Abstract
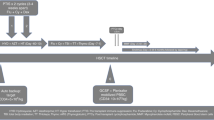
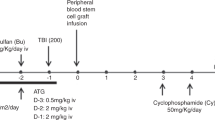
Despite significant improvements in the supportive care, sickle cell disease (SCD) leads to significant morbidity and mortality. Allogeneic hematopoietic stem cell transplantation (HSCT), the only curative option, is limited due to matched donor availability. This could be met with T-cell-depleted haploidentical HSCT. Twenty advanced-stage SCD patients, median age 15 years, and 9 patients, median age 14 years, were transplanted with CD3/CD19- or TCRαβ/CD19-depleted grafts and from matched sibling donors (MSDs). The conditioning consisted of ATG, thiotepa, fludarabine, and treosulfan. The median follow-up in the T-haplo-HSCT and the MSD patients was 21 (9–62) and 25 (7–60) months, respectively. The OS in the T-haplo-HSCT and MSD was 90% and 100%, respectively. In the T-haplo-HSCT group, two patient succumbed to a CMV pneumonitis and a macrophage activation syndrome (MAS). One patient in the T-haplo-HSCT group requires renal replacement therapy because of BK virus nephritis. None developed grade III–IV acute GvHD. In the T-haplo-HSCT and in the MSD, 20% and 22%, respectively, developed a mild or moderate chronic GvHD. These results demonstrate the feasibility, safety, and efficacy of T-haplo-HSCT also for adult advanced stage SCD patients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Gluckman E. Allogeneic transplantation strategies including haploidentical transplantation in sickle cell disease. Hematol Am Soc Hematol Educ Program. 2013;2013:370–6. https://doi.org/10.1182/asheducation-2013.1.370
Ballas SK. Self-management of sickle cell disease: a new frontier. J Natl Med Assoc. 2010;102:1042–3.
Bernaudin F, Socie G, Kuentz M, Chevret S, Duval M, Bertrand Y, et al. Long-term results of related myeloablative stem-cell transplantation to cure sickle cell disease. Blood. 2007;110:2749–56. https://doi.org/10.1182/blood-2007-03-079665.
Gluckman E, Cappelli B, Bernaudin F, Labopin M, Volt F, Carreras J, et al. Sickle cell disease: an international survey of results of HLA-identical sibling hematopoietic stem cell transplantation. Blood. 2017;129:1548–56. https://doi.org/10.1182/blood-2016-10-745711
Walters MC, Patience M, Leisenring W, Eckman JR, Scott JP, Mentzer WC, et al. Bone marrow transplantation for sickle cell disease. New Engl J Med. 1996;335:369–76. https://doi.org/10.1056/NEJM199608083350601.
Justus D, Perez-Albuerne E, Dioguardi J, Jacobsohn D, Abraham A. Allogeneic donor availability for hematopoietic stem cell transplantation in children with sickle cell disease. Pedia Blood Cancer. 2015;62:1285–7. https://doi.org/10.1002/pbc.25439
Gragert L, Eapen M, Williams E, Freeman J, Spellman S, Baitty R, et al. HLA match likelihoods for hematopoietic stem-cell grafts in the U.S. registry. N Engl J Med. 2014;371:339–48. https://doi.org/10.1056/NEJMsa1311707
Shenoy S, Eapen M, Panepinto JA, Logan BR, Wu J, Abraham A, et al. A trial of unrelated donor marrow transplantation for children with severe sickle cell disease. Blood. 2016;128:2561–7. https://doi.org/10.1182/blood-2016-05-715870
Gladstone DE, Zachary A, Fuchs EJ. De-sensitization for mismatched hematopoietic stem cell transplantation (HSCT). Blood. 2011;118:1955.
Bertaina A, Merli P, Rutella S, Pagliara D, Bernardo ME, Masetti R, et al. HLA-haploidentical stem cell transplantation after removal of alphabeta + T and B cells in children with nonmalignant disorders. Blood. 2014;124:822–6. https://doi.org/10.1182/blood-2014-03-563817
Lang P, Teltschik HM, Feuchtinger T, Muller I, Pfeiffer M, Schumm M, et al. Transplantation of CD3/CD19 depleted allografts from haploidentical family donors in paediatric leukaemia. Br J Haematol. 2014;165:688–98. https://doi.org/10.1111/bjh.12810
de la Fuente J, Dhedin N, Koyama T, Bernaudin F, Kuentz M, Karnik L, et al. Haploidentical bone marrow transplant with post-transplant cyclophosphamide plus thiotepa improves donor engraftment in patients with sickle cell anemia: results of an international learning collaborative. Biol Blood Marrow Transplant. 2018. https://doi.org/10.1016/j.bbmt.2018.11.027
Saraf SL, Oh AL, Patel PR, Sweiss K, Koshy M, Campbell-Lee S, et al. Haploidentical peripheral blood stem cell transplantation demonstrates stable engraftment in adults with sickle cell disease. Biol Blood Marrow Transplant. 2018;24:1759–65. https://doi.org/10.1016/j.bbmt.2018.03.031
Bolanos-Meade J, Fuchs EJ, Luznik L, Lanzkron SM, Gamper CJ, Jones RJ, et al. HLA-haploidentical bone marrow transplantation with post-transplant cyclophosphamide expands the donor pool for patients with sickle cell disease. Blood. 2012. https://doi.org/10.1182/blood-2012-07-438408.
Schumm M, Lang P, Bethge W, Faul C, Feuchtinger T, Pfeiffer M, et al. Depletion of T-cell receptor alpha/beta and CD19 positive cells from apheresis products with the CliniMACS device. Cytotherapy. 2013;15:1253–8. https://doi.org/10.1016/j.jcyt.2013.05.014
Stahl T, Bohme MU, Kroger N, Fehse B. Digital PCR to assess hematopoietic chimerism after allogeneic stem cell transplantation. Exp Hematol. 2015;43:462–8 e461. https://doi.org/10.1016/j.exphem.2015.02.006
Payne AB, Mehal JM, Chapman C, Haberling DL, Richardson LC, Bean CJ, et al. Mortality trends and causes of death in persons with sickle cell disease in the United States, 1979-2014. Blood. 2017;130(Suppl 1):865–865.
Lanzkron S, Carroll CP, Haywood C Jr. Mortality rates and age at death from sickle cell disease: U.S., 1979-2005. Public Health Rep. 2013;128:110–6.
Platt OS, Brambilla DJ, Rosse WF, Milner PF, Castro O, Steinberg MH, et al. Mortality in sickle cell disease. Life expectancy and risk factors for early death. N Engl J Med. 1994;330:1639–44. https://doi.org/10.1056/NEJM199406093302303
Quinn CT, Rogers ZR, McCavit TL, Buchanan GR. Improved survival of children and adolescents with sickle cell disease. Blood. 2010;115:3447–52. https://doi.org/10.1182/blood-2009-07-233700
Switzer GE, Bruce JG, Myaskovsky L, DiMartini A, Shellmer D, Confer DL, et al. Race and ethnicity in decisions about unrelated hematopoietic stem cell donation. Blood. 2013;121:1469–76. https://doi.org/10.1182/blood-2012-06-437343
Peters C, Schrappe M, von Stackelberg A, Schrauder A, Bader P, Ebell W, et al. Stem-cell transplantation in children with acute lymphoblastic leukemia: a prospective international multicenter trial comparing sibling donors with matched unrelated donors-The ALL-SCT-BFM-2003 trial. J Clin Oncol. 2015;33:1265–74. https://doi.org/10.1200/JCO.2014.58.9747
Cappelli B, Scigliuolo GM, Volt F, Corbacioglu S, de la Fuente J, Dhedin N, et al. Alternative donor hematopoietic stem cell transplantation for sickle cell disease in Europe. Blood. 2018;132(Suppl 1):4645–4645. https://doi.org/10.1182/blood-2018-99-111129
Luznik L, Fuchs EJ. High-dose, post-transplantation cyclophosphamide to promote graft-host tolerance after allogeneic hematopoietic stem cell transplantation. Immunol Res. 2010;47:65–77. https://doi.org/10.1007/s12026-009-8139-0.
Anurathapan U, Hongeng S, Pakakasama S, Sirachainan N, Songdej D, Chuansumrit A, et al. Hematopoietic stem cell transplantation for homozygous beta-thalassemia and beta-thalassemia/hemoglobin E patients from haploidentical donors. Bone Marrow Transplant. 2016;51:813–8. https://doi.org/10.1038/bmt.2016.7
Katewa S, Kharya G, Karnik L, Kassim AA, de la Fuente J. Pre-transplantation suppression of haemopoiesis is associated with a high rate of macrophage activation syndrome in Ptcy haploidentical transplantation for haemoglobinopathies. Blood. 2017;130(Suppl 1):1931–1931.
Dallas MH, Triplett B, Shook DR, Hartford C, Srinivasan A, Laver J, et al. Long-term outcome and evaluation of organ function in pediatric patients undergoing haploidentical and matched related hematopoietic cell transplantation for sickle cell disease. Biol Blood Marrow Transplant. 2013;19:820–30. https://doi.org/10.1016/j.bbmt.2013.02.010.
Oevermann L, Lang P, Feuchtinger T, Schumm M, Teltschik HM, Schlegel P, et al. Immune reconstitution and strategies for rebuilding the immune system after haploidentical stem cell transplantation. Ann N Y Acad Sci. 2012;1266:161–70. https://doi.org/10.1111/j.1749-6632.2012.06606.x
Gaziev J, Isgro A, Sodani P, Paciaroni K, De Angelis G, Marziali M, et al. Haploidentical HSCT for hemoglobinopathies: improved outcomes with TCRalphabeta( + )/CD19( + )-depleted grafts. Blood Adv. 2018;2:263–70. https://doi.org/10.1182/bloodadvances.2017012005
Shenoy S, Angelucci E, Arnold SD, Baker KS, Bhatia M, Bresters D, et al. Current results and future research priorities in late effects after hematopoietic stem cell transplantation for children with sickle cell disease and thalassemia: a consensus statement from the Second Pediatric Blood and Marrow Transplant Consortium International Conference on Late Effects after Pediatric Hematopoietic Stem Cell Transplantation. Biol Blood Marrow Transplant. 2017;23:552–61. https://doi.org/10.1016/j.bbmt.2017.01.009
Lang P, Feuchtinger T, Teltschik HM, Schwinger W, Schlegel P, Pfeiffer M, et al. Improved immune recovery after transplantation of TCRalphabeta/CD19-depleted allografts from haploidentical donors in pediatric patients. Bone Marrow Transplant. 2015;50(Suppl 2):S6–10. https://doi.org/10.1038/bmt.2015.87
Acknowledgements
We thank the patients and family members. We thank Professor Rubert Handgretinger, Department of General Pediatrics, Oncology, and Hematology, University Hospital Tuebingen, and Professor Matthias Eyrich, Department of Haematology, Oncology, and Stem Cell Transplantation, University Hospital Wuerzburg, for the always excellent preparation of T-cell-depleted peripheral stem cell grafts. We thank Ruediger Eder and Irina Fink from the Central FACS Facility, RCI, University Regensburg, for expert cell sorting.
Author information
Authors and Affiliations
Contributions
JF, JHS, AT, BP, DW, and SC reviewed the literature, analyzed the data, and wrote the manuscript. JF, JHS, AT, BP, PH, AE, CA, ME, PL, DW, EH, and SC were involved in the clinical management of the patients. All authors contributed to the intellectual content of this paper and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Foell, J., Schulte, J.H., Pfirstinger, B. et al. Haploidentical CD3 or α/β T-cell depleted HSCT in advanced stage sickle cell disease. Bone Marrow Transplant 54, 1859–1867 (2019). https://doi.org/10.1038/s41409-019-0550-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41409-019-0550-0
This article is cited by
-
Activity of ex vivo graft and DLI Engineering within the last decade increases, a survey from the EBMT Cellular Therapy & Immunobiology Working Party
Bone Marrow Transplantation (2023)
-
Indications for haematopoietic cell transplantation for haematological diseases, solid tumours and immune disorders: current practice in Europe, 2022
Bone Marrow Transplantation (2022)
-
The role of HLA matching in unrelated donor hematopoietic stem cell transplantation for sickle cell disease in Europe
Bone Marrow Transplantation (2020)