Abstract
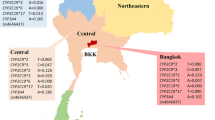
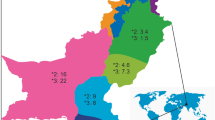
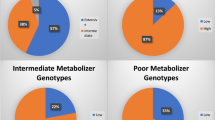
Cytochrome P450 2D6 (CYP2D6) and endothelial nitric oxide synthase (eNOS) are important in the cardiovascular disease susceptibility and drug response. The methylation of cytosine in the promoter of genes could influence expression. The interindividual differences of drug response could only be explained partly by the polymorphisms of drug disposition genes. This study was aimed to investigate the genetic and epigenetic polymorphisms of CYP2D6 and eNOS in Chinese Tibetan, Mongolian, Uygur, and Han volunteers. Blood samples were collected from 842 unrelated Chinese healthy subjects (323 Tibetan, 134 Mongolian, 162 Uygur, and 223 Han) for genotyping and part of the DNA samples were subjected to cytosine methylation analysis. Significant genetic and epigenetic interethnic polymorphisms of eNOS and CYP2D6 were found in the four Chinese groups. The 4b-G-T wild-type haplotype of eNOS was the most common in Chinese Tibetan (87.1%), Mongolian (79.9%), Uygur (76.4%), and Han (79.5%), respectively. eNOS T-786C creates a new CpG site and a significantly higher methylation level was found in −786CC than that in −786TC in Chinese Han (P = 0.0485) and total Chinese (P = 0.0104). CYP2D6 A-678 (rs28633410) makes the symmetrical CpG site changed to be an asymmetrical CpA site, and the methylation level of cytosine in the CpG context of G-678A was significantly higher than that in the CpA. The results of the present study could be useful for understanding the interindividual disparity of cardiovascular disease susceptibility and drug response among different Chinese ethnic groups and would provide more evidences for the precise medication of Chinese.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 print issues and online access
$259.00 per year
only $43.17 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Casas JP, Cavalleri GL, Bautista LE, Smeeth L, Humphries SE, Hingorani AD. Endothelial nitric oxide synthase gene polymorphisms and cardiovascular disease: a HuGE review. Am J Epidemiol. 2006;164:921–35.
Dahgam S, Nyberg F, Modig L, Naluai AT, Olin AC. Single nucleotide polymorphisms in the NOS2 and NOS3 genes are associated with exhaled nitric oxide. J Med Genet. 2012;49:200–5.
Doshi AA, Ziolo MT, Wang H, Burke E, Lesinski A, Binkley P. A promoter polymorphism of the endothelial nitric oxide synthase gene is associated with reduced mRNA and protein expression in failing human myocardium. J Card Fail. 2010;16:314–9.
Pereira TV, Rudnicki M, Cheung BM, Baum L, Yamada Y, Oliveira PS, et al. Three endothelial nitric oxide (NOS3) gene polymorphisms in hypertensive and normotensive individuals: meta-analysis of 53 studies reveals evidence of publication bias. J Hypertens. 2007;25:1763–74.
Alpoim PN, Gomes KB, Pinheiro MB, Godoi LC, Jardim LL, Muniz LG, et al. Polymorphisms in endothelial nitric oxide synthase gene in early and late severe preeclampsia. Nitric Oxide. 2014;42:19–23.
Kumar A, Sagar R, Kumar P, Sahu JK, Grover A, Srivastava AK, et al. Identification of genetic contribution to ischemic stroke by screening of single nucleotide polymorphisms in stroke patients by using a case control study design. BMC Neurol. 2013;13:136.
Santos KG, Crispim D, Canani LH, Ferrugem PT, Gross JL, Roisenberg I. Association of eNOS gene polymorphisms with renal disease in Caucasians with type 2 diabetes. Diabetes Res Clin Pr. 2011;91:353–62.
Hingorani AD, Liang CF, Fatibene J, Lyon A, Monteith S, Parsons A, et al. A common variant of the endothelial nitric oxide synthase (Glu298->Asp) is a major risk factor for coronary artery disease in the UK. Circulation. 1999;100:1515–20.
Luo Y, Chen Y, Zhang Y, Zhou Q, Gao Y. Association of endothelial nitric oxide synthase (eNOS) G894T polymorphism with high altitude pulmonary edema susceptibility: a meta-analysis. Wilderness Environ Med. 2012;23:270–4.
Tanus-Santos JE, Desai M, Flockhart DA. Effects of ethnicity on the distribution of clinically relevant endothelial nitric oxide variants. Pharmacogenetics. 2001;11:719–25.
Gad MZ, Abdel RMF, Hashad IM, Abdel-Maksoud SM, Farag NM, Abou-Aisha K. Endothelial nitric oxide synthase (G894T) gene polymorphism in a random sample of the Egyptian population: comparison with myocardial infarction patients. Genet Test Mol Biomark. 2012;16:695–700.
Zigra AM, Rallidis LS, Anastasiou G, Merkouri E, Gialeraki A. eNOS gene variants and the risk of premature myocardial infarction. Dis Markers. 2013;34:431–6.
Zhou SF. Polymorphism of human cytochrome P450 2D6 and its clinical significance: Part I. Clin Pharmacokinet. 2009;48:689–723.
Teh LK, Bertilsson L. Pharmacogenomics of CYP2D6: molecular genetics, interethnic differences and clinical importance. Drug Metab Pharmacokinet. 2012;27:55–67.
Jirtle RL, Skinner MK. Environmental epigenomics and disease susceptibility. Nat Rev Genet. 2007;8:253–62.
Heyn H, Moran S, Hernando-Herraez I, Sayols S, Gomez A, Sandoval J, et al. DNA methylation contributes to natural human variation. Genome Res. 2013;23:1363–72.
Adkins RM, Krushkal J, Tylavsky FA, Thomas F. Racial differences in gene-specific DNA methylation levels are present at birth. Birth Defects Res A Clin Mol Teratol. 2011;91:728–36.
Wang SM, Zhu AP, Li D, Wang Z, Zhang P, Zhang GL. Frequencies of genotypes and alleles of the functional SNPs in CYP2C19 and CYP2E1 in mainland Chinese Kazakh, Uygur and Han populations. J Hum Genet. 2009;54:372–5.
Qi GZ, Wang X, Miao XJ, Yin SJ, Ren H, Lou YQ, et al. Novel single nucleotide polymorphisms (SNPs) of CYP2W1 gene in Chinese Uygur and Han populations. Drug Metab Pharmacokinet. 2015;30:449–52.
Qi GZ, Zhang ZY, Wang X, Yin SJ, Lou YQ, Zhang GL. Functional allele and genotype frequencies of CYP1A2, CYP2B6 and iNOS among mainland Chinese Tibetan, Mongolian, Uygur and Han populations. J Clin Pharm Ther. 2016;41:84–91.
Naveen AT, Adithan C, Soya SS, Gerard N, Krishnamoorthy R. CYP2D6 genetic polymorphism in South Indian populations. Biol Pharm Bull. 2006;29:1655–8.
Sachse C, Brockmöller J, Bauer S, Roots I. Cytochrome P450 2D6 variants in a Caucasian population: allele frequencies and phenotypic consequences. Am J Hum Genet. 1997;60:284–95.
Cai WM, Nikoloff DM, Pan RM, de Leon J, Fanti P, Fairchild M, et al. CYP2D6 genetic variation in healthy adults and psychiatric African-American subjects: implications for clinical practice and genetic testing. Pharmacogenomics J. 2006;6:343–50.
Ahsan A, Norboo T, Baig MA, Qadar PMA. Simultaneous selection of the wild-type genotypes of the G894T and 4B/ 4A polymorphisms of NOS3 associate with high-altitude adaptation. Ann Hum Genet. 2005;69:260–7.
Erzurum SC, Ghosh S, Janocha AJ, Xu W, Bauer S, Bryan NS, et al. Higher blood flow and circulating NO products offset high-altitude hypoxia among Tibetans. Proc Natl Acad Sci USA. 2007;104:17593–8.
Yi X, Liang Y, Huerta-Sanchez E, Jin X, Cuo ZX, Pool JE, et al. Sequencing of 50 human exomes reveals adaptation to high altitude. Science. 2010;329:75–8.
Ingelman-Sundberg M. Genetic polymorphisms of cytochrome P450 2D6 (CYP2D6): clinical consequences, evolutionary aspects and functional diversity. Pharmacogenomics J. 2005;5:6–13.
Dalai N, Cui H, Yan M, Rile G, Li S, Su X. Risk factors for the development of essential hypertension in a Mongolian population of China: a case-control study. Genet Mol Res. 2014;13:3283–91.
Stoneking M, Delfin F. The human genetic history of East Asia: weaving a complex tapestry. Curr Biol. 2010;20:R188–93.
Lee SJ, Lee SS, Jung HJ, Kim HS, Park SJ, Yeo CW, et al. Discovery of novel functional variants and extensive evaluation of CYP2D6 genetic polymorphisms in Koreans. Drug Metab Dispos. 2009;37:1464–70.
Nishida Y, Fukuda T, Yamamoto I, Azuma J. CYP2D6 genotypes in a Japanese population: low frequencies of CYP2D6 gene duplication but high frequency of CYP2D6*10. Pharmacogenetics. 2000;10:567–70.
Qian JC, Xu XM, Hu GX, Dai DP, Xu RA, Hu LM, et al. Genetic variations of human CYP2D6 in the Chinese Han population. Pharmacogenomics. 2013;14:1731–43.
Gong X, Liu Y, Zhang X, Wei Z, Huo R, Shen L, et al. Systematic functional study of cytochrome P450 2D6 promoter polymorphisms in the Chinese Han population. PLoS ONE. 2013;8:e57764.
Acknowledgements
The study was supported by the National Natural Science Foundation of China (Grants No. 30973585 and No. 91029747), and the Beijing University Center for Human Disease Genomics (Grant No. 2000A-1).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Qi, G., Yin, S., Zhang, G. et al. Genetic and epigenetic polymorphisms of eNOS and CYP2D6 in mainland Chinese Tibetan, Mongolian, Uygur, and Han populations. Pharmacogenomics J 20, 114–125 (2020). https://doi.org/10.1038/s41397-019-0104-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41397-019-0104-2
This article is cited by
-
Human milk cholesterol is associated with lactation stage and maternal plasma cholesterol in Chinese populations
Pediatric Research (2022)
-
Genetic landscape of 125 pharmacogenes in Chinese from the Chinese Millionome Database
Scientific Reports (2021)