Abstract
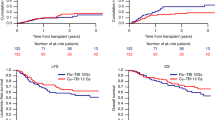
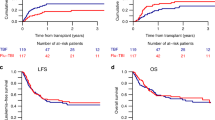
Reduced-intensity conditioning is widely used with hematopoietic stem cell transplantation for non-malignant diseases: however, the optimal conditioning to ensure stable engraftment has not been established. In this study, we retrospectively compared the impact of low-dose (1–6 Gy) irradiation and in vivo T-cell depletion on the clinical outcome of 523 patients with non-malignant disease who underwent a first allogeneic hematopoietic stem cell transplantation using fludarabine-based reduced-intensity conditioning. Use of low-dose irradiation, but not of anti-thymocyte globulin/anti-lymphocyte globulin, showed a beneficial effect on overall survival (adjusted hazard ratio: 0.56; 95% confidence interval: 0.35–0.91, P = 0.018). Furthermore, use of low-dose irradiation was strongly associated with lower transplant-related mortality (adjusted hazard ratio: 0.55; 95% confidence interval: 0.32–0.96, P = 0.034). The addition of low-dose irradiation to the conditioning regimen was beneficial, at least to the short-term clinical outcome. A large prospective study with long-term follow-up is now required to extend these findings and establish the optimal hematopoietic stem cell transplant conditioning for patients with at least some subgroups of non-malignant diseases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Jacobsohn DA, Duerst R, Tse W, Kletzel M. Reduced intensity haemopoietic stem-cell transplantation for treatment of non-malignant diseases in children. Lancet. 2004;364:156–62.
Chiesa R, Veys P. Reduced-intensity conditioning for allogeneic stem cell transplant in primary immune deficiencies. Expert Rev Clin Immunol. 2012;8:255–66.
Kato S, Yabe H, Takakura H, Mugishima H, Ishige M, Tanaka A, et al. Hematopoietic stem cell transplantation for inborn errors of metabolism: a report from the Research Committee on Transplantation for Inborn Errors of Metabolism of the Japanese Ministry of Health, Labour, and Welfare and the Working Group of the Japan Society for Hematopoietic Cell Transplantation. Pediatr Transplant. 2016;20:203–14.
Yabe H, Inoue H, Matsumoto M, Hamanoue S, Koike T, Ishiguro H, et al. Allogeneic haematopoietic cell transplantation from alternative donors with a conditioning regimen of low-dose irradiation, fludarabine and cyclophosphamide in Fanconi anaemia. Br J Haematol. 2006;134:208–12.
Myers KC, Davies SM. Hematopoietic stem cell transplantation for bone marrow failure syndromes in children. Biol Blood Marrow Transplant. 2009;15:279–92.
Marsh RA, Vaughn G, Kim MO, Li D, Jodele S, Joshi S, et al. Reduced-intensity conditioning significantly improves survival of patients with hemophagocytic lymphohistiocytosis undergoing allogeneic hematopoietic cell transplantation. Blood. 2010;116:5824–31.
Umeda K, Adachi S, Tanaka S, Miki M, Okada K, Hashii Y, et al. Comparison of second transplantation and donor lymphocyte infusion for donor mixed chimerism after allogeneic stem cell transplantation for nonmalignant diseases. Pediatr Blood Cancer. 2016;63:2221–9.
Satwani P, Cooper N, Rao K, Veys P, Amrolia P. Reduced intensity conditioning and allogeneic stem cell transplantation in childhood malignant and nonmalignant disease. Bone Marrow Transplant. 2008;41:173–82.
Miyakoshi S, Yuji K, Kami M, Kusumi E, Kishi Y, Kobayashi K, et al. Successful engraftment after reduced-intensity umbilical cord blood transplantation for adult patients with advanced hematological diseases. Clin Cancer Res. 2004;10:3586–92.
Ostronoff F, Milano F, Gooley T, Gutman JA, McSweeney P, Petersen FB, et al. Double umbilical cord blood transplantation in patients with hematologic malignancies using a reduced-intensity preparative regimen without antithymocyte globulin. Bone Marrow Transplant. 2013;48:782–6.
Brunstein CG, Barker JN, Weisdorf DJ, DeFor TE, Miller JS, Blazar BR, et al. Umbilical cord blood transplantation after nonmyeloablative conditioning: impact on transplantation outcomes in 110 adults with hematologic disease. Blood. 2007;110:3064–70.
Howell SJ, Shalet SM. Spermatogenesis after cancer treatment: damage and recovery. J Natl Cancer Monogr Inst. 2005;34:12–17.
Wallace WHB, Thomson AB, Saran F, Kelsey TW. Predicting age of ovarian failure after radiation to a field that includes the ovaries. Int J Rad Oncol Biol Phys. 2005;62:738–44.
Arai Y, Jo T, Matsui H, Kondo T, Takaori-Kondo A. Efficacy of antithymocyte globulin for allogeneic hematopoietic cell transplantation: a systematic review and meta-analysis. Leuk Lymphoma. 2017;58:1840–8.
Finke JF, Bethge WA, Schmoor C, Ottinger HD, Stelljes M, Zander AR, et al. Standard graft-versus-host disease prophylaxis with or without anti-T-cell globulin in haematopoietic cell transplantation from matched unrelated donors: a randomised, open-label, multicenter phase 3 trial. Lancet Oncol. 2009;10:855–64.
Atsuta Y, Suzuki R, Yoshimi A, Gondo H, Tanaka J, Hiraoka A, et al. Unification of hematopoietic stem cell transplantation registries in Japan and establishment of the TRUMP system. Int J Hematol. 2007;86:269–74.
Buckley RH, Schiff SE, Schiff RI, Markert L, Williams LW, Roberts JL, et al. Hematopoietic stem-cell transplantation for the treatment of severe combined immunodeficiency. N Engl J Med. 1999;340:508–16.
Bacigalupo A, Ballen K, Rizzo D, Giralt S, Lazarus H, Ho V, et al. Defining the intensity of conditioning regimens: working definitions. Biol Blood Marrow Transplant. 2009;15:1628–33.
Konopacki J, Porcher R, Robin M, Bieri S, Cayuela JM, Larghero J, et al. Long-term follow up after allogeneic stem cell transplantation in patients with severe aplastic anemia after cyclophosphamide plus antithymocyte globulin conditioning. Haematologica. 2012;97:710–6.
Przepiorka D, Weisdorf D, Martin P, Klingermann HG, Beatty P, Hows J, et al. 1994 Consensus conference on acute GVHD grading. Bone Marrow Transplant. 1995;15:825–8.
Shulman HM, Sullivan KM, Weiden PL, McDonald GB, Strilker GE, Sale GE, et al. A long-term clinicopathologic study of 20 Seattle patients. Am J Med. 1980;69:204–17.
Kanda Y. Investigation of the freely available easy-to-use software ‘EZR’ for medical statistics. Bone Marrow Transplant. 2013;48:452–8.
Socié G, Devergie A, Girinski T, Piel G, Ribaud P, Esperou H, et al. Transplantation for Fanconi’s anaemia: long-term follow-up of 50 patients transplanted from a sibling donor after low-dose cyclophosphamide and thoraco-abdominal irradiation for conditioning. Br J Haematol. 1998;103:249–55.
Alter BP. Cancer in Fanconi Anemia, 1927–2001. Cancer. 2003;97:425–40.
Peffault de Latour R, Porcher R, Dalle JH, Aljurf M, Korthof ET, et al. Allogeneic hematopoietic stem cell transplantation in Fanconi anemia: the European Group for Blood and Marrow Transplantation experience. Blood. 2013;122:4279–86.
Mattsson J, Ringdén O, Storb R. Graft failure after allogeneic hematopoietic cell transplantation. Biol Blood Marrow Transplant. 2008;14:165–70.
Marsh RA, Rao MB, Gefen A, Bellman D, Mehta PA, Khandelwal P, et al. Experience with alemtuzumab, fludarabine, and melphalan reduced-intensity conditioning hematopoietic cell transplantation in patients with nonmalignant diseases reveals good outcomes and that the risk of mixed chimerism depends on underlying disease, stem cell source, and alemtuzumab regimen. Biol Blood Marrow Transplant. 2015;21:1460–70.
Lindemans CA, Chiesa R, Amrolia PJ, Rao K, Nikolajeva O, de Wildt A, et al. Impact of thymoglobulin prior to pediatric unrelated umbilical cord blood transplantation on immune reconstitution and clinical outcome. Blood. 2014;123:126–32.
Fuji S, Ueno N, Hiramoto N, Asakura Y, Yakushijin K, Kamiyama Y, et al. Reduced-intensity conditioning regimen with low-dose ATG-F for unrelated bone marrow transplant is associated with low non-relapse mortality than a regimen with low-dose TBI: a single-center retrospective analysis of 103 cases. Int J Hematol. 2013;98:608–14.
Admiraal R, van Kesteren C, Jol-van der Zijde CM, Lankester AC, Bierings MB, Egberts TC, et al. Association between anti-thymocyte globulin exposure and CD4+ immune reconstitution in paediatric haemopoietic cell transplantation: a multicenter, retrospective phamacodynamic cohort analysis. Lancet Haematol. 2015;2:e194–e203.
Admiraal R, Nierkens S, de Witte MA, Petersen EJ, Fleurke GJ, Verrest L, et al. Association between anti-thymocyte globulin exposure and survival outcomes in adult unrelated haemopoietic cell transplantation: a multicentre, retrospective, pharmacodynamics cohort analysis. Lancet Haematol. 2017;4:e183–e191.
Kanda Y, Mineishi S, Nakai K, Saito T, Tanosaki R, Takaue Y. Frequent detection of rising cytomegalovirus antigenemia after allogeneic stem cell transplantation following a regimen containing antithymocyte globulin. Blood. 2001;97:3676–7.
Brunstein CG, Weisdorf DJ, DeFor T, Barker JN, Tolar J, van Burik JA, et al. Marked increased risk of Epstein-Barr virus-related complications with the addition of antithymocyte globuin to a nonmyeloablative conditioning prior to unrelated umbilical cord blood transplantation. Blood. 2006;108:2874–80.
Rizzo JD, Curtis RE, Socié G, Sobocinski KA, Gilbert E, Landgren O, et al. Solid cancers after allogeneic hematopoietic cell transplantation. Blood. 2009;113:1175–83.
Atsuta Y, Suzuki R, Yamashita T, Fukuda T, Miyamura K, Taniguchi S, et al. Continuing increased risk of oral/esophageal cancer after allogeneic hematopoietic stem cell transplantation in adults in association with chronic graft-versus-host disease. Ann Oncol. 2014;25:435–41.
Mathews V, Savani BN. Conditioning regimens in allo-SCT for thalassemia major. Bone Marrow Transplant. 2014;49:607–10.
Orchard PJ, Fasth AL, Le Rademacher J, He W, Boelens JJ, Horwitz EM, et al. Hematopoietic stem cell transplantation for infantile osteopetrosis. Blood. 2015;126:270–6.
Atta EH, de Sousa AM, Schimer MR, Bouzas LF, Nucci M, Abdelhay E. Different outcome between cyclophosphamide plus horse or rabbit antithymocyte globulin for HLA-identical sibling bone marrow transplant in severe aplastic anemia. Biol Blood Marrow Transplant. 2012;18:1876–82.
Acknowledgements
We thank all the physicians and data managers at the transplant centers who contributed data to the Japan Society for Hematopoietic Cell Transplantation, the Japan Marrow Donor Program, the Japan Cord Blood Bank Network, and the Japanese Society of Pediatric Hematology/Oncology.
Inherited Disease Working Group of the Japan Society for Hematopoietic Cell Transplantation
Katsutsugu Umeda, Hiromasa Yabe, Koji Kato, Kohsuke Imai, Nao Yoshida, Yoji Sasahara, Souichi Adachi, Tomohiro Morio.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Umeda, K., Yabe, H., Kato, K. et al. Impact of low-dose irradiation and in vivo T-cell depletion on hematopoietic stem cell transplantation for non-malignant diseases using fludarabine-based reduced-intensity conditioning. Bone Marrow Transplant 54, 1227–1236 (2019). https://doi.org/10.1038/s41409-018-0418-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41409-018-0418-8
This article is cited by
-
Unresolved issues in allogeneic hematopoietic cell transplantation for non-malignant diseases
International Journal of Hematology (2022)
-
Hematopoietic Cell Transplantation with Reduced Intensity Conditioning Using Fludarabine/Busulfan or Fludarabine/Melphalan for Primary Immunodeficiency Diseases
Journal of Clinical Immunology (2021)
-
Impact of graft-versus-host disease on the clinical outcome of allogeneic hematopoietic stem cell transplantation for non-malignant diseases
International Journal of Hematology (2020)