Abstract
Dezocine, a synthetic opioid, introduced in 1970s as an analgesic, was redeveloped for relieving moderate to severe pain by Yangtze River Pharmaceutical Group in China in 2009. To date, dezocine occupies 45% of China’s opioid analgesic market. Along with dezocine being a dominated painkiller, a certain amount of research was conducted to elucidate dezocine’s action. In this review we summarize the current knowledge on the receptor, preclinical and clinical pharmacology of dezocine. Briefly, preclinical data show that dezocine is effective under varying pain conditions, particularly chronic neuropathic pain and cancer pain, through activation of opioid receptors, and inhibition of norepinephrine reuptake. Clinical data establish the effectiveness of dezocine either as a primary analgesic for postoperative pain management or a supplement for balanced analgesia. The receptor profile of dezocine is different from known pure μ agonists, and allows it to be used in combination with other opioids for additivity in efficacy or lower incidence of adverse effects.
Similar content being viewed by others
Introduction
Acute pain is short-term pain that usually starts suddenly and caused by a possible tissue injury. Chronic pain typically lasts for more than 6 months and is often associated with physical or a combination of physical and mental causes, and the pain continues even after the physical injury has healed [1, 2]. Today, for treatment of acute and moderate to severe chronic pain, opioids are the most commonly used prescribed drugs. Opioids are a class of drugs naturally derived from opium poppy plant, including legally prescription pain relievers morphine, oxycodone, hydrocodone, etc., synthetic opioids fentanyl, tramadol, carfentanil, etc., and illegally heroin. The most of the prescription pain relievers are agonists targeting μ receptors. These opioid drugs produce potent analgesia, but also are highly addictive and can cause respiratory depression and even death [3, 4]. The abuse of these opioids leads to a serious national crisis that presents a significant and ongoing public health burden, is now generally known as the opioid epidemic [5]. It was reported that opioid overdose caused deaths increased nearly ten-fold [6]. Compared with pure full agonists, partial agonists or mixed agonists-antagonists may not be entirely devoid of abuse potential and respiratory depression, but definitely cause less adverse effects. As a well-known mixed agonist-antagonist, dezocine currently is the top 1 painkiller in China, and the sales reaches more than $573 million nationally. It is widely used for postoperative pain and chronic pain management.
Dezocine was redeveloped by Yangtze River Pharmaceutical Group in China and offered as a prescribed analgesic for management of postoperative pain since 2009. The opioid activity and the analgesic effects of dezocine were reviewed previously, with its pharmacokinetic properties and therapeutic trials studies [7]. Marlis’ work was a landmark article, which introduced the pharmacology of dezocine (Wy-16225) and referred dezocine to be a mixed agonist/antagonist with potent analgesic effects. The agonist activity was based on the fact that dezocine produced analgesic effects in the rodent acute pain model [8], and dezocine-induced analgesia was inhibited by naloxone, a nonselective opioid antagonist, whereas the antagonist activity was replied on the finding that dezocine dose-related inhibited morphine-induced loss of righting reflex in rats. As dezocine is marketed in China and becomes dominated, more groups reinvestigate the pharmacodynamic properties of dezocine, which greatly increase the understanding of the basic principle of how dezocine works. Thus, it is critical to have a complete understanding of dezocine pharmacology in order to make well-informed prescribing and therapeutic decisions. This paper is a review of the current stage of knowledge on pharmacology and mechanism of action of dezocine.
Basic pharmacology
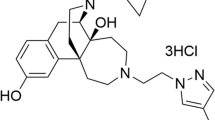
The binding affinities, selectivity, and efficacy of dezocine (Fig. 1) were investigated by in vitro laboratory assays. By using radioligand binding study, Liu et al. did a binding screen for dezocine on variety receptors and transporter proteins and reported that dezocine had Ki values of 3.7 ± 0.7 nM for μ receptors, 31.9 ± 1.9 nM for κ receptors and 527 ± 70 nM for δ receptors [9]. Dezocine’s affinity with μ receptor was ~8 times for κ receptors, 142 times for δ receptors. Increasing evidence also supports that dezocine displays preferential binding to μ receptors than κ and δ receptors [10,11,12,13]. These findings were further confirmed recently by Wang et al. showing that dezocine had Ki values of 1.46 ± 0.10 nM for μ receptors, 22.01 ± 1.52 nM for κ receptors and 398.6 ± 43.25 nM for δ receptors, respectively [14]. Thus, the data from different groups seem consistently supporting that dezocine exhibits higher affinity to μ and κ receptors over δ receptors (Table 1).
However, the intrinsic activity of dezocine seems inconsistent (Table 2). Gharagozlou with colleagues characterized the intrinsic activities of dezocine at different opioid receptors, and reported that dezocine showed agonism at both μ and δ receptors exhibiting inhibitory effect on forskolin-stimulated cAMP production. They also found that dezocine exhibited antagonism at κ receptor, since dezocine was able to antagonize the inhibitory effect of etorphine. Thus, dezocine was classified as a μ agonist, κ antagonist and δ agonist [11,12,13]. Dezocine’s κ antagonist activity was confirmed by Liu et al. by functional [35S]GTPγS binding assay [9]. Liu reported that dezocine did not stimulate κ receptor-mediated G-protein activation when used alone, but was able to inhibit full κ agonist nalbuphine and salvinorin A-induced G-protein activation. However, based on the same assay, Wang et al. evaluated the efficacy and potency of dezocine and reported that dezocine exhibited partial agonist activities at both the κ and μ receptors, with 33%–45% of the maximal stimulation obtained with full κ agonist U50,488H and μ agonist DAMGO. Moreover, dezocine exhibited inhibitory effects on U50,488H and DAMGO-mediated G protein activation [14]. Wang with colleagues further verified the agonist activity of dezocine by in vivo behavioral studies. They found that dezocine-produced significant antinociceptive effects evaluated by animal chemical pain model, and the antinociceptive effects were antagonized by both the κ antagonist nor-BNI and μ antagonist β-FNA, supporting that dezocine had κ and μ agonist activities. When dezocine was coadministered with U50,488H or morphine, it was capable of inhibiting both U50,488H and morphine-induced antinociception, supporting dezocine displayed antagonist activity [14].
In addition to opioid receptors, Liu et al. firstly reported that dezocine had two novel pharmacological targets norepinephrine and serotonin transporter. Dezocine inhibited the norepinephrine and serotonin transporter with pKi of 6.00 ± 0.10 and 6.96 ± 0.08, respectively. In neurotransmitter assay, the pIC50s at NET and SERT were 5.68 ± 0.11 and 5.86 ± 0.17, respectively [9] (Table 1).
Analgesic activities in animals
The analgesic effects of dezocine have been demonstrated in a number of preclinical behavioral paradigms. The literature is reviewed here, particularly focusing on the effects of dezocine on acute and chronic pain with varying experimental conditions on behavioral outcomes [15,16,17,18,19]. The relevant studies are shown in Table 3.
Thermal pain
The hot plate test is a simple behavioral measurement to assess pain sensitivity, and is considered to be supraspinally controlled response [20]. It is based on the principle that when rodents are introduced onto a hot surface, they will perform paw licking or jumping to avoid the thermal stimulus. Marlis et al. reported that in the hot-plate test, dezocine-induced antinociceptive action with ED50 value of 0.06 (0.049–0.081) mg/kg (sc) [8]. However, Wang with colleagues found that in the hot-plate test, 7.5 mg/kg dezocine produced the maximum antinociception around 60%. Neither increasing the dosage nor changing the mode of administration (iv, sc or po) could increase the analgesic potency of dezocine [14].
Both the tail flick and tail withdrawal are well studied spinal reflex, albeit one under the control of supraspinal structures [21]. Intrathecal injection of dezocine (50 μg) can produce analgesic effect in a single injection, and analgesic tolerance does not appear until 1 week of twice daily injection. At 52 °C and 55 °C, the effect of dezocine on the tail contraction phase of mice showed a dose-dependency with ED50 of 0.08 (0.03–0.17) mg/kg and 0.16 (0.1–0.23) mg/kg, respectively [22]. Fischer et al. found that the water temperature is 53 °C, dezocine has a dose-dependent analgesic effect. The ED50 is 0.33 (0.28–0.41) mg/kg, which is 10 times that of morphine, but it does not appear when the water temperature is 56 °C. The analgesic ED50 of morphine at 56 °C is 7.7 (6.2–9.6) mg/kg [23]. When 125 μg of dezocine was administered twice daily, the antinociceptive effect and tolerance development were similar, as with the smaller dose. It was reported that, in the rat tail flick test, dezocine produced analgesic effects with ED50 values of 0.53 (0.37–0.76) mg/kg (ip), 0.12 (0.08–0.17) mg/kg (im), and 2.05 (1.71–2.45) mg/kg (po), respectively. The effect was ~7.7–13 times as potent as morphine [8]. Another study showed that dezocine in dose of 0.625–2.5 μg (it) significantly increased tail withdrawal latency for C57 mice [24]. Huang with colleagues also demonstrated that dezocine increased tail withdrawal latency within the dose range of 0.3125–1.25 μg (it) and produced highest effect around 43% [17].
Mechanical pain
Paw pressure test is used to measure pain response by observing the reaction to gradually increasing pressures on the normal or inflamed paw [25]. Marlis et al. reported that dezocine raised the threshold of both the normal (ED50 1.55 mg/kg, po) and inflamed (ED50 0.53 mg/kg, po) paws in the rat paw pressure test [8]. Barrett et al. found that, across the dose range of 1–100 mg/kg (ip), dezocine increased the paw pressure latency and produced maximal effect of 55% [26].
Chemical pain
The abdominal constriction test is commonly used as a basis for test of analgesic drugs [27]. After 0.6% acetic acid is injected, mice develop acute abdominal pain, which is characterized by abdominal contraction, limb tremor, hind limb extension and S-shaped body. It was reported that dezocine produced a dose-dependent antinociceptive effects, with an ED50 value of 0.2 (0.1–0.2) mg/kg (sc) [14].
Inflammatory pain
Formalin test is a widely used pain model induced by acute tissue injury. Injection of formalin into the hind paw of mice can induce a series of behavioral responses such as foot elevation, licking, and biting [28]. It produces a biphasic response, with phase I pain caused by a direct effect of the primary nociceptive afferent, and phase II pain caused by inflammation of the central sensitization of the spinal dorsal horn [29]. In the mice formalin test, Wang et al. found that dezocine produced significant analgesic effect in both phase I and phase II, with ED50 values of 0.4 (0.2–1.0) mg/kg (sc) and 0.4 (0.2–0.9) mg/kg (sc) [14]. Li et al. found that the mice pretreated with dezocine 3 mg/kg (ip) could only decrease the pain score in phase II, but not in phase I[24].
Complete Freund’s Adjuvant (CFA)-induced pain CFA is another inflammatory pain model. Intra-articular injection of CFA produces localized edema [30]. Ye et al. reported that dezocine (0.4 µg/kg, sc) significantly increased the CFA-induced inflammatory pain threshold compared to a control group [19].
Capasaicin-induced sensitization is considered as a model for hyperalagesic states [31]. Barrett et al. found that subcutaneous injection of dezocine dose-dependently produced analgesic effects with ED50 values of 0.38 (0.29–0.49) mg/kg and 0.26 (0.18–0.38) mg/kg for male and female of F344 rats, respectively. Local tail injection of dezocine produced antinociceptive action with ED50 values of 236.7 (141.9–394.9) μg and 207.7 (131.3–328.5) μg for male and female rats, respectively [32].
Neuropathic pain
Neuropathic pain is often presented as allodynia or hyperalgesia, caused by damage or disease affecting the somatosensory nervous system. The spinal nerve ligation (SNL) model was tightly ligating the L5 spinal nerve [16]. By using SNL model, Wang et al. found that dezocine produced time and dose-dependent mechanical and thermal analgesia, with ED50 values of 0.6 (0.3–1.3) mg/kg (sc) and 0.3 (0.1–0.4) mg/kg (sc), respectively [16]. Chronic constriction injury (CCI) is another commonly used neuropathic pain model, which is produced by type ligatures around the nerve. CCI causes mild compression of the nerves and leads to incomplete injury of the nerves and hyperalgesia. Wu et al. demonstrated that 3 mg/kg (ip) of dezocine can produce mechanical analgesia and thermal analgesia in CCI-induced neuralgia mice [15]. Zhang et al. used the peripheral nerve compression to build a neuralgia model, and the mechanical pain threshold was measured 21 days after the operation. It was found that intraperitoneal injection of 0.1 mg/kg dezocine significantly increased the mechanical pain threshold of the surgical model group [33].
Rat bone cancer pain
When walker 256 mammary gland carcinoma cells are injected into the tibia, mice develop behavioral signs of mechanical allodynica indicative of pain [34]. Mao et al. firstly demonstrated that dezocine produced significant mechanical analgesic effect in rat bone cancer pain model with ED50 value of 0.6 (0.4–1.0) mg/kg (sc) [18].
Molecular mechanism of action
Since the new molecular targets of dezocine are identified, the molecular mechanisms underlying the antinociceptive action with dezocine treatment need to be fully understood. Recently information is garnered from preclinical studies and the findings are described in the following section.
Opioid receptors
The previous hypothesis for dezocine’s mechanism of action focuses on the interaction of dezocine with opioid receptors, based on the fact that dezocine-induced analgesia was dose-related antagonized by naloxone, a nonselective opioid receptor antagonist [7, 8]. Recently, the specific receptor mediating dezocine’s action was identified. By using rat neuropathic pain model, Wang et al. reported that selective μ receptors antagonist CTAP, given by intrathecal, prevented dezocine-produced mechanical antiallodynia, but κ receptors antagonist GNTI and δ receptors antagonist naltrindole did not, suggesting that dezocine produced antinociception was mediated by μ opioid receptor [16]. Wang group further applied bone pain model and confirmed the μ receptor mechanism underlying dezocine-induced mechanical antiallodynia. They reported that intrathecal injection of CTAP, not GNTI, nor-BNI or naltrindole, profoundly inhibited dezocine’s mechanical antiallodynia action [18]. However, by applying different pain model, Wang et al. reported that dezocine’s antinociceptive action were through both the κ and μ opioid receptors in vesical pain, since κ selective antagonist nor-BNI and μ antagonist β-FNA, given by ip, both significantly inhibited dezocine-induced antinociception. These findings suggest that dezocine produces antinociceptive action possibly via activation of κ and μ opioid receptors.
Norepinephrine reuptake
Recent reports have shown that dezocine can inhibit noradrenaline reuptake, and spinal norepinephrine was involved in pain transmission, thus, Wang et al. detected the specific effects of norepinephrine depletory (6-OHDA) and selective α2-adrenoceptors antagonist Yohimbe. They found that intrathecal pretreatment with 6-OHDA for 3 days depleted spinal norepinephrine, and produced inhibitory effect on dezocine-induced antinociception in SNL neuropathic pain model [16]. The dezocine’s mechanical antiallydynia was also inhibited by intrathecal administration of Yohimbe. They further reported that in rat bone cancer pain model, both intrathcal administrations of 6-OHDA and yohimbe significantly reduced dezocine mechanical antiallydynia [18]. These data indicate that inhibition of norepinephrine reuptake contributes to dezocine-induced antinociception.
Therapeutic trials
Using dezocine alone: focus on postoperative pain control
Achieving successful treatment of postoperative pain remains to be a challenge, since poor pain control leads to prolonged hospitalization, delayed wound healing, psychological consequences and higher health care cost. Currently, dezocine is the mainstay opioids used in China for postoperative pain management. Intramuscular injection of dezocine produced effective analgesic effect and comfort in 60 patients undergoing inguinal hernia repair surgery for up to 24 h postoperatively [35]. In an open abdominal surgery, dezocine, 0.15 mg/kg intravenous given, offered significant antihyperalgesic and analgesic effect for up to 48 h postoperatively [36]. Patients intraoperatively received intravenous dezocine 0.1 mg/kg reduced the incidence and severity of postoperative catheter-related bladder discomfort compared to flurbiprofen axetil 1 mg/kg [37]. At same dosage, intravenous dezocine significantly decreased the postoperative sore throat and improved patient satisfaction after maxillofacial procedures compared to flurbiprofen axetil 1 mg/kg [38]. Zhou et al. demonstrated that preoperative 0.15 mg/kg dezocine intravenous administration effectively managed postoperative pain following laparoscopic cholecystectomy and gynecological laparoscopic surgery [39,40,41]. Similar results were reported by Zhu et al. that preoperative administration of intramuscular dezocine (0.1 mg/kg) was effective in reducing postoperative pain for laparoscopic cholecystectomy [42].
A few studies carried out dezocine in elderly patient undergoing abdominal operation surgery. Yao et al. reported that deozcine (0.05, 0.1, 0.15 mg/kg, iv) dose-dependently produced analgesic effects, decreased postoperative tracheal extubation-induced stress response, and reduced the occurrence rate of adverse complication, and the sedative effect of dezocine (0.05 mg/kg) was lower than that of the control group fentanyl (10 μg/kg) [43]. In cervical cancer treated with radical surgery, deocine 0.6 mg/kg was effective for postoperative analgesia, but the efficacy was lower than 1 mg/kg oxycodone [44]. Patients undergoing endoscopic sinus surgery received 0.1 mg/kg dezocine intramuscularly before the start of surgery can reduce the occurrence of restlessness, chills, nausea and lethargy [45]. In inguinal hernia, administration of dezocine 0.1 mg/kg decreased the incidence and severity of emergence agitation in preschool children [46]. The relevant clinical studies are shown in Table 4.
Studies using dezocine combined with other agents
In addition to being used alone for pain therapy, combined use of dezocine with other agents increasingly has attracted the attention with exhibiting better analgesia, anesthesia induction effects, or fewer adverse reactions, including drowsiness, vomiting, nausea, or urine retention. The relevant clinical studies are shown in Table 5.
Dezocine with opioid analgesics
When dezocine was administered in combination with sufentanil after gynecological laparoscopic surgery, the analgesic effects were improved and the consumption of sufentanil was significantly reduced [47]. In liver cancer patients undergoing radiofrequency ablation, the dezocine-remifentanil method has a better analgesic effect, and can effectively shorten wake-up time and regulate the expression of inflammatory cytokines TNF-α and IL-6 compared with midazolam-remifentanil intravenous anesthesia [48]. Wu et al. reported that the low concentration of dezocine in combination with morphine, morphine/dezocine: 1/0.05 and 1/0.1 could enhance the postoperative analgesia following thoracotomy, meanwhile, decrease nausea and pruritus [49]. However, Gal and DiFazio reported that the order of administration affected the efficacy of the combination of dezocine and morphine. Dezocine, when given to patients who had received a prior dose of morphine, produced a dramatic increment in analgesia. But with reverse sequence, no additive action was observed [50]. This phenomenon may result from dezocine’s action at the κ receptor, norepinephrine and serotonin reuptake transporters.
Opioid analgesics have severe respiratory depression and cause patients to cough during general anesthesia operations. Studies show that use of dezocine combined with sufentanil, remifentanil or fentanyl can significantly improve the cough caused by opioids, but have no effect on heart rate [51,52,53,54,55,56,57]. For burn patient, dezocine 0.25 μg/kg and sufentanil 1.5 µg/kg can reduce the sedative effect and nausea after skin grafting [58].
Elderly patients may have greater sensitivity to opioid analgesics. Zhao et al. used 1.3 μg/kg sufentanil and 1 mg/kg dezocine in elderly patients colorectal cancer surgery, while the control group received 2.3 μg/kg sufentanil. The depression index of the patients was evaluated 48 h after the operation. The depression index of the patients in the dezocine and sufentanil group was lower than that in the sufentanil group [59]. Wang et al. found that in elderly patient postoperative patient controlled intravenous analgesia, 0.1 mg/kg dezocine + 1 μg/kg sufentanil + 10 mg metoclopramide can significantly improve patients’ satisfaction [60]. Postoperative cognitive dysfunction is a severe postoperative complication that occurs in elderly patients. In a 40 elderly patient undergoing upper abdominal surgeries or thoracotomies with general anesthesia study, dezocine and butorphanol analgesia had transient effects on postoperative cognitive function, and the effect was lower than that of dezocine and butorphanol only group [61].
Deozocine with different anesthetic agents
When 0.1 mg/kg dezocine was intravenously administered in combination with 0.05 mg/kg midazolam for anesthesia before vitrectomy, improved pain relief occurred and the incidence of vomiting was reduced [62]. When 0.1 mg/kg dezocine was intravenously injected before etomidate to induce anesthesia, it effectively reduced the occurrence of intraoperative myoclonus [63, 64]. In strabismus surgery, intravenous injection of dezocine and dexmedetomidine can effectively reduce pain and anxiety [65]. In open hepatectomy, infiltration anesthesia with dezocine and ropivacaine can inhibit the secretion of pain factors, significantly shorten the anesthesia recovery time of patients, reduce postoperative body stress and cough in the experimental group and produce less side effects such as restlessness and cough [66].
In adult gastroscopy and colonoscopy, propofol was widely used to induce and maintain anesthesia after injection of dezocine. When propofol 1.0–2.5 mg/kg was used in combination with dezocine 0.05 mg/kg, sufentanil 0.10 μg/kg, and fentanyl 1.0 μg/kg, the analgesic usage and pain score of the dezocine group were significantly lower than those of other groups. With regard to postoperative recovery time, it was found that the recovery period was shorter in the dezocine group and sufentanil group [67]. Xu et al. also found that dezocine combined with propofol reduced the dosage of propofol and reduced the occurrence of side effects such as nausea and vomiting [68]. A totaling 630 patients included meta-analysis studies further confirmed that dezocine could prevent propofol injection pain and mitigate its severity [69]. In painless gastroscopy, dezocine 20 µg/kg and propofol 1 mg/kg has lower incidence rate of respiratory depression and body movement than propofol 1 mg/kg alone [70]. In painless abortion, dezocine 0.1 mg/kg combined with propofol 0.5–1.0 mg/kg can reduce postoperative tension as well [71]. Lu et al also reported that dezocine decreased propofol injection pain as effective as lidocaine [72].
Potential role for the treatment of opioid dependence
Opioids have potent analgesic effects, and are commonly used for the management of pain. However, prolonged use, misuse or use without medical supervision can lead to a dependency or addiction. Opioid medicines including methadone and buprenorphine have been used extensively for maintenance treatment of opioid dependence. Wu et al found that dezocine effectively alleviated morphine physical withdrawal syndromes and reinstatement of conditioned place preference behaviors, suggesting that dezocine may have potential in treatment of opioid dependence[73]. Indeed, dezocine and buprenorphine share some similar properties, and both are classified as μ partial agonists. Based on μ partial agonist activity, dezocine may block or displace morphine binding to μ receptor thus contributes to reduced opioid dependence. Further studies, particularly randomized clinical trials and other experiments in human are needed to define the utility of dezocine in opioid addiction.
Adverse effects
Respiratory depression
Several studies have been performed to directly detect the respiratory depressant effect of dezocine. With 5 healthy subjects, it was found that dezocine was 8.6 times as potent as pentazocine in its respiratory depressant over a 3 h monitoring period [74]. Gal and DiFazio further evaluated the respiratory depressant of dezocine in a common clinical doses of 0.15 mg/kg in a group of healthy volunteers. At this dosage, they found that dezocine produced significantly respiratory depression, and this effect reached its peak at 0.3 mg/kg. Dezocine’s ceiling effect of respiratory depression was similar to other agonist-antagonist analgesics such as nalbuphine and nalorphine [50]. Romagnoli and Keats also demonstrated a ceiling effect for respiratory depression of dezocine in man at 30 mg/kg, and the respiratory depression by dezocine was almost completely antagonized by naloxone [75].
Constipation
Constipation is one of the most common adverse reaction of opioid agonists. Bian with colleagues detected the effect of dezocine on intestinal mobility, and made a comparison with morphine and sufentanil. They found that morphine (5, 10, 30 mg/L) and sufentanil (20, 40, 120 μg/L) dose dependently increased the contractile tension of isolated small intestine smooth muscle, but dezocine (1.7, 3.4, 10.2 mg/L) did not cause significant effects. All the opioids decreased small intestinal propulsion. They concluded that compared to morphine and sufentanyl, dezocine might cause less intestinal side effects [76].
Inhibiting human sperm motility
Whether exposure to dezocine and other opioid analgesics could affect sperm motility was investigated by Xu et al. in vitro. It was found that both dezocine and butorphanol induced sperm-immobilizing effect, whereas, fentanyl, alfentanil and sufentanil showed partial inhibitory effects [77].
Conclusion and future directions
Based on the combination of in vitro laboratory assay and in vivo analgesic studies, Dezocine is an opioid with mixed agonist-antagonist activity at κ and μ opioid receptors and norepinephrine/serotonin transporter reuptake inhibitor (Fig. 2) with broad-spectrum analgesic properties, including acute thermal pain, persistent chemical and inflammatory pain, neuropathic pain and bone cancer pain. Dezocine analgesia depends on acting at κ and μ opioid receptor, and inhibiting norepinephrine reuptake. The question remains open whether dezocine’s action is linked to its ability to inhibit serotonin transporter reuptake, and there still remains gaps in our understanding of the molecular and behavioral pharmacology of dezocine. Recent studies introducing of neuropathic pain and bone cancer pain model to detect the analgesic effect of dezocine may shed new light on the potential clinical benefits of dezcoine, although it is reported that neuropathic pain is found to be less responsive to pure μ opioid analgesics, and current guidelines do not specifically recommend use of partial agonists or mixed agonists/antagonists for cancer pain. As a partial agonist, it usually means that the maximal effect produced will be less than that of full agonist. However, studies carried out to date appear to confirm that dezocine produced more potent or equivalent analgesia than morphine in rodents, primates and humans, and there was no ceiling effect on dezocine-induced analgesia in clinical studies. The potent analgesia of dezocine should be due to its action on multiple targets rather than a single specific target. Available clinical evidence also suggests a particular value for dezocine as an anesthetic or as a supplement to balanced anesthesia for surgical procedures. Further controlled studies will also be needed to expand efficacy, improve compliance and enhance safety of dezocine or its combination partner. Limited evidence to date indicates that the primary side effects of dezocine are similar to morphine-like μ agonist (eg, respiratory depression, constipation), but the intensity of these side effects is reduced. Additional clinical studies involving larger patient populations are required to establish the incidence of adverse effects for dezocine. Animal studies have shown that dezocine acts as μ partial agonist. In theory, it should produce fewer abuse potential than other opioids such as morphine, heroin or fentanyl. However, since dezocine is used in China by a clinician administration, little information from controlled clinical trials is available about the abuse potential of dezocine. Studies are also needed to elucidate the abuse liability.
References
Fillingim RB, Loeser JD, Baron R, Edwards RR. Assessment of chronic pain: domains, methods, and mechanisms. J Pain. 2016;17:T10–20.
Gatchel RJ, McGeary DD, McGeary CA, Lippe B. Interdisciplinary chronic pain management: past, present, and future. Am Psychol. 2014;69:119–30.
Algera MH, et al. Opioid-induced respiratory depression in humans: a review of pharmacokinetic-pharmacodynamic modelling of reversal. Br J Anaesth. 2019;122:e168–79.
Kiyatkin EA. Respiratory depression and brain hypoxia induced by opioid drugs: morphine, oxycodone, heroin, and fentanyl. Neuropharmacology. 2019;151:219–26.
Lyden J, Binswanger IA. The United States opioid epidemic. Semin Perinatol. 2019;43:123–31.
Volkow ND, Blanco C. The changing opioid crisis: development, challenges and opportunities. Mol Psychiatry. 2021;26:218–33.
O’Brien JJ, Benfield P. Dezocine. A preliminary review of its pharmacodynamic and pharmacokinetic properties, and therapeutic efficacy. Drugs. 1989;38:226–48.
Malis JL, Rosenthale ME, Gluckman MI. Animal pharmacology of Wy-16,225, a new analgesic agent. J Pharmacol Exp Ther. 1975;194:488–98.
Liu RY, Huang XP, Yeliseev A, Xi J, Roth BL. Novel molecular targets of dezocine and their clinical implications. Anesthesiology. 2014;120:714–23.
Chen JC, Smith ER, Cahill M, Cohen R, Fishman JB. The opioid receptor binding of dezocine, morphine, fentanyl, butorphanol and nalbuphine. Life Sci. 1993;52:389–96.
Gharagozlou P, Demirci H, Clark JD, Lameh J. Activation profiles of opioid ligands in HEK cells expressing delta opioid receptors. BMC Neurosci. 2002;3:19.
Gharagozlou P, Demirci H, David Clark J, Lameh J. Activity of opioid ligands in cells expressing cloned mu opioid receptors. BMC Pharmacol. 2003;3:1.
Gharagozlou P, Hashemi E, DeLorey TM, Clark JD, Lameh J. Pharmacological profiles of opioid ligands at kappa opioid receptors. BMC Pharmacol. 2006;6:3.
Wang YH, Chai JR, Xu XJ, Ye RF, Zan GY, Liu GY, et al. Pharmacological characterization of dezocine, a potent analgesic acting as a κ partial agonist and μ partial agonist. Sci Rep. 2018;8:14087.
Wu FX, Pan RR, Yu WF, Liu R. The anti-nociception effect of dezocine in a rat neuropathic pain model. Transl Perioper Med. 2014;1:5–8.
Wang YX, Mao XF, Li TF, Gong N, Zhang MZ. Dezocine exhibits antihypersensitivity activities in neuropathy through spinal μ-opioid receptor activation and norepinephrine reuptake inhibition. Sci Rep. 2017;7:43137.
Huang YQ, Guo SH, Liu R, Zhu SM, Sun JL, Yao YX. Additive analgesic effect of dexmedetomidine and dezocine administered intrathecally in a mouse pain model. Oncotarget. 2018;9:24391–7.
Mao XF, Ahsan MZ, Apryani E, Tang XQ, Zhao MJ, Li XY, et al. Dual μ-opioid receptor and norepinephrine reuptake mechanisms contribute to dezocine- and tapentadol-induced mechanical antiallodynia in cancer pain. Eur J Pharmacol. 2020;876:173062.
Ye Z, Zhang M, Ding N, Gao P, Hei Y, Wang Y, et al. Antinociceptive effects of dezocine on complete Freund’s adjuvant-induced inflammatory pain in rats. Exp Ther Med. 2018;15:5469–74.
South SM, Smith MT. Apparent insensitivity of the hot plate latency test for detection of antinociception following intraperitoneal, intravenous or intracerebroventricular M6G administration to rats. J Pharmacol Exp Ther. 1998;286:1326–32.
Carstens E, Douglass DK. Midbrain suppression of limb withdrawal and tail flick reflexes in the rat: correlates with descending inhibition of sacral spinal neurons. J Neurophysiol. 1995;73:2179–94.
Cook CD, Rodefer JS, Picker MJ. Selective attenuation of the antinociceptive effects of mu opioids by the putative dopamine D3 agonist 7-OH-DPAT. Psychopharmacology. 1999;144:239–47.
Fischer BD, Miller LL, Henry FE, Picker MJ, Dykstra LA. Increased efficacy of micro-opioid agonist-induced antinociception by metabotropic glutamate receptor antagonists in C57BL/6 mice: comparison with (-)-6-phosphonomethyl-deca-hydroisoquinoline-3-carboxylic acid (LY235959). Psychopharmacology. 2008;198:271–8.
Li NN, Huang YQ, Huang LE, Guo SH, Shen MR, Guo CL, et al. Dezocine antagonizes morphine analgesia upon simultaneous administration in rodent models of acute nociception. Pain Physician. 2017;20:e401–9.
Hu OY, Ho ST, Wang JJ, Lee SC. Paw pressure test in the rabbit: a new animal model for the study of pain. Acta Anaesthesiol Sin. 1996;34:1–8.
Barrett AC, Smith ES, Picker MJ. Sex-related differences in mechanical nociception and antinociception produced by mu- and kappa-opioid receptor agonists in rats. Eur J Pharmacol. 2002;452:163–73.
Collier H, Dinneen LC, Johnson CA, Schneider C. The abdominal constriction response and its suppression by analgesic drugs in the mouse. Br J Pharmacol. 2012;32:295–310.
Hassani FV, Rezaee R, Sazegara H, Hashemzaei M, Shirani K, Karimi G. Effects of silymarin on neuropathic pain and formalin-induced nociception in mice. Iran J Basic Med Sci. 2015;18:715–20.
Yamamoto T, Nozaki-Taguchi N, Chiba T. Analgesic effect of intrathecally administered orexin-A in the rat formalin test and in the rat hot plate test. Br J Pharmacol. 2002;137:170–6.
Yuan Y, Zhang Y, He X, Fan S. Protective effects of sinomenine on CFA-induced inflammatory pain in rats. Med Sci Monit. 2018;24:2018–24.
Kaido M, Inoue S, Kawashima M, Ishida R, Nakamura S, Tsubota K. Capsaicin-induced pain sensitivity in short tear break-up time dry eye. Ocul Surf. 2020;18:620–6.
Barrett AC, Smith ES, Picker MJ. Capsaicin-induced hyperalgesia and mu-opioid-induced antihyperalgesia in male and female Fischer 344 rats. J Pharmacol Exp Ther. 2003;307:237–45.
Zhang X, Chu C, Ma C, Sun J, Liu Z. Analgesic effect of dizocine combined with ropivacaine on recurrent neuropathic pain in peripheral nerve compression rats. Pharmacology. 2020;105:514–21.
Mao-Ying QL, Wang XW, Yang CJ, Li X, Mi WL, Wu GC, et al. Robust spinal neuroinflammation mediates mechanical allodynia in Walker 256 induced bone cancer rats. Mol Brain. 2012;5:16.
Liu X, Hu J, Gao L, Ji X, Zhai D, Song H, et al. Analgesic effect of preoperative dezocine-based local anesthesia in patients undergoing inguinal hernia repair. J Int Med Res. 2018;46:4945–51.
Yu F, Zhou J, Xia S, Xu H, Wang X. Dezocine prevents postoperative hyperalgesia in patients undergoing open abdominal surgery. Evid Based Complement Altern Med. 2015;2015:946194.
Zhang GF, Guo J, Qiu LL, Li SM, Zheng M, Xia JY, et al. Effects of dezocine for the prevention of postoperative catheter-related bladder discomfort: a prospective randomized trial. Drug Des Devel Ther. 2019;13:1281–8.
Jiao L, Liu RC. Effects of dezocine on postoperative sore throat after maxillofacial procedures: a comparison with flurbiprofen axetil. J Peking Univ. 2014;46:104–6.
Zhou M, Wang L, Wu C, Yan L, Wang R, Sun B, et al. Efficacy and safety of different doses of dezocine for preemptive analgesia in gynecological laparoscopic surgeries: a prospective, double blind and randomized controlled clinical trial. Int J Surg. 2017;37:539–45.
Zhou L, Zhang Y, Sun H, Hu R, Wang J, Xu G. Effect of preemptive dezocine before general anesthesia on postoperative analgesia in patients undergoing laparoscopic cholecystectomy: a prospective observational study. Medicine. 2018;97:e12533.
Zhou M, Wang L, Wu C, Yan L, Wang R, Sun B, et al. Reprint of: Efficacy and safety of different doses of dezocine for preemptive analgesia in gynecological laparoscopic surgeries: a prospective, double blind and randomized controlled clinical trial. Int J Surg. 2018;49:84–90.
Zhu Y, Jing G, Yuan W. Preoperative administration of intramuscular dezocine reduces postoperative pain for laparoscopic cholecystectomy. J Biomed Res. 2011;25:356–61.
Yao ZY, Jia Z, Xie YH, Zhang LL, Zhang HS, Wu WQ, et al. Analgesic effect of dezocine in different doses on elderly patients undergoing abdominal operation under general anesthesia and its influence on stress response to postoperative tracheal extubation. Eur Rev Med Pharm Sci. 2017;21:5223–9.
Li Z, Wu H, He R, Hu X, Liu S. Oxycodone versus dezocine for postoperative analgesia in patients with cervical cancer treated with radical surgery. J Cancer Res Ther. 2016;12:27–9.
Su Z, Zhang Y, Liu Y, An LJ, Liu HL. The effect of dezocine in prevention of adverse reaction of tracheal extubation in nasal endoscopic operation. Zhongguo Ying Yong Sheng Li Xue Za Zhi. 2014;30:394–5, 400.
An LJ, Zhang Y, Su Z, Zhang XL, Liu HL, Zhang ZJ, et al. A single dose of dezocine suppresses emergence agitation in preschool children anesthetized with sevoflurane- remifentanil. BMC Anesthesiol. 2017;17:154.
Zhu H, Chen Y, Huang S, Sun X. Interaction of analgesic effects of dezocine and sufentanil for relief of postoperative pain: a pilot study. Drug Des Devel Ther. 2020;14:4717–24.
Jia Q, Tian F, Duan WN, Jia YF, Wang HX, Xia ZY. Effects of dezocine-remifentanil intravenous anaesthesia on perioperative signs, serum TNF-alpha and IL-6 in liver cancer patients undergoing radiofrequency ablation. J Coll Physicians Surg Pak. 2019;29:4–7.
Wu L, Dong YP, Sun L, Sun L. Low concentration of dezocine in combination with morphine enhance the postoperative analgesia for thoracotomy. J Cardiothorac Vasc Anesth. 2015;29:950–4.
Gal TJ, DiFazio CA. Ventilatory and analgesic effects of dezocine in humans. Anesthesiology. 1984;61:716–22.
Ma R, Wei Y, Xu Z. A small dose of dezocine suppresses remifentanil-induced cough in general anesthesia induction: a prospective, randomized, controlled study. BMC Anesthesiol. 2020;20:235.
Sun ZT, Yang CY, Cui Z, Zhang J, Han XP. Effect of intravenous dezocine on fentanyl-induced cough during general anesthesia induction: a double-blinded, prospective, randomized, controlled trial. J Anesth. 2011;25:860–3.
Xu Y, Zhu Y, Wang S, Ren Y, Miao C. Dezocine attenuates fentanyl-induced cough in a dose-dependent manner-a randomized controlled trial. Int J Clin Exp Med. 2015;8:6091–6.
Li YJ, Li YJ. Comparative study of intravenous different dose of dezocine premedication on sufentanil-induced cough. J Clin Pulm Med. 2017;22:2219–22.
Wang H, Qing EM. Effect of intravenous dezocine premedication on sufentanil-induced cough. J Chin Pract Diagnosis Ther. 2015;29:825–6.
Liu XS, Xu GH, Shen QY, Zhao Q, Cheng XQ, Zhang J, et al. Dezocine prevents sufentanil-induced cough during general anesthesia induction: A randomized controlled trial. Pharmacol Rep. 2015;67:52–5.
Xiong Z, Yi P, Song J, Tan M. Dezocine prevents sufentanil-induced cough during general anesthesia induction: a meta-analysis of randomised controlled trials. BMC Anesthesiol. 2020;20:714–23.
Li S, Min S, Wu B, Tang W. Application of patient-controlled intravenous analgesia of dezocine combined with sufentanil in burn patients after surgery. Chin J Burns. 2015;31:48–51. Chinese.
Zhao P, Wu Z, Li C, Yang G, Ding J, Wang K, et al. Postoperative analgesia using dezocine alleviates depressive symptoms after colorectal cancer surgery: A randomized, controlled, double-blind trial. PLoS One. 2020;15:e0233412.
Wang C, Li L, Shen B, Jiang H, Yuan L, Shi D, et al. A multicenter randomized double-blind prospective study of the postoperative patient controlled intravenous analgesia effects of dezocine in elderly patients. Int J Clin Exp Med. 2014;7:530–9.
Ren BX, Zong J, Tang JC, Sun DP, Hui X, Li RQ, et al. Effects of intravenous analgesia with combined dezocine and butorphanol on postoperative cognitive function in elderly patients. Genet Mol Res. 2015;14:5571–6.
Xiang Y, Ye W, Sun N, Jin X. Analgesic and sedative effects of dezocine and midazolam during vitrectomy. Curr Eye Res. 2016;41:1460–4.
He L, Ding Y, Chen H, Qian Y, Li Z. Dezocine pretreatment prevents myoclonus induced by etomidate: a randomized, double-blinded controlled trial. J Anesth. 2015;29:143–5.
Lv Z, Fang J, Zhu J, Liang B, Li F, Jiang S, et al. Intravenous dezocine pretreatment reduces the incidence and intensity of myoclonus induced by etomidate. J Anesth. 2014;28:944–7.
Ye W, Hu Z, Sun C. Application of dezoxine plus dexmedetomidine in strabismus surgery. Natl Med J China. 2014;94:1479–82.
Zhu R, Du T, Gao H. Effects of dezocine and ropivacaine infiltration anesthesia on cellular immune function indicators, anesthesia recovery time and pain factors in patients with open liver resection. Cell Mol Biol. 2020;66:149–54.
Li XT, Ma CQ, Qi SH, Zhang LM. Combination of propofol and dezocine to improve safety and efficacy of anesthesia for gastroscopy and colonoscopy in adults: a randomized, double-blind, controlled trial. World J Clin Cases. 2019;7:3237–46.
Xu BB, Zhao XL, Xu GP. Clinical study of anesthetization by dezocine combined with propofol for indolent colonoscopy. World J Gastroenterol. 2016;22:5609–15.
Zhou C, Yang Y, Zhu Y, Ruan L. Effects of dezocine on prevention of propofol injection pain: a meta-analysis. J Pain Res. 2017;10:1369–75.
Ma J, Zhang P, Zhang Y, Chen Z, Xin W, Zhang D, et al. Effect of dezocine combined with propofol on painless gastroscopy in patients with suspect gastric carcinoma. J Cancer Res Ther. 2016;12:c271–3.
Zheng M, Guo Y, Shan S, Yang S. Dezocine for anesthesia and stress reduction in induced abortion. Patient Prefer Adherence. 2015;9:369–72.
Lu Y, Ye Z, Wong GT, Dong C, Yu J. Prevention of injection pain due to propofol by dezocine: a comparison with lidocaine. Indian J Pharmacol. 2013;45:619–21.
Wu FX, Babazada H, Gao H, Huang XP, Xi CH, Chen CH, et al. Dezocine alleviates morphine-induced dependence in rats. Anesth Analg. 2019;128:1328–35.
Wuest HP, Bellville JW. The respiratory effects of dezocine and pentazocine in man. J Clin Pharmacol. 1979;19:205–10.
Romagnoli A, Keats AS. Ceiling respiratory depression by dezocine. Clin Pharmacol Ther. 1984;35:367–73.
Bian X, Zhou R, Yang Y, Li P, Hang Y, Hu Y, et al. Divergent effect of dezocine, morphine and sufentanil on intestinal motor function in rats. Int J Med Sci. 2015;12:848–52.
Xu B, Wang ZP, Wang YJ, Lu PH, Wang LJ, Wang XH. The toxic effect of opioid analgesics on human sperm motility in vitro. Drug Chem Toxicol. 2013;36:205–8.
Acknowledgements
The research was supported by the National Natural Science Foundation of China (81671322 to JGL and 81773710 to YJW), and the Youth Innovation Promotion Association of the Chinese Academy of Sciences (2017334 to YJW).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Ye, RR., Jiang, S., Xu, X. et al. Dezocine as a potent analgesic: overview of its pharmacological characterization. Acta Pharmacol Sin 43, 1646–1657 (2022). https://doi.org/10.1038/s41401-021-00790-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41401-021-00790-6
Keywords
This article is cited by
-
Intraoperative Esketamine Is Effective at Reducing Acute Postoperative Pain in Bariatric Surgery Patients: a Randomized Control Trial
Obesity Surgery (2023)
-
Risk of esketamine anesthesia on the emergence delirium in preschool children after minor surgery: a prospective observational clinical study
European Archives of Psychiatry and Clinical Neuroscience (2023)