Abstract
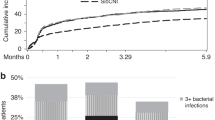
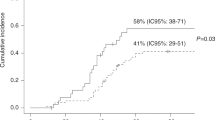
Several approaches have been developed to overcome historical barriers associated with poor outcomes in the setting of HLA-haploidentical allogeneic transplantation (HaploSCT). Here, we examine the outcome of patients with various hematological disorders undergoing HaploSCT with high-dose, post-transplantation cyclophosphamide. We performed a retrospective study on 381 patients from 30 centers between January 2013 and December 2015. At the last follow-up, a total of 1058 infectious episodes were diagnosed, affecting 90.3% of the cohort. Median time to first infection was 13 days for bacterial, 32 days for viral and 20 days for fungal infections. Around 41% of these infections were of bacterial origin and 35% of viral origin, among which 48.8% of patients presented CMV reactivation. Median of GVHD relapse-free survival, progression-free survival and overall survival were 7.1 months, 19.9 months and 33.5 months, respectively. HSCT procedure was the primary or contributing cause of death (55.6%), followed by relapse of the original disease (34.2%). Infections accounted for 45.7% of the HSCT-related deaths. The present multicenter data on a large cohort of patients receiving HaploSCT with PTCy confirmed the feasibility of the procedure with an acceptable incidence of infectious complications, not different as compared to other haploidentical platforms or HLA-matched transplantation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Beatty PG, Clift RA, Mickelson EM, Nisperos BB, Flournoy N, Martin PJ, et al. Marrow transplantation from related donors other than HLA-identical siblings. N Engl J Med. 1985;313:765–71.
Ciurea SO, Mulanovich V, Saliba RM, Bayraktar UD, Jiang Y, Bassett R, et al. Improved early outcomes using a T cell replete graft compared with T cell depleted haploidentical hematopoietic stem cell transplantation. Biol Blood Marrow Transplant. 2012;18:1835–44.
O’Donnell PV, Luznik L, Jones RJ, Vogelsang GB, Leffell MS, Phelps M, et al. Nonmyeloablative bone marrow transplantation from partially HLA-mismatched related donors using posttransplantation cyclophosphamide. Biol Blood Marrow Transplant. 2002;8:377–86.
Luznik L, O’Donnell PV, Symons HJ, Chen AR, Leffell MS, Zahurak M, et al. HLA-haploidentical bone marrow transplantation for hematologic malignancies using nonmyeloablative conditioning and high-dose, posttransplantation cyclophosphamide. Biol Blood Marrow Transplant. 2008;14:641–50.
Cho B-S, Yoon J-H, Shin S-H, Yahng S-A, Lee S-E, Eom K-S, et al. Comparison of allogeneic stem cell transplantation from familial-mismatched/haploidentical donors and from unrelated donors in adults with high-risk acute myelogenous leukemia. Biol Blood Marrow Transplant. 2012;18:1552–63.
Solomon SR, Sizemore CA, Sanacore M, Zhang X, Brown S, Holland HK, et al. Haploidentical transplantation using T cell replete peripheral blood stem cells and myeloablative conditioning in patients with high-risk hematologic malignancies who lack conventional donors is well tolerated and produces excellent relapse-free survival: results of a prospective phase II trial. Biol Blood Marrow Transplant. 2012;18:1859–66.
Raiola AM, Dominietto A, Ghiso A, Di Grazia C, Lamparelli T, Gualandi F, et al. Unmanipulated haploidentical bone marrow transplantation and posttransplantation cyclophosphamide for hematologic malignancies after myeloablative conditioning. Biol Blood Marrow Transplant. 2013;19:117–22.
Sahin U, Toprak SK, Atilla PA, Atilla E, Demirer T. An overview of infectious complications after allogeneic hematopoietic stem cell transplantation. J Infect Chemother. 2016;22:505–14.
Crocchiolo R, Bramanti S, Vai A, Sarina B, Mineri R, Casari E, et al. Infections after T-replete haploidentical transplantation and high-dose cyclophosphamide as graft-versus-host disease prophylaxis. Transpl Infect Dis. 2015;17:242–9.
Slade M, Goldsmith S, Romee R, DiPersio JF, Dubberke ER, Westervelt P et al. Epidemiology of infections following haploidentical peripheral blood hematopoietic cell transplantation. Transpl Infect Dis. 2017;19:1–10.
Castagna L, Crocchiolo R, Furst S, Bramanti S, El Cheikh J, Sarina B, et al. Bone marrow compared with peripheral blood stem cells for haploidentical transplantation with a nonmyeloablative conditioning regimen and post-transplantation cyclophosphamide. Biol Blood Marrow Transplant. 2014;20:724–9.
Brissot E, Alsuliman T, Gruson B, Hermet E, Tirefort Y, Yakoub-Agha I, et al. [How to manage EBV reactivation and EBV-PTLD, CMV and human herpesvirus 6 reactivation and infection after allogeneic stem cell transplantation: A report of the SFGM-TC (update)]. Bull Cancer. 2017;104:S181–S187.
Przepiorka D, Weisdorf D, Martin P, Klingemann HG, Beatty P, Hows J, et al. 1994 Consensus Conference on Acute GVHD Grading. Bone Marrow Transplant. 1995;15:825–8.
Arora M, Klein JP, Weisdorf DJ, Hassebroek A, Flowers MED, Cutler CS, et al. Chronic GVHD risk score: a Center for International Blood and Marrow Transplant Research analysis. Blood. 2011;117:6714–20.
Aversa F, Prezioso L, Manfra I, Galaverna F, Spolzino A, Monti A. Immunity to Infections after Haploidentical Hematopoietic Stem Cell Transplantation. Mediterr J Hematol Infect Dis. 2016;8:e2016057.
Fabricius WA, Ramanathan M. Review on haploidentical hematopoietic cell transplantation in patients with hematologic malignancies. Adv Hematol. 2016;2016:5726132.
Atilla E, Atilla PA, Bozdağ SC, Demirer T. A review of infectious complications after haploidentical hematopoietic stem cell transplantations. Infection. 2017;45:403–11.
Young J-AH, Logan BR, Wu J, Wingard JR, Weisdorf DJ, Mudrick C, et al. Infections after transplantation of bone marrow or peripheral blood stem cells from unrelated donors. Biol Blood Marrow Transplant. 2016;22:359–70.
Mikulska M, Del Bono V, Bruzzi P, Raiola AM, Gualandi F, Van Lint MT, et al. Mortality after bloodstream infections in allogeneic haematopoietic stem cell transplant (HSCT) recipients. Infection. 2012;40:271–8.
Wingard JR, Hsu J, Hiemenz JW. Hematopoietic stem cell transplantation: an overview of infection risks and epidemiology. Infect Dis Clin North Am. 2010;24:257–72.
Mikulska M, Del Bono V, Viscoli C. Bacterial infections in hematopoietic stem cell transplantation recipients. Curr Opin Hematol. 2014;21:451–8.
Valadkhani B, Kargar M, Ashouri A, Hadjibabaie M, Gholami K, Ghavamzadeh A. The risk factors for cytomegalovirus reactivation following stem cell transplantation. J Res Pharm Pract. 2016;5:63–9.
Arthur RR, Shah KV, Charache P, Saral R. BK and JC virus infections in recipients of bone marrow transplants. J Infect Dis. 1988;158:563–9.
Lunde LE, Dasaraju S, Cao Q, Cohn CS, Reding M, Bejanyan N, et al. Hemorrhagic cystitis after allogeneic hematopoietic cell transplantation: risk factors, graft source and survival. Bone Marrow Transplant. 2015;50:1432–7.
Gilis L, Morisset S, Billaud G, Ducastelle-Leprêtre S, Labussière-Wallet H, Nicolini F-E, et al. High burden of BK virus-associated hemorrhagic cystitis in patients undergoing allogeneic hematopoietic stem cell transplantation. Bone Marrow Transplant. 2014;49:664–70.
Silva Lde P, Patah PA, Saliba RM, Szewczyk NA, Gilman L, Neumann J. et al. Hemorrhagic cystitis after allogeneic hematopoietic stem cell transplants is the complex result of BK virus infection, preparative regimen intensity and donor type. Haematologica. 2010;95:1183–90.
Ruggeri A, Roth-Guepin G, Battipaglia G, Mamez A-C, Malard F, Gomez A, et al. Incidence and risk factors for hemorrhagic cystitis in unmanipulated haploidentical transplant recipients. Transpl Infect Dis. 2015;17:822–30.
Girmenia C, Raiola AM, Piciocchi A, Algarotti A, Stanzani M, Cudillo L, et al. Incidence and outcome of invasive fungal diseases after allogeneic stem cell transplantation: a prospective study of the Gruppo Italiano Trapianto Midollo Osseo (GITMO). Biol Blood Marrow Transplant. 2014;20:872–80.
Kontoyiannis DP, Marr KA, Park BJ, Alexander BD, Anaissie EJ, Walsh TJ, et al. Prospective surveillance for invasive fungal infections in hematopoietic stem cell transplant recipients, 2001–6: overview of the Transplant-Associated Infection Surveillance Network (TRANSNET) Database. Clin Infect Dis. 2010;50:1091–1100.
Robinson TM, O’Donnell PV, Fuchs EJ, Luznik L. Haploidentical bone marrow and stem cell transplantation: experience with post-transplantation cyclophosphamide. Semin Hematol. 2016;53:90–7.
Kanakry CG, Fuchs EJ, Luznik L. Modern approaches to HLA-haploidentical blood or marrow transplantation. Nat Rev Clin Oncol. 2016;13:10–24.
Montoro J, Sanz J, Sanz GF, Sanz MA. Advances in haploidentical stem cell transplantation for hematologic malignancies. Leuk Lymphoma. 2016;57:1766–75.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Fayard, A., Daguenet, E., Blaise, D. et al. Evaluation of infectious complications after haploidentical hematopoietic stem cell transplantation with post-transplant cyclophosphamide following reduced-intensity and myeloablative conditioning: a study on behalf of the Francophone Society of Stem Cell Transplantation and Cellular Therapy (SFGM-TC). Bone Marrow Transplant 54, 1586–1594 (2019). https://doi.org/10.1038/s41409-019-0475-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41409-019-0475-7
This article is cited by
-
Post-transplantation cyclophosphamide is associated with increased bacterial infections
Bone Marrow Transplantation (2024)
-
Haploidentical transplant with posttransplant cyclophosphamide vs matched related and unrelated donor transplant in acute myeloid leukemia and myelodysplastic neoplasm
Bone Marrow Transplantation (2023)
-
Patterns of infection and infectious-related mortality in patients receiving post-transplant high dose cyclophosphamide as graft-versus-host-disease prophylaxis: impact of HLA donor matching
Bone Marrow Transplantation (2021)
-
Severe infections and infection-related mortality in a large series of haploidentical hematopoietic stem cell transplantation with post-transplant cyclophosphamide
Bone Marrow Transplantation (2021)
-
Haploidentical Hematopoietic Stem Cell Transplantation in Leukemia’s: Experience from a Cancer Center in India
Indian Journal of Hematology and Blood Transfusion (2021)