Abstract
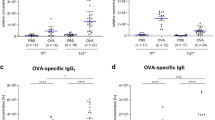
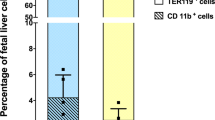
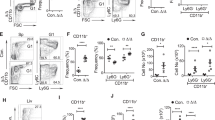
Injections of a crude fetal sheep liver extract (FSLE) containing fetal hemoglobin, MPLA, and glutathione (GSSH) reversed cytokine changes in aged mice. To investigate the role of fetal hemoglobin we derived mice with homzygous deletions for either of the two major βchains, HgbβmaKO or HgbβmiKO. Hgbβmi is the most prominent fetal Hgbβ chain, with Hgbβma more prominent in adult mice. Mice lacking another fetal Hgb chain, HgbεKO, died in utero. CHO cells transfected with cloned Hgb chains were used to produce proteins for preparation of rabbit heteroantibodes. Splenocytes from HgbβmaKO mice stimulated in vitro with Conconavalin A showed a higher IL-2:IL-4 ratio than cells from HgbβmiKO mice. Following immunization in vivo with ovalbumin in alum, HgbβmaKO mice produced less IgE than HgbβmiKO mice, suggesting that in the absence of HgbβmiKO mice had a predeliction to heightened allergic-type responses. Using CHO cells transfected with cloned Hgb chains, we found that only the fetal Hgb chain, Hgbε, was secreted at high levels. Secretion of Hgbβma or Hgbβmi chains was seen only after genetic mutation to introduce the two N-linked glycosylation sites present in Hgbε, but absent in the Hgbβ chains. We speculated that a previously unanticipated biological function of a naturally secreted fetal Hgb chain may be partly responsible for the effects reported following injection of animals with fetal, not adult, Hgb. Mice receiving injections of rabbit anti-Hgbε but not either anti-Hgbβma or anti-Hgbβmi from day 14 gestation also showed a bias towards the higher IL-2:IL-4 ratios seen in HgbβmiKO mice.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 print issues and online access
$259.00 per year
only $43.17 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Gomez CR, AcunaCastillo C, Nishimura S, Perez V, Escobar A, SalazarOnfray F, et al. Serum from aged F344 rats conditions the activation of young macrophages. Mech Age Dev. 2006;127:257–263.
Beverley PCL, GrubeckLoebenstein B. Is immunsenescence reversible? Mech Age Dev. 2000;18:1721–1724.
Kim HR, Hong MS, Dan JM, Kang I. Altered IL-7R alpha expression with aging and the potential implications of IL-7 therapy on CD8(+) T-cell immune responses. Blood. 2006;107:2855–2862.
Gorczynski RM, Alexander C, Bessler W, Brandenburg K, Fournier K, Hoffmann P, et al Role of MIF and glutathione, in association with fetal ovine globin chain (Hbgamma) and LPS, in induction of TNFalpha from cells of young and aged mice, and PBL from healthy human populations. Immunol Lett. 2006;105:140–149.
Carillo EH, Eddy H, Gordon LE, Richardson DJ, Polk HC. Free hemoglobin enhances tumor necrosis factor-alpha production in isolated human monocytes. J Trauma. 2002;52:449–52.
Dunn DL, Nelson RD, Condie RM, Simmons RL. Mechanisms of the adjuvant effect of hemoglobin in experimental peritonitis VI. Effects of stroma-free hemoglobin and red blood cell stroma on mortality and neutrophil function. Surgery. 1983;93:653–9.
Su D, Roth RI, Yoshida M, Levin J. Hemoglobin increases mortality from bacterial endotoxin. Infect Immun. 1999;65:1258–66.
Howe J, Hammer M, Alexander C, Rossle M, Fournier K, Mach JP, et al Biophysical characterization of the interaction of endotoxins with hemoglobins. Med Chem. 2007;3:13–20.
Gorczynski RM, Alexander C, Brandenberg K, Chen Z, Heini A, Heumann D, et al. An altered REDOX environment, assisted by over-expression of fetal hemoglobins, protects from inflammatory colitis and reduces inflammatory cytokine expression. Int J Immunopharmacol. 2017;50: 69–76.
Alter BP, Goff SC. A murine model for the switch from fetal to adult hemoglobin during ontogeny. Blood. 1980;56:1100–1105.
Ogasawara MA, Zhang H. Redox regulation and its emerging roles in stem cells and stem-like cancer cells. Antioxid Redox Signal. 2009;11:1107–1122.
Yoshida Y, Takahashi K, Okita K, Ichisaka T, Yamanaka S. Hypoxia enhances the generation of induced pluripotent stem cells. Cell Stem Cell. 2009;5:237–241.
Wang K, Zhang T, Dong Q, Nice EC, Huang C, Wei Y. Redox homeostasis: the linchpin in stem cell self-renewal and differentiation. Cell Death Dis. 2013; 4: https://doi.org/10.1038/cddis.2013.50
Gorczynski RM, Sadozai H, Zhu F, Khatri I. Effect of infusion of monoclonal antibodies to tumour necrosis factor-receptor super family 25 on graft rejection in allo-immune mice receiving autologous marrow transplantation. Immunology. 2017;150(4):418–431.
Jax labs Technical Support, Master Protocols, 2016. https://www2.jax.org/protocolsdb/f?p=116:5:0::NO:5:P5_MASTER_PROTOCOL_ID,P5_JRS_CODE:18433,002683.
Saldanha JCS, Gargiulo DL, Silva SS, Cara DC. A model of chronic IgE-mediated food allergy in ovalbumin-sensitized mice. Braz J Med Biol Res. 2004;37:809–816.
Gorczynski RM, Dubiski S, Munder PG, Cinader B, Westphal O. Age-related changes in interleukin production in BALB/cNNia and SJL/J mice and their modification after administration of foreign macromolecules. Immunol Lett. 1993;38:243–251.
Hardy K, Hunt NH. Effects of a redox-active agent on lymphocyte activation and early gene expression patterns. Free Radic Biol Med. 2004;37:1550–1563.
Schenk H, Vogt M, Droge W, Schulze-Osthoff K. Thioredoxin as a potent costimulus of cytokine expression. J Immunol. 1996;156:765–771.
Nakamura Y, Takagi D, Homma A, Hatakeyama S, Fukuda S. Oxidative stress regulates IL-4 gene expression in mast cells through the reduction of histone deacetylase. Otolaryngol Head Neck Surg. 2015;152:48–52.
Won HY, Sohn JH, Min HJ, Lee K, Woo HA, Ho YS, et al. Glutathione peroxidase 1 deficiency attenuates allergen-induced airway inflammation by suppressing Th2 and Th17 cell development. Antioxid Redox Signal. 2010;13:575–587.
Hoffman S, Nolin J, McMillan D, Wouters E, Janssen-Heininger Y, Reynaert N. Thiol redox chemistry: role of protein cysteine oxidation and altered redox homeostasis in allergic inflammation and asthma. J Cell Biochem. 2015;116:884–892.
Vural H, Uzun K. Serum and red blood cell antioxidant status in patients with bronchial asthma. Can Respir J. 2000;7:476–480.
Dasgupta T, Fabry ME, Kaul DK. Antisickling property of fetal hemoglobin enhances nitric oxide bioavailability and ameliorates organ oxidative stress in transgenic-knockout sickle mice. Am J Physiol Regul Integr Comp Physiol. 2010;298:R394–402.
Phillips KL, An P, Boyd JH, Strunk RC, Casella JF, Barton BA, et al. Major gene effect and additive familial pattern of inheritance of asthma exist among families of probands with sickle cell anemia and asthma. Am J Hum Biol. 2008;20:149–153.
Palma-Carlos AG, Palma-Carlos ML, Costa AC. “Minor” hemoglobinopathies: a risk factor for asthma. Eur Ann Allergy Clin Immunol. 2005;37:177–182.
Acknowledgements
This work was suppported by the grant from CIHR (Canada) and CLP (Montreux).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Khatri, I., Alexander, C., Brandenburg, K. et al. Analysis of cytokine immune response profile in response to inflammatory stimuli in mice with genetic defects in fetal and adult hemoglobin chain expression. Pharmacogenomics J 18, 546–555 (2018). https://doi.org/10.1038/s41397-017-0008-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41397-017-0008-y