Abstract
Background
Androgen deprivation therapy (ADT), either by medical or surgical castration, is the backbone for standard treatment of locally advanced or metastatic prostate cancer, yet it is also associated with various metabolic and cardiovascular complications. Recent evidence have shown that obesity, insulin resistance, or metabolic disturbances can be associated with changes in the gut microbiome, while animal studies also show that castration is associated with changes in the gut microbiome. This study aims to investigate whether the fecal microbiota in prostate cancer patients who had undergone prostatectomy or ADT are different, and explore changes in phylogeny and pathways that may lead to side effects from ADT.
Methods
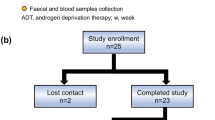
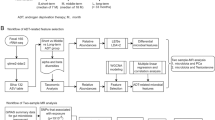
A total of 86 prostate cancer patients (56 patients on ADT and 30 patients with prostatectomy) were recruited. The fecal microbiota was analyzed by the 16S rRNA gene for alpha- and beta-diversities by QIIME2, as well as the predicted metabolic pathways by Phylogenetic Investigation of Communities by Reconstruction of Unobserved States 2.
Results
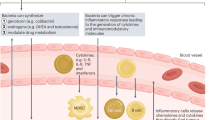
The alpha-diversity was significantly lower in the ADT group. The beta-diversity was significantly different between the groups, in which Ruminococcus gnavus and Bacteroides spp were having higher relative abundance in the ADT group, whereas Lachnospira and Roseburia were reduced. The Firmicutes-to-Bacteroidetes ratio is noted to be lower in the ADT group as well. The functional pathway prediction showed that the biosynthesis of lipopolysaccharide (endotoxin) and propanoate was enriched in the ADT as well as the energy cycle pathways. This study is limited by the cross-sectional design and the clinical heterogeneity.
Conclusions
There is a significant difference in gut microbiome between prostate cancer patients on ADT and prostatectomy. We theorize that this difference may contribute to the development of metabolic complications from ADT. Further longitudinal studies are awaited.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 4 print issues and online access
$259.00 per year
only $64.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Sharifi N, Gulley JL, Dahut WL. Androgen deprivation therapy for prostate cancer. JAMA. 2005;294:238–44.
Teoh JYC, Chiu PKF, Chan SYS, Poon DMC, Cheung H-Y, Hou SSM, et al. Androgen deprivation therapy, diabetes and poor physical performance status increase fracture risk in Chinese men treated for prostate cancer. Aging Male. 2015;18:180–5.
Smith MR. Androgen deprivation therapy and risk for diabetes and cardiovascular disease in prostate cancer survivors. Curr Urol Rep. 2008;9:197–202.
Saylor PJ, Smith MR. Metabolic complications of androgen deprivation therapy for prostate cancer. J Urol. 2009;181:1998–2008.
Levine GN, D’Amico AV, Berger P, Clark PE, Eckel RH, Keating NL, et al. Androgen-deprivation therapy in prostate cancer and cardiovascular risk: a science advisory from the American Heart Association, American Cancer Society, and American Urological Association: endorsed by the American Society for Radiation Oncology. Circulation. 2010;121:833–40.
Teoh JYC, Chiu PKF, Chan SYS, Poon DMC, Cheung H-Y, Hou SSM, et al. Risk of new-onset diabetes after androgen deprivation therapy for prostate cancer in the Asian population. J Diabetes. 2015;7:672–80.
Teoh JYC, Ng C-F. Cardiovascular risk after androgen deprivation therapy for prostate cancer: an Asian perspective. Int Urol Nephrol. 2016;48:1429–35.
Teoh JYC, Chan SYS, Chiu PKF, Poon DMC, Cheung H-Y, Hou SSM, et al. Risk of acute myocardial infarction after androgen-deprivation therapy for prostate cancer in a Chinese population. BJU Int. 2015;116:382–7.
Teoh JYC, Chiu PKF, Chan SYS, Poon DMC, Cheung HY, Hou SSM, et al. Risk of ischemic stroke after androgen deprivation therapy for prostate cancer in the Chinese population living in Hong Kong. Jpn J Clin Oncol. 2015;45:483–7.
Harada N, Hanaoka R, Horiuchi H, Kitakaze T, Mitani T, Inui H, et al. Castration influences intestinal microflora and induces abdominal obesity in high-fat diet-fed mice. Sci Rep. 2016;6:23001.
Aron-Wisnewsky J, Clément K. The gut microbiome, diet, and links to cardiometabolic and chronic disorders. Nat Rev Nephrol. 2016;12:169–81.
Sfanos KS, Markowski MC, Peiffer LB, Ernst SE, White JR, Pienta KJ, et al. Compositional differences in gastrointestinal microbiota in prostate cancer patients treated with androgen axis-targeted therapies. Prostate Cancer Prostatic Dis. 2018;21:539–48.
Kimura M, Tay KJ, Muto S, Horie S. Focal therapy and active surveillance of prostate cancer in East and Southeast Asia. In: Polascik TJ, editor. Imaging and focal therapy of early prostate cancer. Cham: Springer International Publishing; 2017. p. 75–81. Available from: http://link.springer.com/10.1007/978-3-319-49911-6_6.
Lim MY, Park Y-S, Kim J-H, Nam Y-D. Evaluation of fecal DNA extraction protocols for human gut microbiome studies. BMC Microbiol. 2020;20:212.
Cisternas P. Proyecto FONDECYT: study of neuronal glucose metabolism impairment as a molecular link between obesity and the establishment of Alzheimer´s disease: role of adipokines. Nat Methods. 2016;13:4–5.
Bolyen E, Rideout JR, Dillon MR, Bokulich NA, Abnet CC, Al-Ghalith GA, et al. Reproducible, interactive, scalable and extensible microbiome data science using QIIME 2. Nat Biotechnol. 2019;37:852–7.
Quast C, Pruesse E, Yilmaz P, Gerken J, Schweer T, Yarza P, et al. The SILVA ribosomal RNA gene database project: Improved data processing and web-based tools. Nucleic Acids Res. 2013;41:590–6.
Mandal S, Van Treuren W, White RA, Eggesbø M, Knight R, Peddada SD. Analysis of composition of microbiomes: a novel method for studying microbial composition. Micro Ecol Health Dis. 2015;26:1–7.
Segata N, Izard J, Waldron L, Gevers D, Miropolsky L, Garrett WS, et al. Metagenomic biomarker discovery and explanation. Genome Biol. 2011;12:R60.
Wickham H. ggplot2: elegant graphics for data analysis. New York: Springer; 2016.
Afgan E, Baker D, Batut B, van den Beek M, Bouvier D, Čech M, et al. The Galaxy platform for accessible, reproducible and collaborative biomedical analyses: 2018 update. Nucleic Acids Res. 2018;46:W537–44.
Douglas GM, Maffei VJ, Zaneveld J, Yurgel SN, Brown JR, Taylor CM, et al. PICRUSt2: an improved and extensible approach for metagenome inference. bioRxiv. 2019:672295. https://doi.org/10.1101/672295.
Huggins C, Hodges CV. Studies on prostatic cancer. I. The effect of castration, of estrogen and androgen injection on serum phosphatases in metastatic carcinoma of the prostate. Cancer Res. 1941;1:293–7.
Yu I-C, Lin H-Y, Sparks JD, Yeh S, Chang C. Androgen receptor roles in insulin resistance and obesity in males: the linkage of androgen-deprivation therapy to metabolic syndrome. Diabetes. 2014;63:3180–8.
Poljak Z, Hulin I, Maruscakova L, Carter A, Mladosievicova B. Androgen deprivation therapy and cardiovascular complications. Bratisl Med J. 2017;117:557–61.
Dadwal UC, Chang ES, Sankar U. Androgen receptor-CaMKK2 axis in prostate cancer and bone microenvironment. Front Endocrinol. 2018;9:335.
Izard JP, Siemens DR. Androgen deprivation therapy and mental health: impact on depression and cognition. Eur Urol Focus. 2020;6:1162–4.
Liu RT, Rowan-Nash AD, Sheehan AE, Walsh RFL, Sanzari CM, Korry BJ, et al. Reductions in anti-inflammatory gut bacteria are associated with depression in a sample of young adults. Brain Behav Immun. 2020;88:308–24.
Chou H-C. The association of gut microbiome composition and Parkinson’s disease in patient cohort of Central California. UCLA; 2019. Available from: https://escholarship.org/uc/item/09d147pp.
Zhao G, Zhou L, Dong Y, Cheng Y, Song Y. The gut microbiome of hooded cranes (Grus monacha) wintering at Shengjin Lake, China. MicrobiologyOpen. 2017;6:e00447.
Doumatey AP, Adeyemo A, Zhou J, Lei L, Adebamowo SN, Adebamowo C, et al. Gut microbiome profiles are associated with type 2 diabetes in urban Africans. Front Cell Infect Microbiol. 2020;10:63.
Zhang Y, Lu S, Yang Y, Wang Z, Wang B, Zhang B, et al. The diversity of gut microbiota in type 2 diabetes with or without cognitive impairment. Aging Clin Exp Res. 2020. http://link.springer.com/10.1007/s40520-020-01553-9.
Chen Y, Bai J, Wu D, Yu S, Qiang X, Bai H, et al. Association between fecal microbiota and generalized anxiety disorder: severity and early treatment response. J Affect Disord. 2019;259:56–66.
Hsu C-N, Chang-Chien G-P, Lin S, Hou C-Y, Lu P-C, Tain Y-L. Association of trimethylamine, trimethylamine N-oxide, and dimethylamine with cardiovascular risk in children with chronic kidney disease. J Clin Med. 2020;9:336.
Costa M, Garcia M, Costa-Rodrigues J, Costa M, Ribeiro M, Fernandes M, et al. Exploring bioactive properties of marine cyanobacteria isolated from the Portuguese Coast: high potential as a source of anticancer compounds. Mar Drugs. 2013;12:98–114.
Costa M, Rosa F, Ribeiro T, Hernandez-Bautista R, Bonaldo M, Gonçalves Silva N, et al. Identification of cyanobacterial strains with potential for the treatment of obesity-related co-morbidities by bioactivity, toxicity evaluation and metabolite profiling. Mar Drugs. 2019;17:280.
Nguyen TLA, Vieira-Silva S, Liston A, Raes J. How informative is the mouse for human gut microbiota research? Dis Model Mech. 2015;8:1–16.
Lin X-H, Jiang J-K, Luo J-C, Lin C-C, Ting P-H, Yang U-C, et al. The long term microbiota and metabolic status in patients with colorectal cancer after curative colon surgery PLoS ONE. 2019;14:e0218436.
Ley RE, Turnbaugh PJ, Klein S, Gordon JI. Human gut microbes associated with obesity. Nature. 2006;444:1022–3.
Walters WA, Xu Z, Knight R. Meta-analyses of human gut microbes associated with obesity and IBD. FEBS Lett. 2014;588:4223–33.
Magne F, Gotteland M, Gauthier L, Zazueta A, Pesoa S, Navarrete P, et al. The firmicutes/bacteroidetes ratio: a relevant marker of gut dysbiosis in obese patients? Nutrients. 2020;12:1474.
Gurung M, Li Z, You H, Rodrigues R, Jump DB, Morgun A, et al. Role of gut microbiota in type 2 diabetes pathophysiology. EBioMedicine. 2020;51:102590.
Yu F, Han W, Zhan G, Li S, Xiang S, Zhu B, et al. Abnormal gut microbiota composition contributes to cognitive dysfunction in streptozotocin-induced diabetic mice. Aging. 2019;11:3262–79.
Saji N, Murotani K, Hisada T, Tsuduki T, Sugimoto T, Kimura A, et al. The relationship between the gut microbiome and mild cognitive impairment in patients without dementia: a cross-sectional study conducted in Japan. Sci Rep. 2019;9:19227.
Liu P, Wu L, Peng G, Han Y, Tang R, Ge J, et al. Altered microbiomes distinguish Alzheimer’s disease from amnestic mild cognitive impairment and health in a Chinese cohort. Brain Behav Immun. 2019;80:633–43.
Xu Z, Xie Z, Sun J, Huang S, Chen Y, Li C, et al. Gut microbiome reveals specific dysbiosis in primary osteoporosis. Front Cell Infect Microbiol. 2020;10:160.
Liu X, Lu L, Yao P, Ma Y, Wang F, Jin Q, et al. Lipopolysaccharide binding protein, obesity status and incidence of metabolic syndrome: a prospective study among middle-aged and older Chinese. Diabetologia. 2014;57:1834–41.
Awoyemi A, Trøseid M, Arnesen H, Solheim S, Seljeflot I. Markers of metabolic endotoxemia as related to metabolic syndrome in an elderly male population at high cardiovascular risk: a cross-sectional study. Diabetol Metab Syndr. 2018;10:59.
Leigh S-J, Morris MJ. Diet, inflammation and the gut microbiome: mechanisms for obesity-associated cognitive impairment. Biochim Biophys Acta BBA - Mol Basis Dis. 2020;1866:165767.
Ohlsson C, Sjögren K. Effects of the gut microbiota on bone mass. Trends Endocrinol Metab. 2015;26:69–74.
Pastori D, Carnevale R, Nocella C, Novo M, Santulli M, Cammisotto V, et al. Gut‐derived serum lipopolysaccharide is associated with enhanced risk of major adverse cardiovascular events in atrial fibrillation: effect of adherence to mediterranean diet. J Am Heart Assoc. 2017;6. https://doi.org/10.1161/JAHA.117.005784.
Dalile B, Van Oudenhove L, Vervliet B, Verbeke K. The role of short-chain fatty acids in microbiota–gut–brain communication. Nat Rev Gastroenterol Hepatol. 2019;16:461–78.
MacFabe DF, Cain NE, Boon F, Ossenkopp K-P, Cain DP. Effects of the enteric bacterial metabolic product propionic acid on object-directed behavior, social behavior, cognition, and neuroinflammation in adolescent rats: relevance to autism spectrum disorder. Behav Brain Res. 2011;217:47–54.
Wan Y, Yuan J, Li J, Li H, Yin K, Wang F, et al. Overweight and underweight status are linked to specific gut microbiota and intestinal tricarboxylic acid cycle intermediates. Clin Nutr. 2020;39:3189–98.
Spinneker A, Sola R, Lemmen V, Castillo MJ, Pietrzik K, González-Gross M. Vitamin B6 status, deficiency and its consequences-an overview. Nutr Hosp. 2007 ;22:7–24.
Martí-Carvajal AJ, Solà I, Lathyris D, Dayer M. Homocysteine-lowering interventions for preventing cardiovascular events. Cochrane Database Syst Rev. 2017;8:CD006612.
Daisley BA, Chanyi RM, Abdur-Rashid K, Al KF, Gibbons S, Chmiel JA, et al. Abiraterone acetate preferentially enriches for the gut commensal Akkermansia muciniphila in castrate-resistant prostate cancer patients. Nat Commun. 2020;11:4822.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Li, J.K.M., Wang, L.L., Wong, C.Y.P. et al. A cross-sectional study on gut microbiota in prostate cancer patients with prostatectomy or androgen deprivation therapy. Prostate Cancer Prostatic Dis 24, 1063–1072 (2021). https://doi.org/10.1038/s41391-021-00360-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41391-021-00360-1
This article is cited by
-
Gut environment changes due to androgen deprivation therapy in patients with prostate cancer
Prostate Cancer and Prostatic Diseases (2023)
-
Gut OncoMicrobiome Signatures (GOMS) as next-generation biomarkers for cancer immunotherapy
Nature Reviews Clinical Oncology (2023)
-
Changes in the gut microbial profile during long-term androgen deprivation therapy for prostate cancer
Prostate Cancer and Prostatic Diseases (2023)
-
Urinary microbiota and prostatic diseases: the key for the lock? A systematic review
Prostate Cancer and Prostatic Diseases (2023)
-
Dysbiotic microbes and how to find them: a review of microbiome profiling in prostate cancer
Journal of Experimental & Clinical Cancer Research (2022)