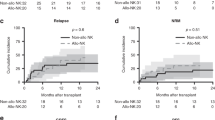
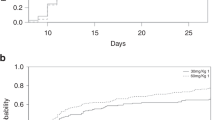
Abstract
HLA-G is a non-classical class I molecule which induces tolerance in allogeneic situations by inhibition of cytotoxic NK and CD8 + T cells and by induction of regulatory T cells. Concordantly, in solid organ transplantation HLA-G is associated with a lower risk for acute and chronic rejection, whereas its role in allogeneic stem cell transplantation (allo-SCT) is less established. We here present detailed analyses of HLA-G-levels in patients after allo-SCT showing a correlation of elevated soluble HLA-G (sHLA-G) levels with less severe acute (p = 0.06) and chronic GvHD (p = 0.0025) and with a superior overall survival (p = 0.03). Soluble HLA-G levels are also positively correlated with the frequency of regulatory T cells in vivo. These clinical data are corroborated by in vitro analyses showing that patients-derived sHLA-G inhibit allogeneic immune responses. ATG-treatment of patients dominantly affects the sHLA-G levels post allo-SCT. Thus, this study highlights the association of elevated sHLA-G levels with less severe acute and chronic GvHD and provides additional functional analyses elucidating possible tolerance-inducing mechanisms of sHLA-G in the context of allo-SCT.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Rebmann V, da Silva Nardi F, Wagner B, Horn PA. HLA-G as a tolerogenic molecule in transplantation and pregnancy. J Immunol Res. 2014;2014:297073.
Verloes A, Van de Velde H, LeMaoult J, Mateizel I, Cauffman G, Horn PA, et al. HLA-G expression in human embryonic stem cells and preimplantation embryos. J Immunol. 2011;186(4):2663–71.
Gonzalez A, Rebmann V, LeMaoult J, Horn PA, Carosella ED, Alegre E. The immunosuppressive molecule HLA-G and its clinical implications. Crit Rev Clin Lab Sci. 2012;49(3):63–84.
Martelli-Palomino G, Pancotto JA, Muniz YC, Mendes-Junior CT, Castelli EC, Massaro JD, et al. Polymorphic sites at the 3’ untranslated region of the HLA-G gene are associated with differential hla-g soluble levels in the Brazilian and French population. PLoS ONE. 2013;8(10):e71742.
Castelli EC, Mendes-Junior CT, Veiga-Castelli LC, Roger M, Moreau P, Donadi EA. A comprehensive study of polymorphic sites along the HLA-G gene: implication for gene regulation and evolution. Mol Biol Evol. 2011;28(11):3069–86.
Hviid TV, Hylenius S, Rorbye C, Nielsen LG. HLA-G allelic variants are associated with differences in the HLA-G mRNA isoform profile and HLA-G mRNA levels. Immunogenetics. 2003;55(2):63–79.
Chen XY, Yan WH, Lin A, Xu HH, Zhang JG, Wang XX. The 14 bp deletion polymorphisms in HLA-G gene play an important role in the expression of soluble HLA-G in plasma. Tissue Antigens. 2008;72(4):335–41.
Hviid TV, Rizzo R, Christiansen OB, Melchiorri L, Lindhard A, Baricordi OR. HLA-G and IL-10 in serum in relation to HLA-G genotype and polymorphisms. Immunogenetics. 2004;56(3):135–41.
Tan Z, Randall G, Fan J, Camoretti-Mercado B, Brockman-Schneider R, Pan L, et al. Allele-specific targeting of microRNAs to HLA-G and risk of asthma. Am J Human Genet. 2007;81(4):829–34.
Veit TD, Chies JA. Tolerance versus immune response—microRNAs as important elements in the regulation of the HLA-G gene expression. Transplant Immunol. 2009;20(4):229–31.
Castelli EC, Moreau P, Oya e Chiromatzo A, Mendes-Junior CT, Veiga-Castelli LC, Yaghi L, et al. In silico analysis of microRNAS targeting the HLA-G 3’ untranslated region alleles and haplotypes. Hum Immunol. 2009;70(12):1020–5.
Carosella ED, Favier B, Rouas-Freiss N, Moreau P, Lemaoult J. Beyond the increasing complexity of the immunomodulatory HLA-G molecule. Blood. 2008;111(10):4862–70.
Carosella ED. The tolerogenic molecule HLA-G. Immunol Lett. 2011;138(1):22–4.
Riteau B, Menier C, Khalil-Daher I, Sedlik C, Dausset J, Rouas-Freiss N, et al. HLA-G inhibits the allogeneic proliferative response. J Reprod Immunol. 1999;43(2):203–11.
Selmani Z, Naji A, Zidi I, Favier B, Gaiffe E, Obert L, et al. Human leukocyte antigen-G5 secretion by human mesenchymal stem cells is required to suppress T lymphocyte and natural killer function and to induce CD4+CD25highFOXP3+regulatory T cells. Stem Cells. 2008;26(1):212–22.
Shi M, Liu ZW, Wang FS. Immunomodulatory properties and therapeutic application of mesenchymal stem cells. Clin Exp Immunol. 2011;164(1):1–8.
Deschaseaux F, Delgado D, Pistoia V, Giuliani M, Morandi F, Durrbach A. HLA-G in organ transplantation: towards clinical applications. Cell Mol Life Sci. 2011;68(3):397–404.
Lila N, Rouas-Freiss N, Dausset J, Carpentier A, Carosella ED. Soluble HLA-G protein secreted by allo-specific CD4+T cells suppresses the allo-proliferative response: a CD4+T cell regulatory mechanism. Proc Natl Acad Sci USA. 2001;98(21):12150–5.
Rebmann V, Bartsch D, Wunsch A, Mollenbeck P, Golda T, Viebahn R, et al. Soluble total human leukocyte antigen class I and human leukocyte antigen-G molecules in kidney and kidney/pancreas transplantation. Hum Immunol. 2009;70(12):995–9.
Le Maux A, Noel G, Birebent B, Grosset JM, Vu N, De Guibert S, et al. Soluble human leucocyte antigen-G molecules in peripheral blood haematopoietic stem cell transplantation: a specific role to prevent acute graft-versus-host disease and a link with regulatory T cells. Clin Exp Immunol. 2008;152(1):50–6.
Liu H, Chen Y, Xuan L, Wu X, Zhang Y, Fan Z, et al. Soluble human leukocyte antigen G molecule expression in allogeneic hematopoietic stem cell transplantation: good predictor of acute graft-versus-host disease. Acta Haematol. 2013;130(3):160–8.
Waterhouse M, Duque-Afonso J, Wasch R, Bertz H, Finke J. Soluble HLA-G molecules and HLA-G 14-base pair polymorphism after allogeneic hematopoietic cell transplantation. Transplant Proc. 2013;45(1):397–401.
Rebmann V, LeMaoult J, Rouas-Freiss N, Carosella ED, Grosse-Wilde H. Quantification and identification of soluble HLA-G isoforms. Tissue Antigens. 2007;69(Suppl 1):143–9.
Hviid TV, Hylenius S, Hoegh AM, Kruse C, Christiansen OB. HLA-G polymorphisms in couples with recurrent spontaneous abortions. Tissue Antigens. 2002;60(2):122–32.
Nuckel H, Castelli EC, Moreau P, Ochsenfarth C, Horn PA, Rebmann V. Simple methods for the detection of HLA-G variants in coding and non-coding regions. Methods Mol Biol. 2012;882:123–42.
Bortolotti D, Gentili V, Melchiorri L, Rotola A, Rizzo R. An accurate and reliable real time SNP genotyping assay for the HLA-G+3142 bp C > G polymorphism. Tissue Antigens. 2012;80(3):259–62.
Zambra FM, Chies JA, Alho CS, Veit TD. Response to Bortolotti et al. 2012--a re-evaluation of our polymerase chain reaction-restriction fragment length polymorphism genotyping method. Tissue Antigens. 2013;82(4):286–7.
Ristich V, Liang S, Zhang W, Wu J, Horuzsko A. Tolerization of dendritic cells by HLA-G. Eur J Immunol. 2005;35(4):1133–42.
Carosella ED, Gregori S, LeMaoult J. The tolerogenic interplay(s) among HLA-G, myeloid APCs, and regulatory cells. Blood. 2011;118(25):6499–505.
Gregori S, Tomasoni D, Pacciani V, Scirpoli M, Battaglia M, Magnani CF, et al. Differentiation of type 1 T regulatory cells (Tr1) by tolerogenic DC-10 requires the IL-10-dependent ILT4/HLA-G pathway. Blood. 2010;116(6):935–44.
Kordelas L, Rebmann V, Ludwig AK, Radtke S, Ruesing J, Doeppner TR, et al. MSC-derived exosomes: a novel tool to treat therapy-refractory graft-versus-host disease. Leukemia. 2014;28(4):970–3.
Sheshgiri R, Gustafsson F, Sheedy J, Rao V, Ross HJ, Delgado DH. Everolimus but not mycophenolate mofetil therapy is associated with soluble HLA-G expression in heart transplant patients. J Heart Lung Transplant. 2009;28(11):1193–7.
Feng X, Kajigaya S, Solomou EE, Keyvanfar K, Xu X, Raghavachari N, et al. Rabbit ATG but not horse ATG promotes expansion of functional CD4+CD25highFOXP3+regulatory T cells in vitro. Blood. 2008;111(7):3675–83.
Moreau P, Adrian-Cabestre F, Menier C, Guiard V, Gourand L, Dausset J, et al. IL-10 selectively induces HLA-G expression in human trophoblasts and monocytes. Int Immunol. 1999;11(5):803–11.
La Nasa G, Littera R, Locatelli F, Lai S, Alba F, Caocci G, et al. The human leucocyte antigen-G 14-basepair polymorphism correlates with graft-versus-host disease in unrelated bone marrow transplantation for thalassaemia. Br J Haematol. 2007;139(2):284–8.
Chiusolo P, Bellesi S, Piccirillo N, Giammarco S, Marietti S, De Ritis D, et al. The role of HLA--G 14-bp polymorphism in allo-HSCT after short-term course MTX for GvHD prophylaxis. Bone Marrow Transplant. 2012;47(1):120–4.
Acknowledgements
This study was supported by the Deutsche José Carreras Leukämie-Stiftung e.V. (DJCLS R 07/06 v). Special thanks go to the patients and donors providing the blood samples for this study. We are grateful for the technical support of Sabine Schramm, Monika Collenburg, Ines Krimphoff, Ursel Hill, and Martina Franke as well as other members of the team of the Institute for Transfusion Medicine and the colleagues from the department of Bone Marrow Transplantation, all University Hospital Essen.
Author contributions
VR and LK conceived, designed and performed the experiments, analyzed the data and wrote the manuscript. FSN, BW, MD, TL, ML, FMH, PAH, and DWB confirmed the analyses and assisted in correcting the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Kordelas, L., da Silva Nardi, F., Wagner, B. et al. Elevated soluble human leukocyte antigen G levels in patients after allogeneic stem cell transplantation are associated with less severe acute and chronic graft-versus-host disease. Bone Marrow Transplant 53, 1149–1156 (2018). https://doi.org/10.1038/s41409-018-0145-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41409-018-0145-1