Abstract
Study design
Multi-centre randomized clinical trial.
Objectives
(1) compare the effects of arm-cycle ergometry (ACET) and body weight supported treadmill training (BWSTT) on quality of life (QOL) and intermediary variables in individuals with spinal cord injury (SCI); (2) examine correlations between baseline measures, and changes in physical activity, QOL, and intermediaries.
Setting
Hospital-based research institutes (Vancouver, Toronto) and University-based exercise program (Hamilton).
Methods
35 participants with motor-complete SCI above T6 completed baseline assessments of physical activity, life satisfaction, independence, autonomy, positive and negative affect, and pain. Twenty-eight participants were randomized to 72 sessions of ACET (n = 14) or BWSTT (n = 14) with measures repeated following 36 sessions, 72 sessions, and 6-months post-intervention.
Results
Neither intervention significantly impacted QOL. Pain was reduced in ACET compared to BWSTT (interaction effect p = 0.022) and was significantly less at 72 sessions vs. baseline in the ACET group (p = 0.009). At baseline, QOL was positively correlated with independence, autonomy, and positive affect and negatively correlated with negative affect (all p < 0.05). Following BWSTT, changes in moderate-vigorous physical activity correlated with changes in QOL (r = 0.87, p = 0.010). Following ACET, changes in autonomy and independence were positively correlated with changes in QOL (both r > 0.64, p < 0.048).
Conclusions
Contrary to previous studies, there was no benefit of either intervention on measures of QOL. The social context of exercise may be important for improving QOL. However, individuals may benefit more from active (ACET) than passive (BWSTT) exercise modalities through reduced pain. Exercise interventions that improve autonomy and independence may lead to improvements in QOL.
Similar content being viewed by others
Introduction
The cardiometabolic benefits of exercise for people with spinal cord injury (SCI) are well established [1]. In contrast, despite the dramatic life impact of SCI [2, 3], relatively few studies have examined the quality of life (QOL) benefits. Overall, those studies suggest exercise can have small, but significant positive effects on QOL following SCI [4,5,6]. However, results have varied and not all have reported a positive effect. Such variability is often attributed to inconsistency among intervention protocols regarding the characteristics of exercise frequency, intensity, time, and type of exercise [7]; i.e., some exercise protocols may induce improvements in QOL whereas others may not.
Several explanations suggest how exercise may improve QOL in people with SCI. In general, these explanations propose that relatively proximal outcomes of exercise training (e.g., enhanced mood, increased function) lead to improvements in the more distal outcome of QOL [8]. For instance, Sweet et al. (2013) [9] used structural equation modeling to test whether the relationship between leisure time physical activity (LTPA; i.e., sports, exercise and other forms of recreational activity) and QOL was mediated by depressive symptoms, independence, participation, and self-efficacy in 395 individuals with SCI. Baseline levels of LTPA were related to independence and depressive symptoms at 6-months, which in turn mediated QOL at 18-months. Similarly, Martin Ginis et al. (2003) [10] found that reductions in pain and stress mediated the effects of a 3-month exercise intervention on improvements in QOL in 34 adults with SCI. Yet while these studies suggest exercise improves QOL through its effects on intermediary variables, it is not known how various exercise protocols may differentially impact QOL and these intermediaries.
Among able-bodied individuals, manipulations of one or more characteristics of an exercise protocol or prescription (i.e., frequency, intensity, time, and type) can significantly alter cardiometabolic outcomes. For example, increasing exercise frequency significantly improves aerobic capacity [11]. Among persons with SCI, there is high-quality evidence that upper-body exercise must be performed at a minimum frequency, intensity and duration to significantly improve cardiometabolic outcomes (i.e., 30 min of moderate-vigorous intensity exercise three times per week) [1]. In contrast, the types of exercise, and minimum thresholds needed to improve QOL in people with SCI, are not known. Given the lower QOL often reported by persons with SCI compared with able-bodied individuals [2, 3], it is critical to design exercise interventions that maximize QOL. Thus, developing a better understanding of optimal exercise prescriptions to improve QOL is a valuable first step. While research is needed to determine the optimal exercise prescription based on all four exercise characteristics, one fundamental question for SCI exercise research is whether the type of exercise influences QOL. Of particular interest is whether there is a difference in active upper-limb versus passive lower-limb exercise.
To date, the majority of exercise interventions that measured, or aimed to improve, QOL among people with SCI have utilized active upper-limb exercise [5]. Few studies have examined such outcomes with passive lower-limb exercise, and reported either no effect on QOL [12,13,14,15] or a small positive effect [16, 17]. However, these studies either did not include a comparison group [13, 16] or were conducted only in ambulatory individuals with SCI for whom the exercise was not entirely passive [12, 14, 15, 17] and may not reflect the experiences of the wider SCI population.
The Cardiovascular Health/Outcomes: Improvements Created by Exercise and education in SCI (CHOICES) study [18] provides an ideal opportunity to compare the impact of two exercise types – an active upper-limb modality (arm cycle ergometry training (ACET)) and passive lower-limb modality (body weight supported treadmill training (BWSTT)) - on QOL and potential intermediary variables of the exercise-QOL relationship. Therefore, the primary purpose of the present report was to compare the effects of ACET and BWSTT on QOL and previously identified mediators (i.e., pain, affect, self-efficacy, independence, participation) [9, 10] in individuals with motor complete SCI. Given minimal previous research and inconsistent findings, it was not possible to formulate a hypothesis regarding which exercise condition would lead to greater improvements. The secondary purpose was to advance understanding of the relationship between LTPA, intermediary variables and QOL, as well as correlations between changes in these variables, over the course of exercise training. Consistent with previous research, it was expected that LTPA would positively correlate with positive affect, self-efficacy, independence and participation, and negatively correlate with pain and negative affect.
Methods
Design and trial overview
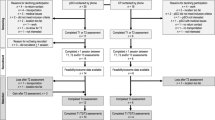
The CHOICES study was a multi-centre, randomized clinical trial (ClinicalTrials.gov identifier: NCT01718977) conducted in collaboration with researchers at the University of British Columbia, Toronto Rehabilitation Institute, and McMaster University. The primary objective of the trial was to compare the effects of BWSTT and ACET on measures of cardiovascular disease risk among individuals with motor-complete SCI between the C4 and T6 spinal level [18]. The present study reports on secondary outcome measures from the larger CHOICES trial.
A detailed description of the CHOICES methodology is published elsewhere [18]. Briefly, participants were randomized to one of two exercise intervention arms and were required to attend three exercise sessions each week for 6 months, totaling 72 sessions. All exercise sessions were conducted in either hospital-based research institutes (Vancouver, Toronto) or a University-based exercise program (Hamilton). Participants completed either 30 min/session of moderate-vigorous intensity ACET (Lode BV, Groningen, The Netherlands; Vancouver site, Monark 881E, Monark Exercise AB, Vansbro, Sweden; Toronto and Hamilton sites) or 60 min/session of BWSTT whereby participants were suspended above a treadmill (Woodway, Weil am Rhein, Germany; Vancouver and Hamilton sites, Thera-Stride, Innoventor Inc., St Louis, Missouri, USA; Toronto site) using a body weight-support system (Andago, Hocoma AG, Volketswil, Switzerland) and volunteers assisted with passive locomotion. The two protocols were matched for exercise volume, calculated as the product of the duration of each session and the session’s ratings of perceived exertion [19]. Descriptions of study methods pertinent to data presented herein are provided below.
Participants
Participants were recruited between January 2013 and October 2018 who (1) were 18–60 years of age; (2) had sustained a motor-complete traumatic SCI between the fourth cervical and sixth thoracic spinal level according to the International Standards for Neurological Classification of Spinal Cord Injury [20] more than one year prior to enrolment, and (3) had a carotid-femoral pulse wave velocity, a measure of cardiovascular disease risk [21], greater than the median normative value of age-matched able-bodied individuals [22] at baseline assessment. All participants were living within the community throughout the duration of the intervention. Each participant provided written informed consent and all experimental procedures conformed to the Declaration of Helsinki.
Measures
A description of each measure is provided below. All questionnaires have been implemented in SCI research previously and, where applicable, the reliability and/or validity in this population are reported.
Quality of Life (QOL) was measured using the Life Satisfaction Questionnaire 9 (LiSAT-9) and Satisfaction With Life Scale (SWLS). The LiSAT-9 assesses satisfaction with life as a whole (i.e. single item) and different aspects of life (eight items) with evidence of validity in persons with SCI [23]. Participants rated each item on a 6-point scale (‘very dissatisfying’ to ‘very satisfying’). Ratings were totaled (range 9–54) with higher scores indicating greater life satisfaction. Cronbach’s α at each time point was 0.87. The SWLS is a 5-item scale that assesses a person’s global life satisfaction. Responses are made on a 7-point Likert scale and item scores are summed with higher scores indicating higher satisfaction (range 5–35). The SWLS has shown temporal reliability [24] and evidence of validity in a sample of persons with SCI [23]. Cronbach’s α at each time point was 0.82-0.93.
Leisure Time Physical Activity (LTPA) was measured using the Leisure Time Physical Activity Questionnaire for People with SCI (LTPAQ-SCI) [25]. Participants recalled how many days they did mild, moderate, and/or heavy intensity LTPA over the previous seven days and for about how many minutes each day. For the purpose of the present study, only moderate- and vigorous-intensity physical activity (MVPA) were used as indicators of LTPA, as these are the intensities recommended in the scientific SCI exercise guidelines [1]. Total weekly MVPA was calculated as the product of the number of days engaged in MVPA in the previous week and the usual time spent doing MVPA each of those days. The LTPAQ-SCI has demonstrated good criterion validity [26] and test-retest reliability [27] in samples of adults with SCI.
Based on previous research and theorizing regarding potential mediators of the effects of exercise on QOL, the following intermediary variables were assessed:
Pain was measured with the 2-item pain subscale from the Medical Outcomes Short-Form 36-Item Health Survey (SF-36) [28]. Participants rated how much bodily pain they experienced and how much pain interfered with their daily life in the past 4 weeks. A higher sum total indicates less pain (range 0-100). Cronbach’s α was between 0.75 and 0.85 at each time point. The SF-36 pain scale has been used in previous studies involving people with SCI and has shown to be responsive to exercise interventions. [29].
Affect was assessed by the Positive and Negative Affect Schedule (PANAS) [30] which measures positive and negative mood states using two ten-item scales. Participants rated various feelings or emotions experienced over the past week using a 5-point scale (‘very slightly or not at all’ to ‘extremely’). A higher total score (range 10–50) indicates greater positive/negative affect. Cronbach’s α at each time point for both subscales was 0.8–0.95 except for negative affect at the 72-session time point (α = 0.74). The PANAS has been used in previous studies of adults with SCI and has demonstrated responsiveness to exercise interventions [31].
Self-Efficacy for performing aerobic exercise was assessed with a 10-item questionnaire [32]. Participants rated their confidence on a 7-point scale (‘not at all confident’ to ‘completely confident’) to perform moderate and heavy-intensity aerobic activity without stopping for different time intervals (e.g., 10 min, 20 min, 30 min). An average score (range 1–7) was calculated. Higher scores reflect greater self-efficacy. Cronbach’s alpha (α) at each time point was 0.8–0.95. This measure was developed specifically for adults with SCI and has demonstrated construct validity through patterns of association with theoretically meaningful constructs [32].
Independence to manage self-care, respiration and sphincter needs, and mobility were assessed with the Spinal Cord Independence Measure (SCIM) version III [33]. Scores from the self-care (range 0–20), respiration and sphincter management (range 0–40), and mobility subscales (range 0–40) were totaled; higher scores indicate greater overall independence (range 0-100). The SCIM III has evidence of reliability and validity in a multi-cultural sample of people with SCI [33]. Cronbach’s α at each time point for the total score was 0.85–0.95.
Participation and Autonomy were assessed with the Impact on Participation and Autonomy Questionnaire (IPAQ) [34]. The questionnaire includes items specific to autonomy indoors (seven items), autonomy outdoors (five items), and social relationships (six items). Participants were asked to rate each item “in the context of illness or disability” on a 7-point scale (‘very poor’ to ‘very good’). Ratings were summed and analyzed as total IPAQ score (range 0–126). Higher scores represent greater participation and autonomy. Cronbach’s α at each time point for autonomy indoors and outdoors was 0.85-0.95 and for social relations was > 0.7.
Procedure
Participants were contacted by email to arrange a time to complete the questionnaires. A trained interviewer administered the questionnaires over the telephone at baseline (prior to randomization), following 36 and 72 sessions of exercise, and six months after completion of exercise training. Order of presentation of the questionnaires was systematically rotated between participants to minimize response bias, with the caveat that measures of LTPA and the intermediary variables always preceded the life satisfaction measures. Participants completed the questionnaires in the same order at each time point. To ensure that baseline fitness tests performed for the CHOICES study [18] were not reported as part of their 7-day recall of LTPA (i.e., LTPAQ-SCI), participants were interviewed either before or 7 days after the fitness test.
Statistical Analyses
As per protocol [18], linear mixed-effects models with fixed effects for timepoint, intervention arm parametrised as a two level-factor (ACET and BWSTT), and timepoint×intervention as fixed effects were performed to assess changes in the study measures. Intraclass correlations for baseline measures across study sites were not significant, and confirmed that data did not violate the assumption of independence of observations. Therefore, study site was not incorporated in our modeling. Following significant interaction or main effects, multiple comparisons were performed using Tukey post-hoc tests. For significant variables only, a further two-way ANOVA (timepoint×intervention) with Tukey post-hoc comparisons tested changes from 72 sessions to follow-up among participants who completed follow-up assessments.
Correlations between LTPA, intermediary variables, and QOL at baseline were assessed by Pearson’s correlation coefficients. To assess correlations between changes in the study measures, residualized change scores were computed to measure changes from baseline to 72 sessions (controlling for baseline scores) and then Pearson’s correlation coefficients were computed for these scores. Correlations were run separately for ACET and BWSTT to examine if exercise modality moderated the relationships. For analyses of correlations between LTPA, intermediary variables, and QOL alpha was not adjusted for multiple correlations given our a priori hypotheses.
Analyses were conducted using SPSS, version 27 (IBM Corp., Armonk, NY, USA) and GraphPad Prism, version 9.1.0 (GraphPad Software, Inc., LaJolla, CA, USA). Data are presented as the mean(SD) and p < 0.05 was considered statistically significant.
Results
Demographics
Baseline questionnaires were completed by 35 participants (University of British Columba n = 20, Toronto Rehabilitation Institute n = 12, McMaster University n = 3). Fourteen participants completed the ACET intervention and 14 completed the BWSTT arm. Seven individuals did not complete either intervention – five chose to withdraw following randomization, one due to injury unrelated to the study, and one due to mood disorder exacerbation. Data from these individuals are only included in baseline analyses. Participant demographics are presented in Table 1.
Effect of intervention
Mean(SD) for each study measure at baseline, following 36 sessions, and following 72 sessions is presented in Table 2.
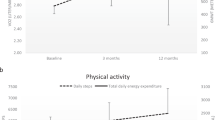
Except for pain, there were no significant interaction effects or main effects for timepoint or intervention for any outcome. A significant interaction effect emerged for the pain subscale whereby pain was significantly less in the ACET condition over the course of the intervention compared to BWSTT (p = 0.022). Tukey post-hoc analysis revealed that following 72 sessions of ACET, pain was significantly lower compared to baseline (p = 0.092, see Fig. 1). At follow-up, pain ratings for each intervention were not different from the 72 session timepoint (see Fig. 1).
Correlations
Correlations between each measure at baseline (n = 35) are presented in Table 3. MVPA was unrelated to intermediary variables or QOL. The intermediary variables of autonomy/participation, and negative affect were significantly correlated with both measures of QOL (rs ranged from −0.36 to 0.75, ps < 0.05). Independence (r = 0.46, p = 0.007) and positive affect (r = 0.50, p = 0.003) were positively correlated with LiSAT scores.
Correlations between changes in measures
Among participants who completed BWSTT, changes in MVPA and life satisfaction were strongly and positively correlated (r = 0.87, p = 0.010). Analyses of participants in the ACET condition showed that changes in the QOL indices were positively correlated with changes in both autonomy (both r > 0.87, p < 0.001) and independence (both r > 0.64, p < 0.048). Pearson correlation coefficients for residualized change from baseline to 72 sessions for each condition are presented in Table 4.
Discussion
This is the first study to examine psychosocial outcomes following an exercise intervention with an exercise prescription consistent with scientific guidelines for improving cardiometabolic health in individuals with SCI [1]. Further, this is the first study to compare the effects of two different exercise modalities on QOL and its potential psychosocial intermediaries in individuals with SCI. We found that participants who completed three 30 min ACET sessions per week for ~24 weeks (i.e., 72 sessions) reported significantly improved pain compared to both baseline and participants who completed BWSTT. There were no significant changes in any of the other study measures. At baseline, autonomy and independence were significant correlates of QOL and, following the ACET intervention only, improvements in these intermediaries were related to improvements in QOL.
Effect of interventions on intermediary variables and QOL
Our finding that ACET reduced pain in individuals with SCI is in agreement with previous work (for review see Todd et al. (2021) [7]). Utilising the SF-36 pain subscale, Martin Ginis et al. (2003) found that three months of combined ACET and resistance training improved pain scores compared to no exercise [10]. Similarly, in a rehabilitation setting, Mulroy et al. (2011) demonstrated that a 12-week stretching and resistance training program (termed ‘exercise and movement optimization’) improved SF-36 pain scores in individuals with thoracic SCI [35]. Interestingly, the baseline pain scores reported by Mulroy et al. were remarkably similar to those of the present study (39.1 and 39.71, respectively) as was the magnitude of change (7.4 and 7.99). It should be noted that Mulroy et al’s study aimed to reduce shoulder pain following SCI and a limitation of the SF-36 is that it does not measure the type or location of pain. To overcome this limitation, future research should incorporate more comprehensive pain measures to elucidate the mechanisms through which exercise can reduce musculoskeletal and/or neuropathic pain following SCI (e.g., by reduced inflammation and/or increased descending inhibitory control) [36]. In this respect, when designing future studies investigators may refer to the International SCI pain dataset – a dataset that was unfortunately not available during the design of the present study [37].
We are unaware of any BWSTT intervention studies that have measured changes in pain. In a study that measured pain following single, acute bouts of BWSTT (n = 14 adults with incomplete injuries) most participants reported no change in pain, while only a few reported decreased pain and some reported increased pain [38]. Based on this evidence, it seems that BWSTT may be a form of exercise that does not consistently alleviate SCI pain. However, it should be noted that at baseline our study participants were very physically active. MVPA levels were sustained across the 72 sessions in the ACET group but there was a large, albeit non-significant, decrease of 110 min/week of MVPA in the BWSTT group. An alternative explanation is that differences in the MVPA performed by the two conditions account for between-group differences in pain; increased sedentary time in the BWSTT group may have exacerbated their pain. Though it is possible that BWSTT could exacerbate pain due to chafing from straps or bruising from repeated stepping, researchers took precautions by placing foam padding to prevent chafing and conducted regular skin checks to ensure participants did not develop tissue injury from the harness [18] and no such adverse events were reported elsewhere [39]. Understanding the psychophysiological mechanisms by which exercise can alleviate SCI pain would help to shed light on the viability of the above explanations.
The large decrease in self-reported MVPA among participants in the BWSTT condition over the course of the intervention was surprising. BWSTT participants may have substituted their regular exercise program for the ~180 min/wk of BWSTT and not considered this to be MVPA. Alternatively, the time requirement to travel to a specialized rehabilitation centre that offered BWSTT and the time taken to set up the harness, etc. may have limited the time they could commit to other exercise modalities.
Whereas previous research has demonstrated improved self-efficacy and mood with exercise in individuals with SCI [6], the present study did not show these effects. As both ACET and BWSTT were progressively overloaded across the intervention (i.e., increased duration and resistance/speed each session) we expected that participants would have felt more confident in their exercise capacity over time. Indeed, mastery experiences are the most important source of self-efficacy [40]. However, the interventionists may not have provided participants with sufficient feedback on changes in their exercise capacity to create mastery experiences. While participants were aware of their weekly progression and rated their perceived exertion each session, they only received feedback on changes in their exercise capacity and cardiopulmonary fitness once – at the 36 session timepoint. The provision of more frequent, meaningful feedback is likely necessary to boost self-efficacy. Without improvements in self-efficacy, exercise-induced improvements in affect are unlikely [41]. It has also been suggested that in order to generate improvements in QOL, individual exercise experiences must satisfy participants’ needs for belonging, autonomy, challenge, mastery, engagement, and meaning [8]. It is unlikely that the clinical research environment of this study, its use of a standardized protocol (rather than protocols tailored to individual needs and preferences), and the lack of opportunity for social interaction among study participants, satisfied these needs.
Finally, we did not observe changes in independence or autonomy across either intervention. Whereas studies with more heterogeneous samples have demonstrated associations between LTPA and independence [9], all participants in the present study had motor-complete injuries. Therefore, it is highly unlikely that we would have observed functional improvements and associated changes in independence and/or autonomy following participation in ACET or BWSTT.
The absence of change in the intermediaries likely explains why neither exercise intervention led to changes in QOL. Studies that have demonstrated a MVPA-QOL relationship in individuals with SCI had much larger samples than the present study [9]. Further, our data highlight that psychosocial benefits are not automatic following exercise interventions. The present study was conducted in clinical settings (i.e., hospital- and university-based) and the results likely do not reflect the impact of community-based exercise programs on measures of QOL. In community exercise settings, individuals are more likely to have a choice over where they exercise, what types of exercises they do, and opportunities to socially engage with other exercisers and participate in a variety of physical activities. These contextual factors likely increase the possibility of experiencing improvements in QOL and other psychosocial benefits of exercise [42, 43]. We believe that the QOL data from the CHOICES study provide an honest reflection of the challenges of implementing clinically oriented interventions with a physiological rationale in a community setting. That is, clinical trials of exercise interventions designed to improve physiological outcomes may not provide an environment conducive to enhancing psychosocial outcomes. These are important considerations when interpreting our findings and issues for investigators to consider when designing future trials that measure and investigate the effects of exercise on QOL.
Correlations between physical activity, intermediary variables, and quality of life
We did not find that improved pain ratings following ACET correlated with improvements in QOL. This is in opposition to previous research [10, 35] but may be explained. First, the MVPA-pain-QOL axis may be a function of the exercise modality. Participants in the present study completed only ACET (or only BWSTT). In studies where pain (assessed by the SF-36 subscale) mediated the effects of exercise on QOL, participants also performed resistance training [10] or combined resistance training and stretching [35]. It may be that resistance training reduces localized upper-extremity pain that is reflected in measures of total bodily pain (i.e., SF-36 pain subscale) whereas ACET in isolation does not. As such, future research should aim to delineate the effects of local and global pain on QOL in individuals with SCI. Second, we were likely underpowered to detect significance. The Martin Ginis et al. (2003) and Mulroy (2011) studies included 34 and 35 participants, respectively.
To our knowledge only one previous study has examined the effects of BWSTT on QOL and potential intermediaries. Hicks et al. (2005) [44] found that BWSTT improved both walking capacity and QOL; it is possible that improved walking capacity improved autonomy and independence, mediating the effects on QOL. Of note, 12/14 of their participants had motor-incomplete SCI (i.e., potential for locomotor recovery) whereas the present study included only participants with motor-complete SCI. As such, we suggest that BWSTT-related changes in QOL may be specific only to those participants who gain, or believe they have the potential to gain, functional improvements; as this study did not assess functional changes in walking, nor were any expected, we did not assess participant expectations of BWSTT at baseline. Hicks et al. (2005) did question whether BWSTT facilitates improved QOL if successful walking is not achieved; our results suggest that it does not. To enhance QOL, exercise may need to impart observable changes in function rather than non-observable changes in physiological parameters (e.g., vascular adaptations).
Our findings, taken together with those of Hicks et al. (2005), raise two prescient questions about how exercise may facilitate changes in QOL in individuals with SCI. First, given that changes in QOL appear to be at least partially mediated by changes in independence and autonomy [9], should exercise interventions for individuals with motor-complete SCI focus on active (e.g., ACET or resistance exercise) rather than passive (e.g., BWSTT, functional electrical stimulation) exercise modalities? Further, does BWSTT have greater potential to improve QOL in individuals with acute SCI who may have greater belief in experiencing functional recovery? These questions are worthy of further investigation.
Methodological considerations
There are a number of considerations to note in the interpretation of the results of the present study. Despite a more homogenous study sample than previous studies, our data was highly variable between individual participants, limiting our ability to detect changes over time. Further, while a homogenous sample may be a strength in some aspects, it limited our recruitment pool and number of participants we were able to assess across all time-points. Nevertheless, despite the strict inclusion/exclusion criteria our total sample size was greater than the mean sample size reported for randomized controlled trials of exercise in individuals with SCI (n = 25) [45]. We also note that the statistical approach of conducting multiple comparisons increases the risk of type I error. Finally, while all participants completed 72 training sessions, adherence was variable as participants missed sessions due to injury and/or illness unrelated to the study procedures. Therefore not all participants completed the interventions within the 24 week timeframe. Additionally, data was not available for all participants at each time-point which limits our ability to identify significant differences between the 72 sessions and follow-up time-points.
Conclusions
The findings of the present study indicate that ~24 weeks of ACET can improve ratings of pain in individuals with motor-complete SCI, whereas BWSTT does not, and improved pain ratings were maintained at 6 months follow-up. Second, while neither intervention led to changes in QOL following the intervention, our results support previous work that suggests improved autonomy and independence to be mediators of enhanced QOL. Further research is warranted to examine the role of the exercise context and psychosocial intermediaries that may account for the effects of exercise on QOL.
Data availability
The datasets generated and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Van Der Scheer JW, Ginis KAM, Ditor DS, Goosey-Tolfrey VL, Hicks AL, West CR, et al. Effects of exercise on fitness and health of adults with spinal cord injury: A systematic review. Neurology 2017;89:736–45.
Dijkers M. Quality of life after spinal cord injury: A meta analysis of the effects of disablement components. Spinal Cord. 1997;35:829–40.
Leduc BE, Lepage Y. Health-related quality of life after spinal cord injury. Disabil Rehabil. 2002;24:196–202.
Franz M, Richner L, Wirz M, Von Reumont A, Bergner U, Herzog T, et al. Physical therapy is targeted and adjusted over time for the rehabilitation of locomotor function in acute spinal cord injury interventions in physical and sports therapy article. Spinal Cord. 2018;56:158–67.
Martin Ginis KA, Jetha A, MacK DE, Hetz S. Physical activity and subjective well-being among people with spinal cord injury: A meta-analysis. Spinal Cord. 2010;48:65–72.
Tomasone JR, Wesch NN, Ginis KAM, Noreau L. Spinal cord injury, physical activity, and quality of life: A systematic review. Kinesiol Rev. 2016;2:113–29.
Todd KR, Lawrason SVC, Shaw RB, Wirtz D, Martin Ginis KA. Physical activity interventions, chronic pain, and subjective well-being among persons with spinal cord injury: A systematic scoping review. Spinal Cord. 2021;59:93–104.
Lox C, Martin Ginis KA, Gainforth HL, Petruzzello S (eds.). The Psychology of Exercise: Integrating Theory and Practice. 5th ed. Routledge: London, 2019.
Sweet SN, Ginis KAM, Tomasone JR. Investigating intermediary variables in the physical activity and quality of life relationship in persons with spinal cord injury. Heal Psychol. 2013;32:877–85.
Martin Ginis KA, Latimer AE, McKechnie K, Ditor DS, McCartney N, Hicks AL, et al. Using exercise to enhance subjective well-being among people with spinal cord injury: The mediating influences of stress and pain. Rehabil Psychol. 2003;48:157–64.
Montero D, Lundby C, Montero D, Lundby C. Refuting the myth of non-response to exercise training: ‘non-responders’ do respond to higher dose of training. Physiol Soc J Physiol. 2017;595:11.
Piira A, Lannem AM, Gjesdal K, Knutsen R, Jørgensen L, Glott T, et al. Quality of life and psychological outcomes of body-weight supported locomotor training in spinal cord injured persons with long-standing incomplete lesions. Spinal Cord. 2020;58:560–9.
Effing TW, Van Meeteren NLU, Van Asbeck FWA, Prevo AJH. Body weight-supported treadmill training in chronic incomplete spinal cord injury: A pilot study evaluating functional health status and quality of life. Spinal Cord. 2006;44:287–96.
Hitzig SL, Craven BC, Panjwani A, Kapadia N, Giangregorio LM, Richards K, et al. Randomized trial of functional electrical stimulation therapy for walking in incomplete spinal cord injury: Effects on quality of life and community participation. Top Spinal Cord Inj Rehabil. 2013;19:245–58.
Wu M, Kim J, Wei F. Facilitating weight shifting during treadmill training improves walking function in humans with spinal cord injury: A randomized controlled pilot study. Am J Phys Med Rehabil. 2018;97:585–92.
Hicks AL, Adams MM, Martin Ginis K, Giangregorio L, Latimer A, Phillips SM, et al. Long-term body-weight-supported treadmill training and subsequent follow-up in persons with chronic SCI: Effects on functional walking ability and measures of subjective well-being. Spinal Cord. 2005;43:291–8.
Alexeeva N, Sames C, Jacobs PL, Hobday L, DiStasio MM, Mitchell SA, et al. Comparison of training methods to improve walking in persons with chronic spinal cord injury: A randomized clinical trial. J Spinal Cord Med. 2011;34:362–79.
Krassioukov AV, Currie KD, Hubli M, Nightingale TE, Alrashidi AA, Ramer L. et al. Effects of exercise interventions on cardiovascular health in individuals with chronic, motor complete spinal cord injury: Protocol for a randomised controlled trial [Cardiovascular Health/Outcomes: Improvements Created by Exercise and education in SCI]. BMJ Open. 2019;9:e023540.
Borg G. Perceived exertion as an indicator of somatic stress. Scand J Rehabil Med. 1970;2:92–8.
Kirshblum SC, Burns SP, Biering-Sorensen F, Donovan W, Graves DE, Jha A, et al. International standards for neurological classification of spinal cord injury (revised 2011). J Spinal Cord Med. 2011;34:535–46.
Mitchell GF, Hwang SJ, Vasan RS, Larson MG, Pencina MJ, Hamburg NM, et al. Arterial stiffness and cardiovascular events: The framingham heart study. Circulation. 2010;121:505–11.
Mattace-Raso FUS, Hofman A, Verwoert GC, Wittemana JCM, Wilkinson I, Cockcroft J, et al. Determinants of pulse wave velocity in healthy people and in the presence of cardiovascular risk factors: ‘Establishing normal and reference values’. Eur Heart J. 2010;31:2338–50.
Post MW, Van Leeuwen CM, Van Koppenhagen CF, De, Groot S. Validity of the life satisfaction questions, the life satisfaction questionnaire, and the satisfaction with life scale in persons with spinal cord injury. Arch Phys Med Rehabil. 2012;93:1832–7.
Diener E, Emmons RA, Larsem RJ, Griffin S. The satisfaction with life scale. J Pers Assess. 1985;49:71–75.
Martin Ginis K, Latimer A. Leisure Time Physical Activity Questionnaire for People with Spinal Cord Injury (LTPAQ-SCI). 2007. http://sciactioncanada.ca/docs/research-publications/leisure-time-physical-activity-questionnaire.pdf.
Martin Ginis KA, Úbeda-Colomer J, Alrashidi AA, Nightingale TE, Au JS, Currie KD, et al. Construct validation of the leisure time physical activity questionnaire for people with SCI (LTPAQ-SCI). Spinal Cord. 2021;59:311–8.
Martin Ginis KA, Phang SH, Latimer AE, Arbour-Nicitopoulos KP. Reliability and validity tests of the leisure time physical activity questionnaire for people with spinal cord injury. Arch Phys Med Rehabil. 2012;93:677–82.
Ware JE, Sherbourne CD. The MOS 36-item short-form health survey (Sf-36): I. conceptual framework and item selection. Med Care. 1992;30:473–83.
Hicks AL, Martin KA, Ditor DS, Latimer AE, Craven C, Bugaresti J, et al. Long-term exercise training in persons with spinal cord injury: Effects on strength, arm ergometry performance and psychological well-being. Spinal Cord. 2003;41:34–43.
Watson D, Clark LA, Tellegen A. Development and validation of brief measures of positive and negative affect: The PANAS scales. J Pers Soc Psychol. 1988;54:1063–70.
Protas EJ, Holmes SA, Qureshy H, Johnson A, Lee D, Sherwood AM. Supported treadmill ambulation training after spinal cord injury: A pilot study. Arch Phys Med Rehabil. 2001;82:825–31.
Latimer-Cheung AE, Arbour-Nicitopoulos KP, Brawley LR, Gray C, Wilson AJ, Prapavessis H, et al. Developing physical activity interventions for adults with spinal cord injury. Part 2: Motivational counseling and peer-mediated interventions for people intending to be active. Rehabil Psychol. 2013;58:307–15.
Itzkovich M, Gelernter I, Biering-Sorensen F, Weeks C, Laramee MT, Craven BC, et al. The Spinal Cord Independence Measure (SCIM) version III: Reliability and validity in a multi-center international study. Disabil Rehabil. 2007;29:1926–33.
Cardol M, De Haan RJ, Van Den Bos GAM, De Jong BA, De, Groot IJM. The development of a handicap assessment questionnaire: The impact on participation and autonomy (IPA). Clin Rehabil. 1999;13:411–9.
Mulroy SJ, Thompson L, Kemp B, Hatchett PP, Newsam CJ, Lupold DG, et al. Strengthening and optimal movements for painful shoulders (STOMPS) in chronic spinal cord injury: A randomized controlled trial. Phys Ther. 2011;91:305–24.
Martin Ginis KA, van der Scheer JW, Todd KR, Davis JC, Gaudet S, Hoekstra F, et al. A pragmatic randomized controlled trial testing the effects of the international scientific SCI exercise guidelines on SCI chronic pain: protocol for the EPIC-SCI trial. Spinal Cord. 2020;58:746–54.
Widerström-Noga E, Biering-Sørensen F, Bryce TN, Cardenas DD, Finnerup NB, Jensen MP, et al. The International spinal cord injury pain basic data set (version 2.0). Spinal Cord. 2014;52:282–6.
Martin Ginis KA, Latimer AE. The effects of single bouts of body-weight supported treadmill training on the feeling states of people with spinal cord injury. Spinal Cord. 2007;45:112–5.
AA A, TE N, KD C, M H, MJ M, A H, et al. Exercise improves cardiorespiratory fitness but not arterial health after spinal cord injury: The CHOICES trial. J Neurotrauma. 2021. https://doi.org/10.1089/NEU.2021.0071.
Bandura A. Self-efficacy: The exercise of control. W.H. Freeman and Company: New York, 1997.
McAuley E, Talbot H-M, Martinez S. Manipulating self-efficacy in the exercise environment in women: Influences on affective responses. Heal Psychol. 1999;18:288–94.
Jackson J, Williams TL, McEachern BM, Latimer-Cheung AE, Tomasone JR. Fostering quality experiences: Qualitative perspectives from program members and providers in a community-based exercise program for adults with physical disabilities. Disabil Health J. 2019;12:296–301.
Shirazipour CH, Evans MB, Leo J, Lithopoulos A, Martin Ginis KA, Latimer-Cheung AE. Program conditions that foster quality physical activity participation experiences for people with a physical disability: A systematic review. 2018; 42:147–55.
Hicks AL, Adams MM, Ginis KM, Giangregorio L, Latimer A, Phillips SM, et al. Long-term body-weight-supported treadmill training and subsequent follow-up in persons with chronic SCI: effects on functional walking ability and measures of subjective well-being. Spinal Cord. 2005;43:291–8.
Lai B, Cederberg K, Vanderbom KA, Bickel CS, Rimmer JH, Motl RW. Characteristics of adults with neurologic disability recruited for exercise trials: A secondary. Anal Adapt Phys Act Q. 2018;35:476–97.
Acknowledgements
The authors would like to thank all study participants and volunteers who assisted with BWSTT. We would also like to thank Dr. Jasmin K. Ma for help with data collection as well as Dr. Joan Ubeda Colomer and Athena Ginis for assistance with data entry.
Funding
The CHOICES study was funded by a project grant from the Canadian Institutes of Health Research with the funding reference number (TCA 118348). Collection of questionnaire data was supported by the Canadian Disability Participation Project which is funded by the Social Sciences and Humanities Research Council of Canada (grant no. 895-2013-1021). AVK holds the Endowed Chair in Spinal Cord Rehabilitation Research, Department of Medicine, UBC. KAMG holds the Reichwald Family Southern Medical Program Chair in Preventive Medicine.
Author information
Authors and Affiliations
Contributions
CMG acquired data, interpreted results, and drafted the manuscript for important intellectual content. ARS developed and implemented the questionnaire administration protocol, and drafted the measurement section. AVK conceived of and designed the work that led to the submission and revised the manuscript for important intellectual content. KAMG conceived of and designed the work that led to the submission, interpreted results, and revised the manuscript for important intellectual content. All authors approved the final version and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics statement
We certify that all applicable institutional and governmental regulations concerning the ethical use of human volunteers were followed during the course of this research. Ethics approval was received from the University of British Columbia (H12-02945-11), McMaster University (12-672) and Toronto Rehabilitation Institute – University Health Network (12-5797).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gee, C.M., Sinden, A.R., Krassioukov, A.V. et al. The effects of active upper-limb versus passive lower-limb exercise on quality of life among individuals with motor-complete spinal cord injury. Spinal Cord 60, 805–811 (2022). https://doi.org/10.1038/s41393-022-00796-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41393-022-00796-9