Abstract
Background
To review the evidence available to support clinical practice guidelines for dietary interventions aimed at mitigating the side effects of androgen deprivation therapy (ADT) in men with prostate cancer, and to identify future research priorities.
Methods
An analytical model was designed to select and interpret evidence for the effect of dietary interventions on ADT side effects. Key terms identified articles that investigated dietary interventions to mitigate ADT side effects among men treated for prostate cancer. Medline, Embase, Proquest, CINAHL, Cochrane databases, and PubMed were searched from inception through June, 2018. Clinical trial registries were also searched for up-to-date study protocols. Articles were not restricted on design. Methodological quality was assessed using the mixed methods appraisal tool.
Results
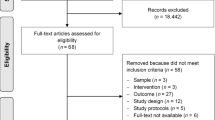
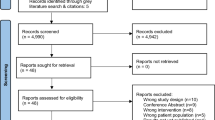
Sixteen articles met inclusion criteria, each with distinct dietary interventions. Twelve studies used interventions that combined diet with physical activity and/or medication and/or counselling. Four articles examined the effect of diet alone on ADT side effects. Of those, three articles measured changes to participants’ dietary intake and influence on ADT side effects. One article showed daily caffeinated beverages improved cancer-related fatigue. Two articles showed no impact of isoflavone supplementation on hot flushes, quality of life, body mass index, or blood lipids. Dietary intake and compliance was poorly reported across all studies limiting knowledge of acceptability and feasibility for dietary interventions. Information on the nutrition care practices and views of clinicians treating men for prostate cancer is limited. No articles measured the impact of diet on long-term ADT side effects. Methodological quality of included papers ranged from weak to strong.
Conclusions
Current evidence for dietary interventions to mitigate ADT side effects is limited. Further investigations are warranted to explore the impact of changes in dietary intake on ADT side effects before practice guidelines can be considered.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 4 print issues and online access
$259.00 per year
only $64.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Wong MC, Goggins WB, Wang HH, et al. Global incidence and mortality for prostate cancer: analysis of temporal patterns and trends in 36 countries. Eur Urol. 2016;70:862–74.
Andrology Australia. Androgen Deprivation Therapy (ADT). 2018. [Available from: https://www.andrologyaustralia.org/androgen-deprivation-therapy/
Kumar R, Barqawi A, Crawford D. Adverse events associated with hormonal therapy for prostate cancer. Rev Urol. 2005;7(Suppl5):S37–43.
Galvão DA, Spry NA, Taaffe DR, et al. Changes in muscle, fat and bone mass after 36 weeks of maximal androgen blockade for prostate cancer. BJU Int. 2008;102:44–7.
Moyad M, Newton R, Tunn U, Gruca D. Integrating diet and exercise into care of prostate cancer patients on androgen deprivation therapy. Res Rep Urol. 2016;8:133–43.
Abete I, Astrup A, Martines J, Thorsdottir I, Zulet M. Obesity and the metabolic syndrome: role of different dietary macrnonutrient distribution patterns and specific nutritional components on weight loss and maintenance. Nutr Rev. 2010;68:214–31.
Prentice A. Diet, nutrition and the prevention of osteoporosis. Public Health Nutr. 2004;7(1A):227–43.
Lai J, Hiles S, Bisquera A, Hure A, McEvoy M, Attia J. A systematic review and meta-anlysis of dietary patterns and depression in community-dwelling adults. Am J Clin Nutr. 2013;99:181–97.
National Heart Foundation of Australia and the Cardiac Society of Australia and New Zealand. Reducing risk in heart disease: an expert guide to clinical practice for secondary prevention of coronary heart disease. Melbourne: National Heart Foundation; 2012.
Tivesten Å, Pinthus JH, Clarke N, Duivenvoorden W, Nilsson J, Cardiovascular risk with androgen deprivation therapy for prostate cancer: potential mechanisms. Urol Oncol. 2015;33:464–75.
Morote J, Gómez-Caamaño A, Alvarez-Ossorio JL, et al. The metabolic syndrome and its components in patients with prostate cancer on androgen deprivation therapy. J Urol. 2015;193:1963–9.
Baguley BJ, Bolam KA, Wright OR, Skinner TL. The effect of nutrition therapy and exercise on cancer-related fatigue and quality of life in men with prostate cancer: a systematic review. Nutrients. 2017;9:1003.
Turner L, Poole K, Faithfull S, Griffin BA. Current and future strategies for the nutritional management of cardiometabolic complications of androgen deprivation therapy for prostate cancer. Nutr Res Rev. 2017;30:220–32.
Owen PJ, Daly RM, Livingston PM, Fraser SF. Lifestyle guidelines for managing adverse effects on bone health and body composition in men treated with androgen deprivation therapy for prostate cancer: An update. Prostate Cancer and Prostatic Diseases. 2017;20:137–45.
Bourke L, Sohanpal R, Nanton V, Crank H, Rosario DJ, Saxton JM. A qualitative study evaluating experiences of a lifestyle intervention in men with prostate cancer undergoing androgen suppression therapy. Trials. 2012;13:208.
Lebret T, Coloby P, Descotes JL, Droupy S, Geraud M, Tombal B. Educational tool-kit on diet and exercise: survey of prostate cancer patients about to receive androgen deprivation therapy. Urology. 2010;76:1434–9.
Wynn K, Trudeau J, Taunton K, Gowans M, Scott I. Nutrition in primary care: current practices, attitudes, and barriers. Can Fam Physician. 2010;56:e109e16.
Crowley J, Ball L, McGill A-T, et al. General practitioners’ views on providing nutrition care to patients with chronic disease: a focus group study. J Prim Health Care. 2016;8:357–64.
Field M, Lohr K. Committee to Advise the Public Health Service on Clinical Practice Guidelines. In: Institute of Medicine, editor. Clinical practice guidelines: directions for a new program. Washington D.C.: National Academy Press; 1990.
Eccles M, Grimshaw J, Sjekelle P, Schunemann H, Woolfe S. Developing clinical practice guideloines: target audiences, identifying topics for guidelines, gduieline group compostion and functioning and conflicts of interest. Implement Sci. 2012;7:60–68.
Woolfe S, Schunemann H, Eccles M, Grimshaw J, Shekelle P. Developing clinical practice guidelines: types of evidence and outcomes; values and economics; synthesis, grading and presentation and deriving recommendations. Implement Sci. 2012;7:61.
Shekelle P, Woolfe S, Grimshaw J, Schunemann H, Eccles M. Developing clinical practice guidelines: reviewing reporting and publishing guidelines; updating guidelines; and the emerging issues of enghancing guidleines implementability and accounting for comorbid conditions in guideline development. Implement Sci. 2012;7:62.
Moher D, Liberati A, Tetzlaff J, Altman D, Group. P. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009;6:e1000097.
World Health Organisation. International Clinical Trials Registry Platform Search Portal. 2018. Available from: http://apps.who.int/trialsearch/.
U.S. National Library of Medicine. ClinicalTrials.gov. 2018. Available from: https://clinicaltrials.gov/.
Cochrane Library. Cochrane Central Register of Controlled Clinical Trials. 2018. Available from: http://cochranelibrary-wiley.com/cochranelibrary/search?searchRow.searchOptions.searchProducts=clinicalTrialsDoi.
Pace R, Pluye P, Bartlett G, et al. Testing the reliability and efficiency of the pilot Mixed Methods Appraisal Tool (MMAT) for systematic mixed studies review. Int J Nurs Stud. 2012;49:47–53.
Focht BC, Lucas AR, Grainger E, et al. Effects of a group-mediated exercise and dietary intervention in the treatment of prostate cancer patients undergoing androgen deprivation therapy: Results from the IDEA-P trial. Ann Behav Med. 2018;52:412–28.
Kiwata JL, Dorff TB, Todd Schroeder E, et al. A pilot randomised controlled trial of a periodised resistance training and protein supplementation intervention in prostate cancer survivors on androgen deprivation therapy. BMJ Open. 2017;7:e016910.
Napora JK, Short RG, Muller DC, et al. High‐dose isoflavones do not improve metabolic and inflammatory parameters in androgen‐deprived men with prostate cancer. J Androl. 2011;32:40–8.
Richard PO, Abramsky H, Bhatt JR, et al. Impact of energy drinks on androgen deprivation-induced fatigue: results of a pilot study. J Caffeine Res. 2015;5:50–4.
Sharma P, Wisniewski A, Braga-Basaria M, et al. Lack of an effect of high dose isoflavones in men with prostate cancer undergoing androgen deprivation therapy. J Urol. 2009;182:2265–73.
Bourke L, Gilbert S, Hooper R, et al. Lifestyle changes for improving disease-specific quality of life in sedentary men on long-term androgen-deprivation therapy for advanced prostate cancer: a randomised controlled trial. Eur Urol. 2014;65:865–72.
Gilbert SE, Tew GA, Fairhurst C, et al. Effects of a lifestyle intervention on endothelial function in men on long-term androgen deprivation therapy for prostate cancer. Br J Cancer. 2016;114:401–8.
Nobes JP, Langley SEM, Klopper T, Russell-Jones D, Laing RW. A prospective, randomized pilot study evaluating the effects of metformin and lifestyle intervention on patients with prostate cancer receiving androgen deprivation therapy. BJU Int. 2012;109:1495–502.
O’Neill RF, Haseen F, Murray LJ, O’Sullivan JM, Cantwell MM. A randomised controlled trial to evaluate the efficacy of a 6-month dietary and physical activity intervention for patients receiving androgen deprivation therapy for prostate cancer. J Cancer Surviv: Res Pract. 2015;9:431–40.
Mareschal J, Weber K, Rigoli P, et al. The ADAPP trial: a two-year longitudinal multidisciplinary intervention study for prostate cancer frail patients on androgen deprivation associated to curative radiotherapy. Acta Oncol. 2017;56:569–74.
Hanson ED, Nelson AR, West DWD, et al. Attenuation of resting but not load-mediated protein synthesis in prostate cancer patients on androgen deprivation. J Clin Endocrinol Metab. 2017;102:1076–83.
Owen PJ, Daly RM, Livingston PM, et al. Efficacy of a multi-component exercise programme and nutritional supplementation on musculoskeletal health in men treated with androgen deprivation therapy for prostate cancer (IMPACT): study protocol of a randomised controlled trial. Trials. 2017;18:451.
Baguley BJ, Skinner TL, Leveritt MD, Wright ORL. Nutrition therapy with high intensity interval training to improve prostate cancer-related fatigue in men on androgen deprivation therapy: a study protocol. BMC Cancer. 2017;17:1.
Davison BJ, Wiens K, Cushing M. Promoting calcium and vitamin D intake to reduce the risk of osteoporosis in men on androgen deprivation therapy for recurrent prostate cancer. Support Care Cancer. 2012;20:2287–94.
Galvao DA, Taaffe DR, Spry N, Joseph D, Newton RU. Combined resistance and aerobic exercise program reverses muscle loss in men undergoing androgen suppression therapy for prostate cancer without bone metastases: a randomized controlled trial. J Clin Oncol. 2010;28:340–7.
Wall BA, Galvão DA, Fatehee N, et al. Exercise improves V˙ O2max and body composition in androgen deprivation therapy-treated prostate cancer patients. Med Sci Sports Exerc. 2017;49:1503–10.
Galvao D, Taaffe D, Spry N, Newton R. Exercise can prevent and even reverse adverse effects of androgen suppression treatment in men with prostate cancer. Prostate Cancer Prostatic Dis. 2007;10:340.
Wu T, Gao X, Chen M, Van Dam R. Long‐term effectiveness of diet‐plus‐exercise interventions vs. diet‐only interventions for weight loss: a meta‐analysis. Obes Rev. 2009;10:313–23.
Brown T, Avenell A, Edmunds LD, et al. Systematic review of long-term lifestyle interventions to prevent weight gain and morbidity in adults. Obes Rev. 2009;10:627–38.
Acknowledgements
The authors would like to thank Griffith University Health Librarian, Ms Bonnie Dixon, for her assistance in preparing the search strategy. This review was supported by the National Health and Medical Research Council Centre for Research Excellence in Prostate Cancer Survivorship.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Barnes, K.A., Ball, L.E., Galvão, D.A. et al. Nutrition care guidelines for men with prostate cancer undergoing androgen deprivation therapy: do we have enough evidence?. Prostate Cancer Prostatic Dis 22, 221–234 (2019). https://doi.org/10.1038/s41391-018-0099-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41391-018-0099-9