Abstract
Backgrounds
Aside from Gleason score few factors accurately identify the subset of prostate cancer (PCa) patients at high risk for metastatic progression. We hypothesized that copy number alterations (CNAs), assessed using CpG methylation probes on Illumina Infinium® Human Methylation450 (HM450K) BeadChip arrays, could identify primary prostate tumors with potential to develop metastatic progression.
Methods
Epigenome-wide DNA methylation profiling was performed in surgically resected primary tumor tissues from two cohorts of PCa patients with clinically localized disease who underwent radical prostatectomy (RP) as primary therapy and were followed prospectively for at least 5 years: (1) a Fred Hutchinson (FH) Cancer Research Center-based cohort (n = 323 patients); and (2) an Eastern Virginia (EV) Medical School-based cohort (n = 78 patients). CNAs were identified using the R package ChAMP. Metastasis was confirmed by positive bone scan, MRI, CT or biopsy, and death certificates confirmed cause of death.
Results
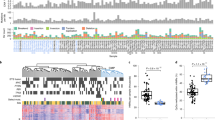
We detected 15 recurrent CNAs were associated with metastasis in the FH cohort and replicated in the EV cohort (p < 0.05) without adjusting for Gleason score in the model. Eleven of the recurrent CNAs were associated with metastatic progression in the FH cohort and validated in the EV cohort (p < 0.05) when adjusting for Gleason score.
Conclusions
This study shows that CNAs can be reliably detected from HM450K-based DNA methylation data. There are 11 recurrent CNAs showing association with metastatic-lethal events following RP and improving prediction over Gleason score. Genes affected by these CNAs may functionally relate to tumor aggressiveness and metastatic progression.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 4 print issues and online access
$259.00 per year
only $64.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2020. CA: A Cancer J Clin. 2020;70:7–30.
Pound CR, Partin AW, Eisenberger MA, Chan DW, Pearson JD, Walsh PC. Natural history of progression after PSA elevation following radical prostatectomy. JAMA.1999;281:1591–7.
Roehl KA, Han M, Ramos CG, Antenor JAV, Catalona WJ. Cancer progression and survival rates following anatomical radical retropubic prostatectomy in 3,478 consecutive patients: long-term results. J Urol. 2004;172:910–4.
Bruce JY, Lang JM, McNeel DG, Liu G. Current controversies in the management of biochemical failure in prostate cancer. Clin Adv Hematol Oncol. 2012;10:716–22.
American Urological Association. 2017 AUA Clinical Guidelines. https://www.auanet.org/guidelines/prostate-cancer-clinically-localized-guideline.
Moyer VA. Screening for prostate cancer: US preventive services task force recommendation statement. Ann Intern Med. 2012;157:120–34.
Delpierre C, Lamy S, Kelly-Irving M, Molinié F, Velten M, Tretarre B, et al. Life expectancy estimates as a key factor in over-treatment: the case of prostate cancer. Cancer Epidemiol. 2013;37:462–8.
Lee YJ, Park JE, Jeon BR, Lee SM, Kim SY, Lee YK. Is prostate-specific antigen effective for population screening of prostate cancer? A systematic review. Ann Lab Med. 2013;33:233–41.
Klotz J. The future of active surveillance. Transl Androl Urol. 2018;7:256–9.
Taylor BS, Schultz N, Hieronymus H, Gopalan A, Xiao Y, Carver BS. et al. Integrative genomic profiling of human prostate cancer. Cancer Cell.2010;18:11–22.
Cancer Genome Atlas Research Network. The molecular taxonomy of primary prostate cancer. Cell. 2015;163:1011–25.
Grasso CS, Wu Y, Robinson DR, Cao X, Dhanasekaran SM, Khan AP, et al. The mutational landscape of lethal castrate resistant prostate cancer. Nature. 2012;487:239–43.
Robinson D, Van Allen EM, Wu Y, Schultz M, Lonigro RJ, Mosquera J, et al. Integrative clinical genomics of advanced prostate cancer. Cell. 2015;161:1215–28.
Liu W. DNA alterations in the tumor genome and their associations with clinical outcome in prostate cancer. Asian J Androl. 2016;18:533–42.
El Gammal AT, Brüchmann M, Zustin J, Isbarn H, Hellwinkel OJ, Köllermann J, et al. Chromosome 8p deletions and 8q gains are associated with tumor progression and poor prognosis in prostate cancer. Clin Cancer Res. 2010;16:56–64.
Kluth M, Runte F, Barow P, Omari J, Abdelaziz ZM, Paustian L, et al. Concurrent deletion of 16q23 and PTEN is an independent prognostic feature in prostate cancer. Int J Cancer. 2015;137:2354–63.
Lalonde E, Ishkanian A, Sykes J. Tumour genomic and microenvironmental heterogeneity for integrated prediction of 5-year biochemical recurrence of prostate cancer: a retrospective cohort study. Lancet Oncol. 2014;15:1521–32.
Choucair K, Ejdelman J, Brimo F, Aprikian A, Chevalier S, Lapointe J. PTEN genomic deletion predicts prostate cancer recurrence and is associated with low AR expression and transcriptional activity. BMC Cancer. 2012;12:543.
Lotan TL, Gurel B, Sutcliffe S, Esopi D, Liu W, Xu J, et al. PTEN protein loss by immunostaining: analytic validation and prognostic indicator for a high risk surgical cohort of prostate cancer patients. Clin Cancer. 2011;17:6563–73.
Liu W, Xie CC, Thomas CY, Kim ST, Lindberg J, Egevad L, et al. Genetic markers associated with early cancer‐specific mortality following prostatectomy. Cancer. 2013;119:2405–12.
Hieronymus H, Schultz N, Gopalan A, Carver BS, Chang MT, Xiao Y, et al. Copy number alteration burden predicts prostate cancer relapse. Proc Natl Acad Sci. 2014;111:11139–44.
Feber A, Guilhamon P, Lechner M, Fenton T, Wilson GA, Thirlwell C. Using high-density DNA methylation arrays to profile copy number alterations. Genome Biol. 2014;15:R30.
Nordlund J, Bäcklin CL, Zachariadis V, Cavelier L, Dahlberg J, Öfverholm I, et al. DNA methylation-based subtype prediction for pediatric acute lymphoblastic leukemia. Clin Epigenetics. 2015;7:11.
Kwee I, Rinaldi A, Rancoita P, Rossi D, Capello D, Forconi F, et al. Integrated DNA copy number and methylation profiling of lymphoid neoplasms using a single array. Br J Haematol. 2012;156:354–7.
Chao S, Kim H, Zeiger MA, Umbricht CB, Cope LM. Measuring DNA copy number variation using high-density methylation microarrays. J Comput Biol. 2019;26:295–304.
Rubicz R, Zhao S, Wright JL, Coleman L, Grasso CS, Geybels MS, et al. Gene expression panel predicts metastatic‐lethal prostate cancer outcomes in men diagnosed with clinically localized prostate cancer. Mol Oncol. 2017;11:140–50.
Cheng A, FitzGerald LM, Wright JL, Kolb S, Karnes RJ, Kenkins RB, et al. A four-gene transcript score to predict metastatic-lethal progression in men treated for localized prostate cancer: development and validation studies. Prostate. 2019;79:1589–96.
Zhao S, Geybels MS, Leonardson A, Rubicz R, Kolb S, Yan Q, et al. Epigenome-wide tumor DNA methylation profiling identifies novel prognostic biomarkers of metastatic-lethal progression in men diagnosed with clinically localized prostate cancer. Clin Cancer Res. 2017;23:311–9.
Zhao S, Leonardson A, Geybels MS, McDaniel AS, Yu M, Kolb S, et al. A five-CpG DNA methylation score to predict metastatic‐lethal outcomes in men treated with radical prostatectomy for localized prostate cancer. Prostate. 2018;78:1084–91.
Stanford JL, Wicklund KG, McKnight B, Daling JR, Brawer MK. Vasectomy and risk of prostate cancer. Cancer Epidemiol Biomark Prev. 1999;8:881–6.
Agalliu I, Salinas CA, Hansten PD, Ostrander EA, Stanford JL. Statin use and risk of prostate cancer: results from a population-based epidemiologic study. Am J Epidemiol. 2008;168:250–60.
Bibikova M, Barnes B, Tsan C, Ho V, Klotzle B, Le JM, et al. High density DNA methylation array with single CpG site resolution. Genomics. 2011;98:288–95.
Morris TJ, Butcher LM, Feber A. ChAMP: 450k chip analysis methylation pipeline. Bioinformatics. 2014;30:428–30.
Tian Y, Morris TJ, Webster AP. ChAMP: updated methylation analysis pipeline for Illumina BeadChips. Bioinformatics. 2017;33:3982–4.
Seshan VE, Olshen A, DNAcopy: DNA copy number data analysis. R package version 1.56.0;2018.
Merme lGH, Schumacher SE, Hill B. GISTIC2.0 facilitates sensitive and confident localization of the targets of focal somatic copy-number alteration in human cancers. Genome Biol. 2011;12:R41.
Robin X, Turck N, Hainard A, Tiberti N, Lisacek F, Sanchez JZ. pROC: an open-source package for R and S+ to analyze and compare ROC curves. BMC Bioinform. 2011;12:77.
Whitton B, Okamoto H, Packham G, Crabb SJ. Vacuolar ATPase as a potential therapeutic target and mediator of treatment resistance in cancer. Cancer Med. 2018;7:3800–11.
Li B, Thrasher JB, Terranova P. Glycogen synthase kinase-3: a potential preventive target for prostate cancer management. Urol Oncol. 2015;33:456–63.
Kobayashi N, Uemura H, Nagahama K. Identification of miR-30d as a novel prognostic maker of prostate cancer. Oncotarget. 2012;3:1455–71.
Song Y, Song C, Yang S. Tumor-suppressive function of miR-30d-5p in prostate cancer cell proliferation and migration by targeting NT5E. Cancer Biother Radiopharm. 2018;33:203–11.
Wang R, Asangani IA, Chakravarthi BV, Ateeq B, Lonigro RJ, Cao Q, et al. Role of transcriptional corepressor CtBP1 in prostate cancer progression. Neoplasia. 2012;14:905–14.
Bluemn EG, Spencer ES, Mecham B, Gordon RR, Coleman I, Lewinshtein D, et al. PPP2R2C loss promotes castration-resistance and is associated with increased prostate cancer-specific mortality. Mol Cancer Res. 2013;11:568–78.
Vainio P, Gupta S, Ketola K, Mirtti T, Mpindi J, Kohonen P, et al. Arachidonic acid pathway members PLA2G7, HPGD, EPHX2, and CYP4F8 identified as putative novel therapeutic targets in prostate cancer. Am J Pathol. 2011;178:525–36.
Chu IM, Hengst L, Slingerland JM. The Cdk inhibitor p27 in human cancer: prognostic potential and relevance to anticancer therapy. Nat Rev Cancer. 2008;8:253–67.
Zhang Z, She J, Yang J. NDRG4 in gastric cancer determines tumor cell proliferation and clinical outcome. Mol Carcinog. 2018;57:762–71.
Hail N, Chen P, Bushman LR. Teriflunomide (leflunomide) promotes cytostatic, antioxidant, and apoptotic effects in transformed prostate epithelial cells: evidence supporting a role for teriflunomide in prostate cancer chemoprevention. Neoplasia. 2010;12:464–75.
Yang L, Ravindranathan P, Ramanan M, Kapur P, Hammes SR, Hsieh J, et al. Central role for PELP1 in nonandrogenic activation of the androgen receptor in prostate cancer. Mol Endocrinol. 2012;26:550–61.
Kluth M, Jung S, Habib O, Eshagzaiy M, Heinl A, Amschler N, et al. Deletion lengthening at chromosomes 6q and 16q targets multiple tumor suppressor genes and is associated with an increasingly poor prognosis in prostate cancer. Oncotarget. 2017;8:108923–35.
Osman I, Scher H, Daibacni G, Reuter V, Zhang Z, Cordon-Cardo C. Chromosome 16 in primary prostate cancer: a microsatellite analysis. Int J Cancer. 1997;71:580–4.
Acknowledgements
This work was supported by grants from the National Cancer Institute (R01 CA222833, R01 CA056678, R01 CA092579, K05 CA175147 (JLS), and P50 CA097186), with additional support provided by the Fred Hutchinson Cancer Research Center (P30 CA015704). We acknowledge the support of the Eastern Virginia Medical School Biorepository, Norfolk, Virginia.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Wang, X., Grasso, C.S., Jordahl, K.M. et al. Copy number alterations are associated with metastatic-lethal progression in prostate cancer. Prostate Cancer Prostatic Dis 23, 494–506 (2020). https://doi.org/10.1038/s41391-020-0212-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41391-020-0212-8
This article is cited by
-
Acquired copy number variation in prostate tumours: a review of common somatic copy number alterations, how they are formed and their clinical utility
British Journal of Cancer (2024)
-
GATA2 co-opts TGFβ1/SMAD4 oncogenic signaling and inherited variants at 6q22 to modulate prostate cancer progression
Journal of Experimental & Clinical Cancer Research (2023)
-
A DNA copy number alteration classifier as a prognostic tool for prostate cancer patients
British Journal of Cancer (2023)
-
Immunohistochemical markers as predictors of prognosis in multifocal prostate cancer
Virchows Archiv (2023)
-
Copy number variation of urine exfoliated cells by low-coverage whole genome sequencing for diagnosis of prostate adenocarcinoma: a prospective cohort study
BMC Medical Genomics (2022)