Abstract
Total glycoside of paeony (TGP) has been widely used to treat inflammation and immune diseases in China. Paeoniflorin (Pae) is the major active component of TGP. Although TGP has few adverse drug reactions, the slow onset and low bioavailability of Pae limit its clinical use. Enhanced efficacy without increased toxicity is pursued in developing new agents for inflammation and immune diseases. As a result, paeoniflorin-6′-O-benzene sulfonate (CP-25) derived from Pae, is developed in our group, and exhibits superior bioavailability and efficacy than Pae. Here we describe the development process and research advance on CP-25. The pharmacokinetic parameters of CP-25 and Pae were compared in vivo and in vitro. CP-25 was also compared with the first-line drugs methotrexate, leflunomide, and hydroxychloroquine in their efficacy and adverse effects in arthritis animal models and experimental Sjögren’s syndrome. We summarize the regulatory effects of CP-25 on inflammation and immune-related cells, elucidate the possible mechanisms, and analyze the therapeutic prospects of CP-25 in inflammation and immune diseases, as well as the diseases related to its potential target G-protein-coupled receptor kinases 2 (GRK2). This review suggests that CP-25 is a promising agent in the treatment of inflammation and immune diseases, which requires extensive investigation in the future. Meanwhile, this review provides new ideas about the development of anti-inflammatory immune drugs.
Similar content being viewed by others
Introduction
Paeonia lactiflora pall, as a traditional Chinese medicine, has played an important role in disease prevention and treatment for thousands of years. Since “Shen Nong’s Herbal Classic”, the analgesic effect of P. lactiflora pall has been described in detail in many medical manuals from the past dynasties, inspiring the research and development of modern drugs [1, 2]. Total glucosides of paeony (TGP) is an effective component of the dried roots of the white peony. The TGP capsule developed by our institution was officially approved in 1998 as the first anti-inflammatory immunoregulatory drug in China. TGP mainly contains paeoniflorin (Pae), albiflorin, paeonin, hydroxyl-Pae, and benzoylpaeoniflorin, among which Pae is the most important component, making upmore than 40% of TGP [3].
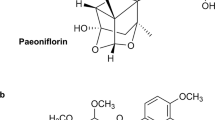
It has been shown that TGP effectively treats inflammation and immune diseases without inducing obvious toxicity [4]. However, the slow onset of action and low bioavailability of TGP, which have been suggested to be attributable to the low oral absorption of Pae, limit its clinical application [4, 5]. Pae is a water-soluble monoterpene with strong hydrophilicity, weak lipophilicity, and weak transmembrane absorption [4, 5]. The low absorption of Pae is related to the intestinal first-pass effect and efflux transporter protein [6,7,8,9] in addition to the physicochemical properties of Pae. Extensive studies on improving the bioavailability of Pae have been conducted. Removing the hydrophilic pyranose structure of Pae increases its ability to penetrate Caco-2 cells by 48 times [10]. Furthermore, acetylation improves the absorption and lipophilicity of Pae in vitro [11] and increases the bioavailability of benzoylpaeoniflorin sulfonate in a mouse model [12]. Based on previous studies, we esterified the pyranose 6-hydroxyl of Pae with benzene sulfonyl chloride and synthesized paeoniflorin-6′-O-benzene sulfonate (CP-25). CP-25 is a modern compound from a natural herb. The chemical structure of CP-25 was determinedby mass spectrometry and nuclear magnetic resonance (Fig. 1).
In this review, we summarize the physiological disposition of CP-25 and its pharmacological effects in an animal model of arthritis and in experimental Sjögren’s syndrome (ESS). We compared the efficacy and adverse effects of CP-25 with those of TGP as well as the first-line drugs methotrexate (MTX), leflunomide (LEF), and hydroxychloroquine (HCQ). In addition, we explored the regulatory effects of CP-25 on various cells and the possible mechanisms and analyzed the therapeutic prospects of CP-25 based on its potential target GRK2 to determine its potential as an agent for subsequent research and clinical treatment of immune and inflammatory diseases.
Physiological disposition of CP-25
Physicochemical properties
Our laboratory has found that CP-25 is slightly soluble in strongly polar water and the nonpolar solvent petroleum ether and is less affected by pH than other compounds [13], indicating that the solubility of orally administered CP-25 delivered by capsules, granules, tablets, or other conventional preparations in the gastrointestinal tract is poor and is not conducive to drug absorption and efficacy. Emulsions, liposomes, and cyclodextrin encapsulation techniques may be used to improve the intestinal absorption of drugs. The study of the physicochemical properties of CP-25 provided experimental bases for establishing quality standards, designing different formulations, and improving the storage conditions of CP-25.
Physiological disposition
Our laboratory first demonstrated that the absolute bioavailability of CP-25 (technical material) is much better than that of Pae in rats [14]. When administered by intravenous injection and gavage, CP-25 (25 or 50 mg/kg) exhibits better oral and intravenous pharmacokinetic parameters, respectively, than Pae (25 or 50 mg/kg) [14] (Table 1). The same result has been observed in an in situ model of single-pass intestinal perfusion in the rat, and passive transport has been found to be the main absorption mechanism of CP-25 in the small intestines of rats [15]. Furthermore, the transport volume of CP-25 is positively correlated with its concentration in Caco-2 cells [16]. Improvements in the biological utilization of CP-25 are mainly attributed to its increased lipid solubility [17] and increased resistance of the P-gp protein [14]. P-gp is an efflux transporter that relies on energy generated by ATP hydrolysis to pump drugs out of the cell to maintain a low intracellular drug concentration [18]. Reportedly, Pae combined with the P-gp inhibitors verapamil and quinidine or pharmaceutical excipients that inhibit P-gp (such as Tween-80) can enhance the absorption of Pae in the intestinal tract [19]. It has been found that inhibiting the expression and function of the P-gp protein increases the intracellular concentration of CP-25. MTX is currently recognized as a first-line drug against RA, and its resistance is thought to be related to P-gp-mediated efflux. Our group found that CP-25 combined with MTX can improve MTX resistance by inhibiting the expression and function of P-gp in adjuvant-induced arthritis (AA) rats, indicating the feasibility of CP-25 combined with MTX for the treatment of RA [20, 21].
CP-25 is distributed in homogeneously throughout all tissues and organs in SD rats (mainly distributed in the heart, lung, synovium, kidney, and muscle) and is found at dynamic concentrations in the circulating blood [14]. The concentration of CP-25 in both the synovium and brain tissues of rats is significantly higher than that in Pae rats (unpublished), suggesting that CP-25 has a higher affinity for the synovium and more easily penetrates the blood–brain barrier than Pae. The distribution of CP-25 in vivo is mainly related to the plasma protein-binding rate, lipid solubility, tissue affinity, and drug transporter levels [14, 19, 20, 22, 23]. In addition, the concentration of CP-25 in peripheral blood lymphocytes (PBMCs) of rats is 1.94 times that of Pae, which may be attributed to the decrease in the expression and function of P-gp and the increase in its concentration in PBMCs [22].
The ester bond and glycoside bond of CP-25 are cleaved by esterase and glycosidase. Under the action of methyltransferase, CP-25 combines with a methionine-derived methyl donor to produce monomethylated and dimethylated products [19]. Our laboratory has also observed that the weakened esterification modification of CP-25 results from dual metabolic effects of enteresterase and hepatic drug enzymes in the intestine and liver, thus improving the absorption of Pae [19].
CP-25 (50 mg/kg) administered i.g. to rats is mostly excreted through the feces, but low amounts are also excreted through bile and urine [14]. Furthermore, the metabolism and excretion of CP-25 in rats are affected by gender [14, 24]. Maximum excretion through the feces and urine requires more time in female rats than in male rats. Maximum excretion rate through the bile is achieved 6 h after drug administration in both male and female rats.
Pharmacological effects of CP-25
TGP has been widely used to treat autoimmune diseases, including RA, systemic lupus erythematosus (SLE), psoriasis, allergic contact dermatitis, etc. Our previous studies indicated that the anti-inflammatory and immunoregulatory effects of CP-25 are superior to those of TGP and Pae. CP-25 inhibits chronic inflammation in dermatitis mice with a lower IC50 value than Pae and TGP (unpublished). The same results have been confirmed in animal models of arthritis and Sjögren’s syndrome (SS) models.
Autoimmune arthritis
RA is a systemic autoimmune disease that is characterized by systemic immune dysfunction and synovial inflammation and has a high disability rate and a serious impact on quality of life. The AA model and collagen-induced arthritis (CIA) model have been used to investigate the anti-arthritic activity of CP-25. In general, CP-25 treatment delays the onset of arthritis and alleviates clinical manifestations in AA rats and CIA mice in a dose-dependent manner [25, 26]. CP-25 significantly decreases ankle joint and spleen histopathological scores in arthritis models, indicating that it has anti-inflammatory and protective effects against joint damage. Cytokine networks regulate a wide range of inflammatory processes that have been identified to play a role in the pathogenesis of RA. A significant decrease in inflammatory factors along with an increase in anti-inflammatory factors has been detected in the serum and the supernatant of fibroblast-like synovial cells (FLSs) and macrophages after CP-25 treatment [25]. Our group also studied the role of CP-25 in rats with carrageen an-induced inflammation and mice with delayed allergic reaction (unpublished). CP-25 can significantly improve spleen histopathology, inhibit the proliferation of T cells in the thymus and spleen, and restore the balance of cytokine secretion.
CP-25 improves clinical symptoms in AA rats and CIA mice at an earlier time point than TGP and Pae at the same dose (50 mg/kg) [25, 26]. For example, in a previous study CP-25 (50 or 100 mg/kg), but not TGP or Pae, had a significant effect starting from day 26 after AA induction. In addition, CP-25 exerts better anti-rheumatism effects than TPG and Pae. For example, CP-25 strongly inhibits cartilage erosion, cellular infiltration, and synovial proliferation, whereas TGP and Pae exert mild effects against these processes [25]. Specifically, CP-25 (25, 50, or 100 mg/kg), but not TGP or Pae, decreases the production of the Th17 cytokine IL-17.
MTX, as the gold standard for the treatment of RA, exerts obvious therapeutic effects. CP-25 (100 mg/kg) shows similar efficacy as MTX in the AA and CIA models [25, 26]. These findings indicate that CP-25 is a potent agent for inhibiting the progression of RA. Notably, disease-modifying antirheumatic drugs (DMARDs), including MTX, have side effects when administered at high doses or for a long period of time. Consistent with a previous report, MTX (0.5 mg/kg) treatment induced obvious lack of movement and weight, and appetite loss in our study [25]. However, these symptoms were not observed following CP-25 treatment, indicating that CP-25 causes fewer adverse reactions at the tested doses. DMARD monotherapy often fails to achieve good efficacy in middle- and late-stage RA patients [27, 28]. For example, although MTX is the anchor drug for RA, 1/3 of RA patients respond poorly to MTX therapy [29]. According to the American College of Rheumatology and European League Against Rheumatism, combination DMARD treatment is recommended for RA since DMARD monotherapy does not achieve the expected efficacy. MTX and LEF are widely used as the corner stones of combination DMARD treatment for RA [30,31,32]. Reportedly, TGP reduces hepatotoxicity caused by combination MTX treatment in RA patients [33, 34]. Moreover, CP-25 has been demonstrated to be effective in relieving the clinical manifestation of arthritis in animal models without inducing significant adverse effects [25]. Therefore, our laboratory compared the efficacy of MTX (0.5 mg/kg) or LEF (10 mg/kg) monotherapy and that of CP-25 (50 mg/kg) combined with MTX or LEF in an AA rat model [35]. Combination treatments exerted better therapeutic effects than all of the monotherapies based on clinical assessment, H&E staining of the joints and spleen, and analysis of cytokine regulation. However, MTX- or LEF-induced liver damage and decreased white blood cell counts were observed in the MTX or LEF monotherapy and combination groups. Considering that dose is an important factor affecting drug toxicity, the therapeutic effects and adverse effects of CP-25 (50 mg/kg) combined with a semiconventional dose of MTX (0.25 mg/kg) or LEF (5 mg/kg) were also determined [35]. The same therapeutic effects were induced by semiconventional combination therapy as MTX or LEF monotherapy, but combination therapy resulted in fewer adverse effects. The above mentioned studies suggest the use of a new strategy involving combination therapy with CP-25 and semiconventional doses of DMARDs for RA that minimizes toxicity and side effects, while ensuring efficacy, providing a reliable experimental basis for the clinical treatment of RA with CP-25.
Primary Sjögren’s syndrome
Primary Sjögren’s syndrome (pSS) is a chronic inflammatory autoimmune disease characterized by various immune abnormalities in moisture-producing glands. The first-line treatment for pSS is artificial teardrops and saliva substitutes [36]. Unfortunately, there are no specific agents to treat pSS, and current treatments are primarily based on those used for other autoimmune diseases. According to the latest Sjogren’s Syndrome Foundation Clinical Practice Guidelines, HCQ is prescribed as a first-line therapy for inflammatory musculoskeletal pain associated with pSS [37]. Pae alleviates SS-like manifestations by improving inflammatory infiltration and cytokine production in vivo [38]. In addition, Pae ameliorates the manifestation of ESS associated with cysteine angiogenesis inducer 61 (Cyr61) expression [38, 39], which is upregulated in the salivary gland epithelial cells of autoantigen-induced experimental Sjögren’s syndrome (ESS) mice. Therefore, the therapeutic effects of CP-25,which has better bioavailability and efficacy than Pae, on pSS have been studied.
NOD/Ltj mice and salivary gland protein-injected C57BL/6 mice are used as ESS models. CP-25 (35 mg/kg, 70 mg/kg) decreases the salivary gland index and thymus index and increases the salivary flow rate [36, 40]. Although HCQ also decreases the salivary gland index, it has no significant effect on the thymus index. CP-25 also reduces the viability of thymocytes and splenocytes [36], the infiltration of Th1/Th2 cells in the salivary glands, and the ratio of Th17/Treg cells in the spleen [40]. Moreover, the levels of anti-La/SSB and IgG antibodies and inflammatory cytokines in the serum are reduced by CP-25 treatment [40]. Taken together, these findings suggest that CP-25 may act as a therapeutic agent for pSS by regulating the function of lymphocytes.
Effects of CP-25 on cells and possible mechanisms
Effect of CP-25 on diverse cellular populations
The dysfunction of immune cells and nonimmune cells in inflammation and immune diseases has been widely reported. CP-25 has been demonstrated to exert its anti-inflammatory immunoregulatory effect by regulating the functions of diverse cellular populations (Table 2, Fig. 2).
CP-25 regulates the function of various cells, influencing the numbers of B-cell subtypes, Th1/Th2 and Th17/Treg balance, FLS-secreted cytokines and FLS proliferation, EC activation, macrophage polarization, and DC maturation. The anti-inflammatory and immunomodulatory functions of CP-25 are attributed to its target, GRK2, which is a multifunctional hub of signaling networks and GPCR pathways. In addition, CP-25 can also inhibit the BAFF-induced TRAF2/NF-κB pathway. CP-25 has been proven to be effective in improving the clinical manifestations of arthritis in animal models and ESS in mice, but further clinical trials are needed. The role of CP-25 in immune, inflammatory and GRK2-related diseases is also worthy of further study. BAFF B-cell-activating factor, NF-κB nuclear transcription factor-κB, TRAF2 tumor necrosis factor receptor-associated factor 2, RA rheumatoid arthritis, pSS primary Sjögren’s syndrome, MS multiple sclerosis, AS ankylosing spondylitis, ACD allergic contact dermatitis, PsA psoriasis arthritis, SLE systemic lupus erythematosus.
Activated T cells secrete large amounts of proinflammatory cytokines, causing multiple organ damage. Our laboratory found that the infiltration of Th1/Th2 cells in the salivary glands is inhibited in CP-25-treated ESS mice. Th17, Treg, Th1, and Th2 cells play significant roles in mediating joint inflammatory cytokines, such as IL-17, IL-2, and IFN-γ. CP-25 reduces T-cell proliferation and regulates the balance between Th1/Th2 and Th17/Treg in the spleens of AA rats and CIA mice [25, 26]. Furthermore, CP-25 can downregulate the level of IL-17 and upregulate the level TGF-β secreted by T cells [25]. Immunoglobulin D (IgD), as a hallmark of autoimmune diseases in patients, binds to the IgD receptor (IgDR) on CD4+ T cells from human PBMCs, leading to the activation and proliferation of T cells [41,42,43]. CP-25 represses the IgD-induced activation and proliferation of CD4+ T cells by inhibiting Lck (Tyr 394) phosphorylation [43].
The excessive proliferation and activation of B cells have been reported to be involved in the pathogenesis of RA and SS [26, 36, 44, 45]. B-cell-activating factor (BAFF) or TNF-α can induce B-cell proliferation in vitro, which is suppressed by CP-25 treatment [45]. CP-25 also reduces the numbers of B-cell subtypes, including CD19+, CD19+CD20+, CD19+CD27+, and CD19+CD20+CD27+ B cells [44]. Moreover, CP-25 moderately restores hyperactivated B-cell function, whereas rituximab and etanercept have more obvious effects on B-cell function [44]. Consistent with the findings in vitro, CP-25 reduces the activation and number of CD19+ B cells and the level of inflammatory factors in CIA mice [26, 45]. CP-25 also improves the pathology of the salivary glands and spleen by reducing the numbers of B-cell subtypes, including CD19+, CD19+CD27+, PDCA1+CD19− B cells, and regulating B-cell function in ESS [36].
In addition, dendritic cells (DCs), as antigen-presenting cells, also participate in the pathological process of RA. In bone marrow DCs isolated from BALB/c mice and stimulated with PGE2 and TNF-α [46, 47], the expression of surface markers (CD40, CD80, CD83, CD86, and MHC-II) and DC antigen uptake are inhibited by CP-25 treatment. In addition, DC-mediated proliferation of T cells is also inhibited by CP-25 when DCs are cocultured with T cells [47].
Hyperplastic macrophages in the synovium are an early hallmark of RA [48]. Our laboratory found that CP-25 can regulate the balance of cytokines secreted by macrophages in AA rats [25], suggesting that CP-25 might restore macrophage polarization to alleviate inflammation. Consistent with this finding, CP-25 was further suggested to decrease proinflammatory macrophage markers (CD86 and iNOS) and increase proresolving macrophage markers (CD206 and Arg1) in the peritoneal macrophages, bone marrow-derived macrophages, and synovial macrophages of CIA mice (unpublished).
Activated FLSs migrate to cartilage and bone, leading to pannus formation and joint destruction. Our laboratory found that CP-25 in vitro inhibited the enhanced migration of CXCL12-treated MH7A cells [49]. Excessive FLS proliferation and elevated levels of PGE2 and TNF-α are detected in AA rats, and CIA mice and are alleviated by CP-25 treatment [26, 35, 50]. In a T cell and FLS coculture system, CP-25 was found to inhibit the proliferation of FLSs and production of proinflammatory cytokines in FLSs by inhibiting the BAFF receptor in CD4+ T cells, suggesting that CP-25 can interfere with the crosstalk between T cells and FLSs in vitro [51].
One of the typical histopathological features of arthritis in RA is the development of the pannus [52]. Previous studies have found that CP-25 can improve pannus formation in AA rats and CIA mice and inhibit the proliferation, migration, and tube formation of endothelial cells (ECs) stimulated by PGE2 or CXCL12, suggesting that CP-25 has an antiangiogenic effect in RA [53, 54].
In addition to the T/B cells mentioned earlier, we also investigated the effect of CP-25 on human salivary gland epithelial (HSGE) cells in vitro [55]. Increased expression of proteins associated with the JAK1-STAT1-CXCL13 axis and IFN has been found in human tissue isolated from pSS patients. After stimulation with IFN-α, the levels of CXCL13 in HSGE cells increase, while CP-25 counteracts the secretion of CXCL13 by inhibiting the JAK1-STAT1/2-CXCL13 signaling pathway.
Potential target of CP-25
Structurally, the cyclohexane with dioxane in CP-25 is similar to the benzo diamene ring in paroxetine, which has been reported to inhibit the activity of G-protein-coupled receptor kinase 2 (GRK2). Through molecular simulation docking and microscale thermophoresis experiments, CP-25 has been confirmed to inhibit GRK2 activity (unpublished). The binding capacity of CP-25 for GRK2 is higher than that of paroxetine for GRK2, indicating that GRK2 is a potential target of CP-25. Based on the inhibitory effect of CP-25 on GRK2, our laboratory found that GRK2 is overexpressed in FLSs [35, 50], DCs [48], macrophages [56], CD4+ T cells [26] and ECs [53] in the peripheral blood of RA patients and arthritis animal models. Regulation of GRK2 activity by CP-25 can inhibit excessive FLS proliferation, proinflammatory macrophage polarization and angiogenesis in RA through related signaling pathways (the GPCR pathway, MAPK pathway, TRAF2/NF-κB pathway, PI3K/AKT pathway, etc.), suggesting the potential importance of targeting GRK2 for RA treatment.
GRKs are kinases associated with the rapid desensitization of G protein-coupled receptors (GPCRs) [56]. Reportedly, GRK2 is widely expressed in immune cells and nonimmune cells, and is involved in the development of multiple diseases [57,58,59]. Under normal physiology, GRK2 is mainly located in the cytoplasm, affects the desensitization and resensitization of GPCRs and regulates (inhibits or activates) a variety of signaling molecules [35, 53, 56]. Under constant stimulation by inflammatory factors, GRK2 is transported to the membrane, leading to (1) abnormal signal transduction downstream of GRK2-phosphorylated GPCRs [60]. (2) GRK2 loses its original ability to inhibit or activate molecules, resulting in abnormal signal transduction related to these molecules [60].
Prostaglandin E2 (PGE2) is a potent lipid mediator, and its level is significantly increased in the serum and synovium in RA [56]. Reportedly, PGE2 regulates the proliferation of FLSs, the activation of ECs, and the maturation of DCs and promotes proresolving macrophage polarization by increasing the cAMP level. PGE2 mediates inflammation through its four PGE2 receptors (EPs)(EP1–4). Our laboratory found that constant stimulation of FLSs [35, 47], DCs [46], macrophages [56], and ECs [53] by PGE2 in AA and CIA models increases GRK2 transport to the membrane, leading to over desensitization of EP4 and abnormal transduction of EP4/cAMP signals. CP-25 can restore PGE2/EP4/cAMP signal transduction by inhibiting GRK2 activity both in vivo and in vitro, thereby restoring the functions of these cells and allowing it to exert its anti-inflammatory and immunoregulatory functions.
On the other hand, it is worth noting that the changes in GRK2 expression and functionality in cells might also affect non-GPCR targets. The level of CXCL12 in the synovium of RA patients is correlated with disease activity, and CXCR4 is associated with joint degeneration [61, 62]. Our laboratory has demonstrated that CXCL12/CXCR4 signals result in FLS migration and EC activation in AA rats, which is attributed to decreased inhibition of ERK1/2 by GRK2. As a member of the MAPK family, ERK1/2 can be activated by inflammatory factors, mitogens, and growth factors and plays an important role in cell growth, proliferation, differentiation, and survival [63]. In general, under physiological conditions, GRK2 and ERK1/2 are mainly located in the cytoplasm, and GRK2 inhibits the phosphorylation of ERK1/2 [64, 65]. Under CCL12 stimulation, GRK2 is transported to the cell membrane, and p-ERK1/2 is transported to the nucleus, leading to decreased inhibition of ERK1/2 by GRK2, which is restored by CP-25 treatment [49]. In addition, B-cell hyperactivity has been confirmed to be involved in the pathogenic mechanism of pSS. CXCL13, which is secreted by the salivary glands, plays a vital role in the progression of pSS and directs B-cell chemotaxis [66]. CXCR5, as the receptor of CXCL13, is related to lymphocyte migration and B-cell follicle migration [67, 68]. Our laboratory has demonstrated that CXCL13/CXCR5 signal-induced p-ERK1/2 activation results in B lymphocyte migration in ESS, which is restored by CP-25-induced inhibition of GRK2 [36]. Consistent with these findings, PGE2/EP4 signal-induced GRK2 transfer to the membrane has been confirmed to promote FLS proliferation [35] and EC activation [53] through decreased inhibition of the ERK1/2 pathway in AA rats. CP-25 inhibits excessive proliferation of FLSs and ECs activation through context-specific molecular mechanisms.
BAFF activates NF-κB signaling, promoting B-cell survival via the BAFF receptor. Tumor necrosis factor receptor-associated factor 2 (TRAF2) interacts with multiple signaling molecules (GRKs, JAKs, NF-κB, etc.) to mediate signal transduction disorders [69]. Our laboratory found that CP-25 inhibits activated B-cell function by inhibiting the BAFF/TRAF2/NF-κB pathways in CIA mice [45], while the direct effect of CP-25 on TRAF2 requires further study. TNF-α-induced TNFR1, TRADD, TRAF2, and NF-κB are also inhibited by CP-25 in DCs, suggesting that CP-25 modulates DC immune function by regulating the TNF-α/TNFR1/TRADD/TRAF2/NF-κB pathways [46].
Therapeutic prospects of CP-25
Our previous study proved that CP-25 has anti-inflammatory and immune regulatory effects in AA, CIA, and ESS, while further clinical trials of CP-25 in RA and pSS patients are still needed. TGP has been widely used in inflammation and immune diseases, and Pae has been reported to exhibit anti-inflammatory [70, 71], anti-neoplastic [72], anti-hyperglycemia [73], and neuroprotective effects [74]. Because CP-25 is derived from Pae, its efficacy in the existing applications of TGP and Pae can be further studied.
Because GRK2 is the regulator of GPCR pathways and a multifunctional hub of numerous signaling networks. CP-25 is a promising candidate for the treatment of multiple disorders. The levels and activity of GRK2 are increased in patients and preclinical models of heart failure (HF), cardiac hypertrophy, hypertension, obesity, insulin resistance, and nonalcoholic fatty liver disease, and these increases favor pathological processes induced by mechanisms related to the multifunctional roles of GRK2 [75,76,77,78,79,80]. Specifically, enhanced GRK2 expression has been reported in failing human hearts and in experimental models of HF [77, 78]. Cardiac GRK2 mRNA and protein levels appear to be increased in HF patients with dilated or ischemic cardiomyopathy. GRK2 levels are increased in peripheral blood samples from HF or acute myocardial infarction patients, but the potential pathophysiological impact of such altered levels remains to be determined. In addition, GRK2 is overexpressed in obese animals and humans, and downregulation of GRK2 levels in hemizygous GRK2+/− mice can protect against TNF-induced alterations in glucose homeostasis and insulin signaling, which provides evidence for a key role for GRK2 in the modulation of insulin sensitivity [79]. GRK2 inhibits insulin receptor (InsR) expression and tyrosine phosphorylation of insulin receptor substrate 1 (IRS1), resulting in impaired insulin action and insulin resistance via inhibition of the InsR/IRS1/PI3K signaling pathway. Inhibition of GRK2 improves glycogen synthesis in mouse liver FL83B cells and glucose metabolism in db/db mice. In autoimmune diseases, GRK2 expression is increased in the synovium of CIA rats during inflammation [80]. GRK2 is also associated with an increase in NF-κB transcriptional activity and upregulation of NF-κB activity, leading to the production of inflammatory cytokines, enhancement of inflammatory cell infiltration, and damage to cartilage and bone [45]. Therefore, genetic GRK2 deletion or pharmacological inhibition is protective in animal models recapitulating these pathological settings.
Drugs targeting GRK2 are still being developed. Recent studies have utilized paroxetine, Takeda’s CMPD101, and two amide derivatives of GSK180736A [81], termed CCG215022 and CCG224063, in in vivo and in vitro experiments. By regulating the level and function of GRK2 in these disease-related preclinical models, disease progression is inhibited to varying degrees, suggesting that CP-25 may play the same role by regulating GRK2. Moreover, GRK2 also interacts with non-GPCR substrates and cellular interactors, including signal transduction kinases (p38MAPK, ERK1/2, PI3K/Akt, MEK1, and AMPK), receptor tyrosine kinases and their downstream molecules (IRS1, EGFR, and PDGFR), transcription factors and their regulatory proteins (Smad2/3 and IκBα), ubiquitin ligases (Mdm2 and Nedd4-2), cytoskeletal proteins and modulators (ezrin, tubulin, GIT1, and HDAC6), G protein subunits and modulators (Gαq, Gβγ, phosducin, RhoA, RalA, and EPAC1), and different enzymes relevant for signaling (Pin1 and eNOS) [82, 83]. These molecules and signals are widely involved in various diseases, and CP-25 might regulate GRK2 to improve disease progression.
Conclusion
Nonsteroidal anti-inflammatory drugs (NSAIDs), corticosteroids, DMARDs, and biologicals are currently available to treat immune and inflammatory diseases [27, 28]. However, NSAIDs reduce disease symptoms without changing the course of the disease. DMARD monotherapies often fail to achieve good efficacy in middle- and late-stage RA patients. High costs and the need for repeated injections limit the widespread use of biologicals. Poor response rates and high costs associated with treatments mandate the search for new therapies.
CP-25, derived from the active ingredient of the natural herb P. lactiflora pall, presents special advantages in arthritis animal models and ESS model. First, CP-25 has better bioavailability and therapeutic effects than TGP and Pae [14, 25]. In addition, similar to first-line drugs such as MTX and LEF, CP-25 (50, 100 mg/kg) affects the course of arthritis early on, while TGP does not [25]. Moreover, CP-25 (100 mg/kg) has equivalent efficacy as MTX and HCQ in the treatment of AA, CIA, and ESS [25, 26, 36, 40]. More importantly, the optimal efficacy and low adverse effects of CP-25 ensure that it has similar effects as MTX (0.5 mg/kg) and LEF (10 mg/kg) when combined with a semiconventional dose of MTX or LEF in AA rats [35]. In particular, the universality of CP-25 in regulating the function of various types of cells is largely due to its targets, i.e., GRK2 and GRK2-related pathways.
The excessive inflammatory immune response (IIR) is the pathological basis of the occurrence and development of multiple systemic diseases. Inhibition of the pathogenic effects of cytokines is often a target in the development of therapeutic drugs for IIR in multiple diseases. In fact, moderate IIR plays an important role in protecting the body from pathological damage to the internal and external environment [84]. However, the excessive regulation of the network balance of cytokines results in serious adverse drug reactions. For example, the JAK-STAT pathway is involved in many important biological processes, such as cell proliferation, differentiation, and apoptosis [85]. Inactivated mutant JAK3 causes immune deficiency. JAK3 is an intracellular molecule necessary for the maintenance of normal immune cell development by various cytokines, hematopoietic cell survival signals, and immunoglobulin class conversion. Tofacitinib, a JAK3 inhibitor used for the treatment of RA, inhibits multiple inflammatory factors through a common target, but inhibition of the activity of JAK3 also disrupts cell physiological functions. Therefore, we propose that soft regulation of inflammatory immune responses (SRIIR) with regards to balanced regulation of the activity of common key molecules in multiple target cells is an important direction for treating inflammatory immune responses [84]. Because GRK2 acts as a multifunctional hub of GPCR and non-GPCR pathways and various signal networks, CP-25 may exert a soft regulatory role in inflammatory immune diseases. Accordingly, better knowledge of the time course of changes in GRK2 levels in pathological situations and their functional impacts is required to better determine the correct timing of therapeutic strategies targeting GRK2.
The concept of SRIIR is ideal and should be used for drug development. Balanced regulation of the common key molecular activities of multiple target cells is an important research direction for the development of drugs for the treatment of IIR-related diseases. However, there are still many topics, including structural modification, dose adjustment, and deeper exploration of mechanisms of CP-25, that need to be studied. Altogether, the data suggest that CP-25 has the potential to be useful for subsequent research and clinical application for immune and inflammatory diseases.
References
Jin Z. To explore the medicine of Cassia twig and Paeonia lactiflora in “Synopsis of Golden Chamber”. forum on traditional Chinese. Medicine. 2008;23:5–6.
Ma SH, Wang HF, Liu JL, Huo XP, Zhao XR, Cao QW, et al. Inhibition of Paeoniflorin on TNF-α-induced TNF-α receptor type I /nuclear factor-κB signal transduction in endothelial cells. Zhongguo Zhong Xi Yi Jie He Za Zhi. 2016;36:339–44.
Kaneda M, Litaka Y, Shibata S. The absolute structures of paeoniflorin, albiflorin, oxypaeoniflorin and benzoylpaeoniflorin isolated from chinese paeony root. Tetrahedron. 1972;28:4309–17.
Wang C, Yuan J, Yang ZY, Nie XX, Song LH, Wei W. Pharmacokinetics of paeoniflorin microemulsion after repeated dosing in rats with adjuvant arthritis. Pharmazie. 2012;67:997–1001.
Takeda S, Isono T, Wakui Y, Matsuzaki Y, Sasaki H, Amagaya S, et al. Absorption and excretion of paeoniflorin in rats. J Pharm Pharmacol. 1995;47:1036–40.
He JX, Akao T, Tani T. Influence of co-administered antibiotics on the pharmacokinetic fate in rats of paeoniflorin and its active metabolite paeonimetabolin-I from Shaoyao-Gancao-tang. J Pharm Pharmacol. 2003;55:313–21.
Hsiu SL, Lin YT, Wen KC, Hou YC, Chao PD. A deglucosylated metabolite of paeoniflorin of the root of Paeonia lactiflora and its pharmacokinetics in rats. Planta Med. 2003;69:1113–8.
Liu ZQ, Jiang ZH, Liu L, Hu M. Mechanisms responsible for poor oral bioavailability of paeoniflorin: Role of intestinal disposition and interactions with sinomenine. Pharm Res. 2006;23:2768–80.
Chan K, Liu ZQ, Jiang ZH, Zhou H, Wong YF, Xu HX, et al. The effects of sinomenine on intestinal absorption of paeoniflorin by the everted rat gut sac model. J Ethnopharmacol. 2006;103:425–32.
Wu H, Zhu Z, Zhang G, Zhao L, Zhang H, Zhu D, et al. Comparative pharmacokinetic study of paeoniflorin after oral administration of pure paeoniflorin, extract of Cortex Moutan and Shuang-Dan prescription to rats. J Ethnopharmacol. 2009;125:444–9.
Yang X, Guo J, Xu W. Absorption and transport characteristic of paeoniflorin and its derivatives in model of Caco-2 cell monolayers. Chinese Traditional and Herbal. Chin Tradit Herb Drugs. 2013;44:2097–104.
Cheng Y, Peng C, Wen F, Zhang H. Pharmacokinetic comparisons of typical constituents in white peony root and sulfur fumigated white peony root after oral administration to mice. J Ethnopharmacol. 2010;129:167–73.
Wang C, Yuan J, Wei W. Study on paeoniflorin-6’O-benzene sulfonate’s physicochemical property. Acta Univ Med Anhui. 2014;49:202–5.
Zhao M, Zhou P, Yu J, James A, Xiao F, Wang C, et al. The tissue distribution and excretion study of paeoniflorin-6’-O-benzene sulfonate (CP-25) in rats. Inflammopharmacology. 2019;27:969–74.
Yang XD, Wang C, Zhou P, Yu J, Asenso J, Ma Y, et al. Absorption characteristic of paeoniflorin-6’-O-benzene sulfonate (CP-25) in in situ single-pass intestinal perfusion in rats. Xenobiotica. 2016;46:775–83.
Wang J, Wang C, Xiao F, Ma Y, Wei W. Study on absorption mechanism of paeoniflorin-6’-O-benzene sulfonate (CP-25) in Caco-2 cells. Acta Univ Med Anhui. 2018;53:1751–6.
Wang C, Yuan J, Zhang LL, Wei W. Pharmacokinetic comparisons of Paeoniflorin and Paeoniflorin-6’O-benzene sulfonate in rats via different routes of administration. Xenobiotica. 2016;46:1142–50.
Szakács G, Váradi A, Ozvegy-Laczka C, Sarkadi B. The role of ABC transporters in drug absorption, distribution, metabolism, excretion and toxicity (ADME-Tox). Drug Discov Today. 2008;13:379–93.
Wang C, Wei W. Research progress of pharmacokinetic study on paeoniflorin. Chin Pharmacol Bull. 2014;30:1646–50.
Tang H, Wu YJ, Xiao F, Wang B, Asenso J, Wang Y, et al. Regulation of CP-25 on P-glycoprotein in synoviocytes of rats with adjuvant arthritis. Biomed Pharmacother. 2019;119:109432.
Asenso J, Yu J, Xiao F, Zhao M, Wang J, Wu Y, et al. Methotrexate improves the anti-arthritic effects of Paeoniflorin-6’-O-benzene sulfonate by enhancing its pharmacokinetic properties in adjuvant-induced arthritis rats. Biomed Pharmacother. 2019;112:108644.
Wu YJ, Zhao MY, Wang J, Tang H, Wang B, Xiao F, et al. Absorption and efflux characteristics of CP-25 in plasma and peripheral blood mononuclear cells of rats by UPLC-MS/MS. Biomed Pharmacother 2018;108:1651–7.
Daohua Shi, Jie Deng, Qiuyan Lian. Relationship between the protein concentration and total concentration of valproate acid in plasma on free concentration. Chin J Clin Pharmacol. 2014;7:1006–8.
Yu J, Xiao F, Asenso J, Peng Z, Yang XD, Wang C, et al. Simultaneous determination of paeoniflorin-6′-O-benzene sulfonate (CP-25) and its active paeoniflorin (Pae) metabolite in rat plasma using UPLC-MS/MS: an application for pharmacokinetic studies. RSC Adv. 2016;113209–18.
Chang Y, Jia X, Wei F, Wang C, Sun X, Xu S, et al. CP-25, a novel compound, protects against autoimmune arthritis by modulating immune mediators of inflammation and bone damage. Sci Rep. 2016;6:26239.
Chen J, Wang Y, Wu H, Yan S, Chang Y, Wei W. A modified compound from paeoniflorin, CP-25, suppressed immune responses and synovium inflammation in collagen-induced arthritis mice. Front Pharmacol. 2018;9:563.
Burmester GR, Pope JE. Novel treatment strategies in rheumatoid arthritis. Lancet. 2017;389:2338–48.
Tanaka Y. Rheumatoid arthritis: DMARD de-escalation - let the patient guide you. Nat Rev Rheumatol. 2017;13:637–8.
McInnes IB, Schett G. Pathogenetic insights from the treatment of rheumatoid arthritis. Lancet. 2017;389:2328–37.
Kavanaugh A, van Vollenhoven RF, Fleischmann R, Emery P, Sainsbury I, Florentinus S, et al. Testing treat-to-target outcomes with initial methotrexate monotherapy compared with initial tumour necrosis factor inhibitor (adalimumab) plus methotrexate in early rheumatoid arthritis. Ann Rheum Dis. 2018;77:289–92.
Nogueira E, Lager F, Le Roux D, Nogueira P, Freitas J, Charvet C, et al. Enhancing methotrexate tolerance with folate tagged liposomes in arthritic mice. J Biomed Nanotechnol. 2015;11:2243–52.
Aletaha D, Stamm T, Kapral T, Eberl G, Grisar J, Machold KP, et al. Survival and effectiveness of leflunomide compared with methotrexate and sulfasalazine in rheumatoid arthritis: a matched observational study. Ann Rheum Dis. 2003;62:944–51.
Xiang N, Li XM, Zhang MJ, Zhao DB, Zhu P, Zuo XX, et al. Total glucosides of paeony can reduce the hepatotoxicity caused by Methotrexate and Leflunomide combination treatment of active rheumatoid arthritis. Int Immunopharmacol. 2015;28:802–7.
Chen Z, Li XP, Li ZJ, Xu L, Li XM. Reduced hepatotoxicity by total glucosides of paeony in combination treatment with leflunomide and methotrexate for patients with active rheumatoid arthritis. Int Immunopharmacol. 2013;15:474–7.
Yang X, Zhao Y, Jia X, Wang C, Wu Y, Zhang L, et al. CP-25 combined with MTX/ LEF ameliorates the progression of adjuvant-induced arthritis by the inhibition on GRK2 translocation. Biomed Pharmacother. 2019;110:834–43.
Chen X, Zhang P, Liu Q, Zhang Q, Gu F, Xu S, et al. Alleviating effect of paeoniflorin-6’-O-benzene sulfonate in antigen-inducedexperimental Sjögren’s syndrome by modulating B lymphocyte migrationvia CXCR5-GRK2-ERK/p38 signaling pathway. Int Immunopharmacol. 2020;80:106199. https://doi.org/10.1016/j.intimp.2020.106199.
Vivino FB, Carsons SE, Foulks G, Daniels TE, Parke A, Brennan MT, et al. New treatment guidelines for Sjögren’s disease. Rheum Dis Clin N Am. 2016;42:531–51.
Li H, Sun X, Zhang J, Sun Y, Huo R, Li H, et al. Paeoniflorin ameliorates symptoms of experimental Sjogren’s syndrome associated with down-regulating Cyr61 expression. Int Immunopharmacol. 2016;30:27–35.
Zhang L, Wei W. Anti-inflammatory and immunoregulatory effects of paeoniflorin and total glucosides of paeony. Pharmacol Ther. 2020;207:107452.
Gu F, Xu S, Zhang P, Chen X, Wu Y, Wang C, et al. CP-25 alleviates experimental Sjögren’s syndrome features in NOD/Ltj mice and modulates T lymphocyte subsets. Basic Clin Pharmacol Toxicol. 2018;123:423–34. https://doi.org/10.1111/bcpt.13025.
Wu Y, Chen W, Chen H, Zhang L, Chang Y, Yan S, et al. The elevated secreted immunoglobulin D enhanced the activation of peripheral blood mononuclear cells in rheumatoid arthritis. PLoS ONE. 2016;11:e0147788.
Wu YJ, Chen WS, Chen HS, Dai X, Dong J, Wang Y, et al. The immunoglobulin D Fc receptor expressed on fibroblast-like synoviocytes from patients with rheumatoid arthritis contributes to the cell activation. Acta Pharmacol Sin. 2017;38:1466–74.
Wu YJ, Chen HS, Chen WS, Dong J, Dong XJ, Dai X, et al. CP-25 attenuates the activation of CD4+ T cells stimulated with immunoglobulin D in human. Front Pharmacol. 2018;9:4. https://doi.org/10.3389/fphar.2018.00004.
Zhang F, Shu JL, Li Y, Wu YJ, Zhang XZ, Han L, et al. CP-25, a novel anti-inflammatory and immunomodulatory drug, inhibits the functions of activated human B cells through regulating BAFF and TNF-alpha signaling and comparative efficacy with biological agents. Front Pharmacol. 2017;8:933.
Shu JL, Zhang XZ, Han L, Zhang F, Wu YJ, Tang XY, et al. Paeoniflorin-6’-O-benzene sulfonate alleviates collagen-induced arthritis in mice by downregulating BAFF-TRAF2-NF-κB signaling: comparison with biological agents. Acta Pharmacol Sin. 2019;40:801–13.
Li Y, Sheng K, Chen J, Wu Y, Zhang F, Chang Y, et al. Regulation of PGE2 signaling pathways and TNF-alpha signaling pathways on the function of bone marrow-derived dendritic cells and the effects of CP-25. Eur J Pharmacol. 2015;769:8–21.
Jia XY, Chang Y, Sun XJ, Wei F, Wu YJ, Dai X, et al. Regulatory effects of paeoniflorin-6’-O-benzene sulfonate (CP-25) on dendritic cells maturation and activation via PGE2-EP4 signaling in adjuvant-induced arthritic rats. Inflammopharmacology. 2019;27:997–1010.
Haringman JJ, Gerlag DM, Zwinderman AH, Smeets TJ, Kraan MC, Baeten D, et al. Synovial tissue macrophages: a sensitive biomarker for response to treatment in patients with rheumatoid arthritis. Ann Rheum Dis. 2005;64:834–8.
Wang DD, Jiang MY, Wang W, Zhou WJ, Zhang YW, Yang M, et al. Paeoniflorin-6’-O-benzene sulfonate down-regulates CXCR4-Gβγ-PI3K/AKT mediated migration in fibroblast-like synoviocytes of rheumatoid arthritis by inhibiting GRK2 translocation. Biochem Biophys Res Commun. 2020;526:805–12.
Jia XY, Chang Y, Wei F, Dai X, Wu YJ, Sun XJ, et al. CP-25 reverses prostaglandin E4 receptor desensitization-induced fibroblast-like synoviocyte dysfunction via the G protein-coupled receptor kinase 2 in autoimmune arthritis. Acta Pharmacol Sin. 2019;40:1029–39.
Jia X, Wei F, Sun X, Chang Y, Xu S, Yang X, et al. CP-25 attenuates the inflammatory response of fibroblast-like synoviocytes co-cultured with BAFF-activated CD4+ T cells. J Ethnopharmacol. 2016;189:194–201.
Yang X, Chang Y, Wei W. Endothelial dysfunction and inflammation: immunity in rheumatoid arthritis. Mediators Inflamm. 2016;2016:6813016.
Han CC, Liu Q, Zhang Y, Li YF, Cui DQ, Luo TT, et al. CP-25 inhibits PGE2-induced angiogenesis by down-regulating EP4/AC/cAMP/PKA-mediated GRK2 translocation. Clin Sci. 2020;134:331–47.
Zhang M, Gao M, Chen J, Song L, Wei W. CP-25 exerts anti-angiogenic effects on a rat model of adjuvant-induced arthritis by promoting GRK2-induced downregulation of CXCR4-ERK1/2 signaling in endothelial cells. Mol Med Rep. 2019;20:4831–42.
Wu H, Chen X, Gu F, Zhang P, Xu S, Liu Q, et al. CP-25 alleviates antigen-induced experimental Sjögren’s syndrome in mice by inhibiting JAK1-STAT1/2-CXCL13 signaling and interfering with B-cell migration. Lab Investig. 2020. https://doi.org/10.1038/s41374-020-0453-0.
Yang X, Li S, Zhao Y, Li S, Zhao T, Tai Y, et al. GRK2 mediated abnormal transduction of PGE2-EP4-cAMP-CREB signaling induces the imbalance of macrophages polarization in collagen-induced arthritis mice. Cells. 2019;8:E1596.
Steury MD, McCabe LR, Parameswaran N. G protein-coupled receptor kinases in the inflammatory response and signaling. Adv Immunol. 2017;136:227–77.
Hagen SA, Kondyra AL, Grocott HP, El-Moalem H, Bainbridge D, Mathew JP, et al. Cardiopulmonary bypass decreases G protein-coupled receptor kinase activity and expression in human peripheral blood mononuclear cells. Anesthesiology. 2003;98:343–8.
Vroon A, Heijnen CJ, Lombardi MS, Cobelens PM, Mayor F Jr, Caron MG, et al. Reduced GRK2 level in T cells potentiates chemotaxis and signaling in response to CCL4. J Leukoc Biol. 2004;75:901–9.
Penela P, Ribas C, Sánchez-Madrid F, Mayor F Jr. G protein-coupled receptor kinase 2 (GRK2) as a multifunctional signaling hub. Cell Mol Life Sci. 2019;76:4423–46.
Grassi F, Cristino S, Toneguzzi S, Piacentini A, Facchini A, Lisignoli G. XCL12 chemokine up-regulates bone resorption and MMP-9 release by human osteoclasts: CXCL12 levels are increased in synovial and bone tissue of rheumatoid arthritis patients. J Cell Physiol. 2004;199:244–51.
Kanbe K, Chiba J, Inoue Y, Taguchi M, Yabuki A. SDF-1 and CXCR4 in synovium are associated with disease activity and bone and joint destruction in patients with rheumatoid arthritis treated with golimumab. Mod Rheumatol. 2016;26:46–50.
Whiteman M, Spencer JP, Zhu YZ, Armstrong JS, Schantz JT. Peroxynitrite-modified collagen-II induces p38/ERK and NF-kappaB-dependent synthesis of prostaglandin E2 and nitric oxide in chondrogenically differentiated mesenchymal progenitor cells. Osteoarthr Cartil. 2006;14:460–70.
Elorza A, Penela P, Sarnago S, Mayor F Jr. MAPK-dependent degradation of G protein-coupled receptor kinase 2. J Biol Chem. 2003;278:29164–73.
Fu X, Koller S, Abd Alla J, Quitterer U. Inhibition of G-protein-coupled receptor kinase 2 (GRK2) triggers the growth-promoting mitogen-activated protein kinase (MAPK) pathway. J Biol Chem. 2013;288:7738–55.
Han FL, Liang F, Jiang TC, Liu M. Increased expression of CXCR5 and CXCL13 in mice with experimental autoimmune myocarditis. Eur Rev Med Pharmacol Sci. 2017;21:1860–7.
Förster R, Mattis AE, Kremmer E, Wolf E, Brem G, Lipp M. A putative chemokine receptor, BLR1, directs B cell migration to defined lymphoid organs and specific anatomic compartments of the spleen. Cell 1996;87:1037–47.
Müller G, Lipp M. Signal transduction by the chemokine receptor CXCR5: structural requirements for G protein activation analyzed by chimeric CXCR1/CXCR5 molecules. Biol Chem. 2001;382:1387–97.
Grech AP, Amesbury M, Chan T, Gardam S, Basten A, Brink R. TRAF2 differentially regulates the canonical and noncanonical pathways of NF-kappaB activation in mature B cells. Immunity. 2004;21:629–42.
Chang Y, Zhang L, Wang C, Jia XY, Wei W. Paeoniflorin inhibits function of synoviocytes pretreated by rIL-1α and regulates EP4 receptor expression. J Ethnopharmacol. 2011;137:1275–82.
Wang C, Yuan J, Wu HX, Chang Y, Wang QT, Wu YJ, et al. Paeoniflorin inhibits inflammatory responses in mice with allergic contact dermatitis by regulating the balance between inflammatory and anti-inflammatory cytokines. Inflamm Res. 2013;62:1035–44.
Wu JJ, Sun WY, Hu SS, Zhang S, Wei W. A standardized extract from Paeonia lactiflora and Astragalus membranaceus induces apoptosis and inhibits the proliferation, migration and invasion of human hepatoma cell lines. Int J Oncol. 2013;43:1643–51.
Yang HO, Ko WK, Kim JY, Ro HS. Paeoniflorin: an antihyperlipidemic agent from Paeonia lactiflora. Fitoterapia. 2004;75:45–9.
Liu DZ, Xie KQ, Ji XQ, Ye Y, Jiang CL, Zhu XZ. Neuroprotective effect of paeoniflorin on cerebral ischemic rat by activating adenosine A1 receptor in a manner different from its classical agonists. Br J Pharmacol. 2005;146:604–11.
Mushegian A, Gurevich VV, Gurevich EV. The origin and evolution of G protein-coupled receptor kinases. PLoS ONE. 2012;7:e33806.
Penela P, Nogués L, Mayor F Jr. Role of G protein-coupled receptor kinases in cell migration. Curr Opin Cell Biol. 2014;27:10–7.
Gurevich EV, Gainetdinov RR, Gurevich VV. G protein-coupled receptor kinases as regulators of dopamine receptor functions. Pharmacol Res. 2016;111:1–16.
Hullmann J, Traynham CJ, Coleman RC, Koch WJ. The expanding GRK interactome: Implications in cardiovascular disease and potential for therapeutic development. Pharmacol Res. 2016;110:52–64.
Ciccarelli M, Chuprun JK, Rengo G, Gao E, Wei Z, Peroutka RJ, et al. G protein-coupled receptor kinase 2 activity impairs cardiac glucose uptake and promotes insulin resistance after myocardial ischemia. Circulation. 2011;123:1953–62.
Wang Q, Wang L, Wu L, Zhang M, Hu S, Wang R, et al. Paroxetine alleviates T lymphocyte activation and infiltration to joints of collagen-induced arthritis. Sci Rep. 2017;7:45364.
Waldschmidt HV, Homan KT, Cruz-Rodríguez O, Cato MC, Waninger-Saroni J, Larimore KM, et al. Structure-based design, synthesis, and biological evaluation of highly selective and potent G protein-coupled receptor kinase 2 inhibitors. J Med Chem. 2016;59:3793–807.
Nogués L, Reglero C, Rivas V, Neves M, Penela P, Mayor F Jr. G-protein-coupled receptor kinase 2 as a potential modulator of the hallmarks of cancer. Mol Pharmacol. 2017;91:220–8.
Mayor F Jr, Cruces-Sande M, Arcones AC, Vila-Bedmar R, Briones AM, Salaices M, et al. G protein-coupled receptor kinase 2 (GRK2) as an integrative signalling node in the regulation of cardiovascular function and metabolic homeostasis. Cell Signal. 2018;41:25–32.
Wei W. Soft regulation of inflammatory immune responses. Chin Pharmacol Bull. 2016;32:297–303.
Boyle DL, Soma K, Hodge J, Kavanaugh A, Mandel D, Mease P, et al. The JAK inhibitor tofacitinib suppresses synovial JAK1-STAT signalling in rheumatoid arthritis. Ann Rheum Dis. 2015;74:1311–6.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (Nos. 81673444, 81330081and 81973332).
Author information
Authors and Affiliations
Contributions
Study concept and design: WW and XZY; Writing of the paper: XZY and WW; All authors reviewed the manuscript prior to submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Yang, Xz., Wei, W. CP-25, a compound derived from paeoniflorin: research advance on its pharmacological actions and mechanisms in the treatment of inflammation and immune diseases. Acta Pharmacol Sin 41, 1387–1394 (2020). https://doi.org/10.1038/s41401-020-00510-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41401-020-00510-6
Keywords
This article is cited by
-
CP-25 enhances OAT1-mediated absorption of methotrexate in synoviocytes of collagen-induced arthritis rats
Acta Pharmacologica Sinica (2023)
-
BAFF Promotes FLS Activation Through BAFFR-Mediated Non-canonical NF-κB Pathway and the Effects of CP-25
Inflammation (2023)