludarabine, melphalan, and low dose total body irradiation. Bone Marrow Transplant 55, 804–810 (2020). https://doi.org/10.1038/s41409-019-0715-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41409-019-0715-x
This article is cited by
-
Clinical trials: design, endpoints and interpretation of outcomes
Bone Marrow Transplantation (2022)