Abstract
Background
Metabolic syndrome and obesity are linked with hyperuricemia, and it has also been proposed that oxidative stress associated with hyperuricemia may promote benign prostatic hyperplasia (BPH). However, it is currently unknown whether use of antihyperuricemic medication is associated with risk of developing BPH. We studied the association between BPH and use of antihyperuricemic allopurinol in a Finnish population-based cohort.
Methods
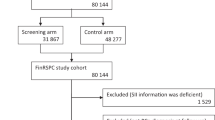
The study cohort consisted of 74,754 men originally identified for the Finnish Randomized Study of Screening for Prostate Cancer (FinRSPC). Information on gout and BPH medication usage (5α-reductase inhibitors, 5ARIs) during 1996–2014 was obtained from the National medication reimbursement database. Information on BPH diagnoses from in- and outpatient hospital visits and BPH-related surgery was obtained from the National Health Care Registry. Men with a record of BPH at baseline was excluded. We used Cox regression to analyze risk of starting BPH medication, having a recorded diagnosis or undergoing BPH surgery by allopurinol use with adjustment for age and simultaneous use of statins, antidiabetic or antihypertensive drugs and aspirin or other NSAIDs. Medication use was analyzed as a time-dependent variable to minimize immortal time bias.
Results
Men using allopurinol had a decreased risk for all three BPH endpoints: BPH medication (HR 0.81; 95% CI 0.75–0.88), BPH diagnosis (HR 0.78; 95% CI 0.71–0.86) and BPH-related surgery (HR 0.67; 95% CI 0.58–0.76) after multivariable adjustment. The risk association did not change by cumulative use. The risk decrease disappeared after 1–2 years lag time. Only BMI modified the risk association; the risk decrease was observed only among men with BMI above the median (27.3 kg/m2); p for interaction <0.05 for each endpoint.
Conclusions
We found that allopurinol use is associated with lowered risk of BPH medication, diagnosis and surgery. A possible explanation could be antioxidative effects of urate-lowering allopurinol.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 4 print issues and online access
$259.00 per year
only $64.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Vignozzi L, Gacci M, Maggi M. Lower urinary tract symptoms, benign prostatic hyperplasia and metabolic syndrome. Nat Rev Urol. 2016;13:108–19.
Hammarsten J, Högstedt B, Holthuis N, Mellström D. Components of the metabolic syndrome-risk factors for the development of benign prostatic hyperplasia. Prostate Cancer Prostatic Dis. 1998;1:157–62.
De Nunzio C, Presicce F, Tubaro A. Inflammatory mediators in the development and progression of benign prostatic hyperplasia. Nat Rev Urol. 2016;13:613–26.
Udensi UK, Tchounwou PB. Oxidative stress in prostate hyperplasia and carcinogenesis. J Exp Clin Cancer Res. 2016. [cited 14 Sep 2017];35. Available at http://www.ncbi.nlm.nih.gov/pmc/articles/PMC5017015/.
Vital P, Castro P, Ittmann M. Oxidative stress promotes benign prostatic hyperplasia. Prostate. 2016;76:58–67.
Sangkop F, Singh G, Rodrigues E, Gold E, Bahn A. Uric acid: a modulator of prostate cells and activin sensitivity. Mol Cell Biochem. 2016;414:187–99.
Ørsted DD, Bojesen SE. The link between benign prostatic hyperplasia and prostate cancer. Nat Rev Urol. 2013;10:49–54.
Chen C-J, Yen J-H, Chang S-J. Gout patients have an increased risk of developing most cancers, especially urological cancers. Scand J Rheumatol. 2014;43:385–90.
Castro GD, Costantini MH, Castro JA. Rat ventral prostate xanthine oxidase-mediated metabolism of acetaldehyde to acetyl radical. Hum Exp Toxicol. 2009;28:203–8.
Zhang Z, Blake DR, Stevens CR, Kanczler JM, Winyard PG, Symons MC, et al. A reappraisal of xanthine dehydrogenase and oxidase in hypoxic reperfusion injury: the role of NADH as an electron donor. Free Radic Res. 1998;28:151–64.
Bove M, Cicero AFG, Veronesi M, Borghi C. An evidence-based review on urate-lowering treatments: implications for optimal treatment of chronic hyperuricemia. Vasc Health Risk Manag. 2017;13:23–8.
Pea F. Pharmacology of drugs for hyperuricemia. Mechanisms, kinetics and interactions. Contrib Nephrol. 2005;147:35–46.
Parikesit D, Mochtar CA, Umbas R, Hamid ARAH. The impact of obesity towards prostate diseases. Prostate Int. 2016;4:1–6.
Kilpeläinen TP, Tammela TL, Malila N, Hakama M, Santti H, Määttänen L, et al. Prostate cancer mortality in the Finnish randomized screening trial. J Natl Cancer Inst. 2013;105:719–25.
Sarre S, Määttänen L, Tammela TLJ, Auvinen A, Murtola TJ. Postscreening follow-up of the Finnish Prostate Cancer Screening Trial on putative prostate cancer risk factors: vitamin and mineral use, male pattern baldness, pubertal development and non-steroidal anti-inflammatory drug use. Scand J Urol. 2016;50:267–73.
WHOCC - ATC/DDD Index. [cited 14 Sep 2017]. Available at https://www.whocc.no/atc_ddd_index/.
Veitonmäki T, Murtola TJ, Määttänen L, Taari K, Stenman U-H, Tammela TLJ, et al. Use of non-steroidal anti-inflammatory drugs and prostate cancer survival in the Finnish prostate cancer screening trial. Prostate. 2015;75:1394–402.
Cao Y, Giovannucci E. Obesity and Prostate Cancer. Recent Results Cancer Res. 2016;208:137–53.
Huang Y, Zhang C, Xu Z, Shen J, Zhang X, Du H, et al. Clinical Study on efficacy of allopurinol in patients with acute coronary syndrome and its functional mechanism. The article is in press. Therefore: Hellenic J Cardiol 2017 (in press)
Ellulu MS. Obesity, cardiovascular disease, and role of vitamin C on inflammation: a review of facts and underlying mechanisms. Inflammopharmacology. 2017;25:313–28.
Glantzounis GK, Tsimoyiannis EC, Kappas AM, Galaris DA. Uric acid and oxidative stress. Curr Pharm Des. 2005;11:4145–51.
Szewczyk-Golec K, Tyloch J, Czuczejko J. Antioxidant defense system in prostate adenocarcinoma and benign prostate hyperplasia of elderly patients. Neoplasma. 2015;62:119–23.
Rupérez AI, Gil A, Aguilera CM. Genetics of oxidative stress in obesity. Int J Mol Sci. 2014;15:3118–44.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
T.L.J. Tammela: consultant fees from Astellas, Bayer, and Roche. T.J. Murtola: consultant fees from Astellasand Janssen Cilag. Lecture fees from Astellas, GSK, and Janssen Cilag. K Taari: Consultant fee from Abbvie, research funding from Medivation, travel support from Astellas, and Orion. Remaining authors declare no conflict of interest.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Kukko, V., Kaipia, A., Talala, K. et al. Allopurinol and risk of benign prostatic hyperplasia in a Finnish population-based cohort. Prostate Cancer Prostatic Dis 21, 373–378 (2018). https://doi.org/10.1038/s41391-017-0031-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41391-017-0031-8
This article is cited by
-
Allopurinol and prostate cancer survival in a Finnish population-based cohort
Prostate Cancer and Prostatic Diseases (2022)
-
Allopurinol and the risk of prostate cancer in a Finnish population-based cohort
Prostate Cancer and Prostatic Diseases (2019)