Abstract
Background
Preterm birth adversely impacts brain development and contributes to neurodevelopmental impairment; the temporal lobe may be particularly vulnerable to the impact of very preterm (VP) birth. Yet, no prior magnetic resonance imaging (MRI) scoring system incorporated a method to quantify temporal lobe size in VP infants.
Methods
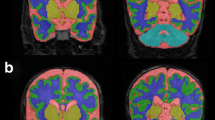
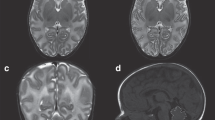
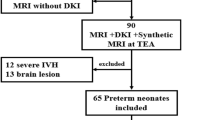
We developed and applied three metrics (temporal lobe length, extra-axial space, and temporal horn width) to quantify temporal lobe structure on term-equivalent brain MRIs obtained from 74 VP and 16 term infants. We compared metrics between VP and term infants and explored associations of each metric with perinatal risk factors.
Results
All metrics had excellent reliability (intra-class correlation coefficient 0.62–0.98). VP infants had lower mean temporal lobe length (76.8 mm versus 79.2 mm, p = 0.02); however, the difference attenuated after correction for postmenstrual age. VP infants had larger temporal horn widths compared with term infants (2.6 mm versus 1.8 mm, p < 0.001). Temporal lobe length was positively associated with gestational age, birth weight, and male sex, and negatively associated with the duration of parenteral nutrition.
Conclusions
The proposed metrics are reliable and sensitive in distinguishing differences in temporal lobe development between VP and full-term infants.
Impact
-
We developed a novel method for quantifying temporal lobe size among very preterm infants at term equivalent using simple metrics performed on brain MRI.
-
Temporal lobe metrics were reliable, correlated with brain volume from volumetric analysis, and were sensitive in identifying differences in temporal lobe development among preterm compared with term infants, specifically larger temporal horn size in preterm infants.
-
This temporal lobe metric system will enable future work to delineate the perinatal and postnatal factors that impact temporal lobe growth, and better understand the relationship between temporal lobe disturbance and neurodevelopment in very preterm infants.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 14 print issues and online access
$259.00 per year
only $18.50 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Data availability
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
References
Cheong, J. L. Y., Spittle, A. J., Burnett, A. C., Anderson, P. J. & Doyle, L. W. Have outcomes following extremely preterm birth improved over time. Semin. Fetal Neonatal Med. 25, 101114 (2020).
Marlow, N. et al. No change in neurodevelopment at 11 years after extremely preterm birth. Arch. Dis. Child. Fetal Neonatal Ed. 106, 418–424 (2021).
Anderson, P., Doyle, L. W. & Victorian Infant Collaborative Study Group. Neurobehavioral outcomes of school-age children born extremely low birth weight or very preterm in the 1990s. JAMA 289, 3264 (2003).
Hack, M. et al. Chronic conditions, functional limitations, and special health care needs of school-aged children born with extremely low-birth-weight in the 1990s. JAMA 294, 318 (2005).
Erdei, C., Austin, N. C., Cherkerzian, S., Morris, A. R. & Woodward, L. J. Predicting school-aged cognitive impairment in children born very preterm. Pediatrics 145, e20191982 (2020).
Woodward, L. J., Horwood, L. J., Darlow, B. A. & Bora, S. Visuospatial working memory of children and adults born very preterm and/or very low birth weight. Pediatr. Res. 91, 1436–1444 (2022).
Eves, R. et al. Association of very preterm birth or very low birth weight with intelligence in adulthood: an individual participant data meta-analysis. JAMA Pediatr. 175, e211058 (2021).
Robinson R. et al. ADHD symptoms and diagnosis in adult preterms: systematic review, IPD meta-analysis, and register-linkage study. Pediatr. Res. https://doi.org/10.1038/s41390-021-01929-1 (2022).
Woodward, L. J., Clark, C. A. C., Bora, S. & Inder, T. E. Neonatal white matter abnormalities an important predictor of neurocognitive outcome for very preterm children. PLoS ONE 7, e51879 (2012).
Woodward, L. J., Anderson, P. J., Austin, N. C., Howard, K. & Inder, T. E. Neonatal MRI to predict neurodevelopmental outcomes in preterm infants. N. Engl. J. Med. 355, 685–694 (2006).
Belfort, M. B. et al. Infant growth before and after term: effects on neurodevelopment in preterm infants. Pediatrics 128, e899–e906 (2011).
Logan, J. W. et al. Adverse effects of perinatal illness severity on neurodevelopment are partially mediated by early brain abnormalities in infants born very preterm. J. Perinatol. 41, 519–527 (2021).
Tierney, A. L. & Nelson, C. A. Brain development and the role of experience in the early years. Zero Three 30, 9–13 (2009).
Pineda, R. G. et al. Alterations in brain structure and neurodevelopmental outcome in preterm infants hospitalized in different neonatal intensive care unit environments. J. Pediatr. 164, 52.e2–60.e2 (2014).
Pineda, R. G. et al. Enhancing sensory experiences for very preterm infants in the NICU: an integrative review. J. Perinatol. 37, 323–332 (2017).
Peterson, B. S. et al. Regional brain volumes and their later neurodevelopmental correlates in term and preterm infants. Pediatrics 111, 939–948 (2003).
Tich S. N. T. et al. Neurodevelopmental and perinatal correlates of simple brain metrics in very preterm infants. Arch. Pediatr. Adolesc. Med. 165, 216–222 (2011).
Cheong, J. L. Y. et al. Brain volumes at term-equivalent age are associated with 2-year neurodevelopment in moderate and late preterm children. J. Pediatr. 174, 91.e1–97.e1 (2016).
Rogers, C. E. et al. Regional cerebral development at term relates to school-age social-emotional development in very preterm children. J. Am. Acad. Child Adolesc. Psychiatry 51, 181–191 (2012).
Inder, T. E. et al. Neuroimaging of the preterm brain: review and recommendations. J. Pediatr. 237, 276.e4–287.e4 (2021).
Kesler, S. R. et al. Increased temporal lobe gyrification in preterm children. Neuropsychologia 44, 445–453 (2006).
Peterson, B. S. et al. Regional brain volume abnormalities and long-term cognitive outcome in preterm infants. JAMA 284, 1939 (2000).
Kesler, S. R. et al. Volumetric analysis of regional cerebral development in preterm children. Pediatr. Neurol. 31, 318–325 (2004).
Thompson, D. K. et al. Neonate hippocampal volumes: prematurity, perinatal predictors, and 2-year outcome. Ann. Neurol. 63, 642–651 (2008).
Thompson, D. K. et al. Hippocampal shape variations at term equivalent age in very preterm infants compared with term controls: perinatal predictors and functional significance at age 7. Neuroimage 70, 278–287 (2013).
Thompson, D. K. et al. Longitudinal growth and morphology of the hippocampus through childhood: Impact of prematurity and implications for memory and learning. Hum. Brain Mapp. 35, 4129–4139 (2014).
Jansen, L. et al. Associations between neonatal magnetic resonance imaging and short- and long-term neurodevelopmental outcomes in a longitudinal cohort of very preterm Children. J. Pediatr. 234, 46.e2–53.e2 (2021).
Kidokoro, H. et al. Brain injury and altered brain growth in preterm infants: predictors and prognosis. Pediatrics 134, e444–e453 (2014).
Kidokoro, H., Neil, J. J. & Inder, T. E. New MR imaging assessment tool to define brain abnormalities in very preterm infants at term. AJNR Am. J. Neuroradiol. 34, 2208–2214 (2013).
Nguyen The Tich, S. et al. A novel quantitative simple brain metric using MR imaging for preterm infants. AJNR Am. J. Neuroradiol. 30, 125–131 (2009).
Bell, K. A. et al. Associations of body composition with regional brain volumes and white matter microstructure in very preterm infants. Arch. Dis. Child. Fetal Neonatal Ed. 107, 533–538 (2022).
Fedorov, A. et al. 3D Slicer as an image computing platform for the Quantitative Imaging Network. Magn. Reson. Imaging 30, 1323–1341 (2012).
Yushkevich, P. A. et al. User-guided 3D active contour segmentation of anatomical structures: Significantly improved efficiency and reliability. Neuroimage 31, 1116–1128 (2006).
Beare R. J. et al. Neonatal brain tissue classification with morphological adaptation and unified segmentation. Front. Neuroinform. 10, 12 (2016).
Vermont Oxford Network. 2019 Vermont Oxford Network Manual of Operations, Part 2, Release 23.2. https://vtoxford.zendesk.com/hc/en-us/articles/360013115393-2019-Manual-of-Operations-Part-2-Release-23-2-PDF (2019).
Koo, T. K. & Li, M. Y. A guideline of selecting and reporting intraclass correlation coefficients for reliability research. J. Chiropr. Med. 15, 155–163 (2016).
Isaacs, E. B. et al. Hippocampal volume and everyday memory in children of very low birth weight. Pediatr. Res. 47, 713–720 (2000).
Cheong et al. Contribution of brain size to IQ and educational underperformance in extremely preterm adolescents. PLoS ONE 8, e77475 (2013).
Keunen, K. et al. Brain tissue volumes in preterm infants: prematurity, perinatal risk factors and neurodevelopmental outcome: a systematic review. J. Matern. Fetal Neonatal Med. 25, 89–100 (2012).
Volpe, J. J. Dysmaturation of premature brain: importance, cellular mechanisms, and potential interventions. Pediatr. Neurol. 95, 42–66 (2019).
Utsunomiya, H., Takano, K., Okazaki, M. & Mitsudome, A. Development of the temporal lobe in infants and children: analysis by MR-based volumetry. AJNR Am. J. Neuroradiol. 20, 717–723 (1999).
Bell, B., Lin, J. J., Seidenberg, M. & Hermann, B. The neurobiology of cognitive disorders in temporal lobe epilepsy. Nat. Rev. Neurol. 7, 154–164 (2011).
Ramos-Escobar, N. et al. Hippocampal and auditory contributions to speech segmentation. Cortex 150, 1–11 (2022).
Dien, J. et al. fMRI characterization of the language formulation area. Brain Res. 1229, 179–192 (2008).
Gallagher, M. & Chiba, A. A. The amygdala and emotion. Curr. Opin. Neurobiol. 6, 221–227 (1996).
Phelps, E. A. Human emotion and memory: interactions of the amygdala and hippocampal complex. Curr. Opin. Neurobiol. 14, 198–202 (2004).
Rogers, C. E. et al. Regional cerebral development at term relates to school-age social-emotional development in very preterm children. J. Am. Acad. Child Adolesc. Psychiatry 51, 181–191 (2012).
Kersbergen et al. Longitudinal regional brain development and clinical risk factors in extremely preterm infants. J. Pediatr. 178, 93.e6–100.e6 (2016).
Belfort, M. B. & Ramel, S. E. NICU diet, physical growth and nutrient accretion, and preterm infant brain development. Neoreviews 20, e385–e396 (2019).
Alexander, B. et al. Changes in neonatal regional brain volume associated with preterm birth and perinatal factors. Neuroimage 185, 654–663 (2019).
Maitre, N. L. et al. The dual nature of early-life experience on somatosensory processing in the human infant brain. Curr. Biol. 27, 1048–1054 (2017).
Pineda, R. G. et al. Alterations in brain structure and neurodevelopmental outcome in preterm infants hospitalized in different neonatal intensive care unit environments. J. Pediatr. 164, 52.e2–60.e2 (2014).
Acknowledgements
We thank the infants and their families who participated in this study.
Funding
Portions of this study were supported by a Marshall Klaus Perinatal Research Award from the American Academy of Pediatrics (KAB); Brigham and Women’s Hospital Department of Pediatric Newborn Medicine and Stork Fund, and the Brigham Research Institute Fund to Sustain Research Excellence (MBB); and the Harvard Clinical and Translational Science Center (National Center for Advancing Translational Science, grants 1UL1TR001102 and 1UL1TR002541-01). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
Author information
Authors and Affiliations
Contributions
C.E. and T.E.I. conceptualized the study; T.E. and M.B.B. contributed to data acquisition; C.E., K.A.B., A.G., and C.B. analyzed the data; all authors contributed to data interpretation; C.E., K.A.B., A.A.G., and C.B. wrote the initial manuscript draft; all authors provided critical review and editing of the manuscript and approved the final draft.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
The parents of all infants participating in this study provided written informed consent to undergo brain magnetic resonance imaging for research purposes, under protocols approved by the Mass General Brigham Institutional Review Board.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Erdei, C., Bell, K.A., Garvey, A.A. et al. Novel metrics to characterize temporal lobe of very preterm infants on term-equivalent brain MRI. Pediatr Res 94, 979–986 (2023). https://doi.org/10.1038/s41390-023-02567-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41390-023-02567-5
This article is cited by
-
A systematic review of immune-based interventions for perinatal neuroprotection: closing the gap between animal studies and human trials
Journal of Neuroinflammation (2023)
-
Predicting neurodevelopmental outcome in children born very preterm – does neonatal MRI have a role?
Pediatric Research (2023)