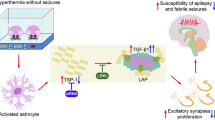
Abstract
Background
Neonatal epileptic seizures cause postictal dysregulation of cerebral blood flow. Hydrogen sulfide (H2S), a mediator with vasodilator and antioxidant properties, is produced in the brain by astrocyte cystathionine β-synthase (CBS). This study investigated whether H2S improves the cerebral vascular outcome of seizures.
Methods
Epileptic seizures were induced in newborn pigs using bicuculline. The effects of the CBS inhibitor aminooxyacetate (AOA) and the H2S donor NaHS on cerebral vascular outcome of seizures were examined in live pigs, cerebral endothelial cells, and cortical astrocytes.
Results
Brain H2S was elevated during seizures. AOA blocked H2S and reduced functional hyperemia in the epileptic brain. The endothelium- and astrocyte-dependent vasodilation of pial arterioles was impaired 48 h after seizures suggesting cerebral vascular dysfunction. Systemic NaHS elevated brain H2S and blocked reactive oxygen species in the epileptic brain and in primary endothelial cells and astrocytes during inflammatory and excitotoxic conditions. Postictal cerebrovascular dysfunction was exaggerated in H2S-inhibited pigs and minimized in NaHS-treated pigs.
Conclusions
H2S elevation in the epileptic brain via activation of CBS contributes to functional hyperemia and exhibits cerebroprotective properties. The H2S donor NaHS enhances brain antioxidant defense and provides a therapeutic approach for preventing adverse cerebral vascular outcome of neonatal epileptic seizures.
Impact
-
Epileptic seizures in neonates lead to prolonged postictal cerebral vascular dysregulation.
-
The role of hydrogen sulfide (H2S), a mediator with vasodilator and antioxidant properties, in the epileptic brain has been explored.
-
Astrocytes are major sites of enzymatic H2S production in the epileptic brain.
-
Postictal cerebral vascular dysfunction is exaggerated when astrocyte H2S production is pharmacologically inhibited during seizures.
-
Postictal cerebral vascular dysfunction is minimized when the brain H2S is elevated by systemic administration of NaHS during seizures.
-
NaHS provides a therapeutic approach for improving cerebrovascular outcome of epileptic seizures via a mechanism that involves the antioxidant potential of H2S.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 14 print issues and online access
$259.00 per year
only $18.50 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study. All data generated or analyzed during this study are included in this published article.
References
Kimura, H. Hydrogen sulfide (H2S) and polysulfide (H2Sn) signaling: the first 25 years. Biomolecules 11, 896 (2021).
Szabo, C. A timeline of hydrogen sulfide (H2S) research: From environmental toxin to biological mediator. Biochem. Pharmacol. 149, 5–19 (2018).
Enokido, Y. et al. Cystathionine beta-synthase, a key enzyme for homocysteine metabolism, is preferentially expressed in the radial glia/astrocyte lineage of developing mouse CNS. FASEB J. 19, 1854–1856 (2005).
Zuhra, K., Augsburger, F., Majtan, T. & Szabo, C. Cystathionine-β-synthase: molecular regulation and pharmacological inhibition. Biomolecules 10, 697 (2020).
Lechpammer, M. et al. Upregulation of cystathionine β-synthase and p70S6K/S6 in neonatal hypoxic ischemic brain injury. Brain Pathol. 27, 449–458 (2017).
Leffler, C. W. et al. Hydrogen sulfide and cerebral microvascular tone in newborn pigs. Am. J. Physiol. Heart Circ. Physiol. 300, H440–H456 (2011).
Liang, G. H. et al. Hydrogen sulfide dilates cerebral arterioles by activating smooth muscle cell plasma membrane KATP channels. Am. J. Physiol. Heart Circ. Physiol. 300, H2088–H2095 (2011).
Liang, G. H., Xi, Q., Leffler, C. W. & Jaggar, J. H. Hydrogen sulfide activates Ca2+ sparks to induce cerebral arteriole dilatation. J. Physiol. 590, 2709–2720 (2012).
Wang, R. Physiological implications of hydrogen sulfide: a whiff exploration that blossomed. Physiol. Rev. 92, 791–896 (2012).
Yang, G. et al. H2S as a physiologic vasorelaxant: hypertension in mice with deletion of cystathionine gamma-lyase. Science 322, 587–590 (2008).
Zhao, W. & Wang, R. H2S-induced vasorelaxation and underlying cellular and molecular mechanisms. Am. J. Physiol. Heart Circ. Physiol. 283, H474–H480 (2002).
Calvert, J. W., Coetzee, W. A. & Lefer, D. J. Novel insights into hydrogen sulfide-mediated cytoprotection. Antioxid. Redox Signal. 12, 1203–1217 (2010).
Kabil, O., Motl, N. & Banerjee, R. H2S and its role in redox signaling. Biochim. Biophys. Acta 1844, 1355–1366 (2014).
Bai, C. & Zhao, C. Sodium hydrosulfide post-conditioning protects hippocampal CA1 neurons from neuronal cell injury in the rat model of transient global cerebral ischemia through activation of extracellular-regulated kinases signaling. Curr. Neurovasc. Res. 16, 156–165 (2019).
Jia, J., Li, J. & Cheng, J. H2S-based therapies for ischaemic stroke: opportunities and challenges. Stroke Vasc. Neurol. 4, 63–66 (2019).
Kamat, P. K. et al. Hydrogen sulfide attenuates neurodegeneration and neurovascular dysfunction induced by intracerebral-administered homocysteine in mice. Neuroscience 252, 302–319 (2013).
Harsono, M. et al. Selective head cooling during neonatal seizures prevents postictal cerebral vascular dysfunction without reducing epileptiform activity. Am. J. Physiol. Heart Circ. Physiol. 311, H1202–H1213 (2016).
Liu, D., Pourcyrous, M., Fedinec, A. L., Leffler, C. W. & Parfenova, H. Preventing harmful effects of epileptic seizures on cerebrovascular functions in newborn pigs: Does sex matter? Pediatr. Res. 82, 881–887 (2017).
Parfenova, H., Leffler, C. W., Basuroy, S., Liu, J. & Fedinec, A. L. Antioxidant roles of heme oxygenase, carbon monoxide, and bilirubin in cerebral circulation during seizures. J. Cereb. Blood Flow. Metab. 32, 1024–1034 (2012).
Parfenova, H., Daley, M. L., Carratu, P. & Leffler, C. W. Heme oxygenase inhibition reduces neuronal activation evoked by bicuculline in newborn pigs. Brain Res. 1014, 87–96 (2004).
Liu, J., Chandaka, G. K., Zhang, R. & Parfenova, H. Acute antioxidant and cytoprotective effects of sulforaphane in brain endothelial cells and astrocytes during inflammation and excitotoxicity. Pharmacol. Res. Perspect. 8, e00630 (2020).
Basuroy, S., Tcheranova, D., Bhattacharya, S., Leffler, C. W. & Parfenova, H. Nox4 NADPH oxidase–derived reactive oxygen species, via endogenous carbon monoxide, promote survival of brain endothelial cells during TNF-a-induced apoptosis. Am. J. Physiol. Cell Physiol. 300, C256–C265 (2011).
Murphy, M. P. et al. Guidelines for measuring reactive oxygen species and oxidative damage in cells and in vivo. Nat. Metab. 4, 651–662 (2022).
Parfenova, H., Liu, J., Hoover, D. T. & Fedinec, A. L. Vasodilator effects of sulforaphane in cerebral circulation: A critical role of endogenously produced hydrogen sulfide and arteriolar smooth muscle KATP and BK channels in the brain. J. Cereb. Blood Flow. Metab. 40, 1987–1996 (2020).
Patel, S. et al. H2S mediates the vasodilator effect of endothelin-1 in the cerebral circulation. Am. J. Physiol. Heart Circ. Physiol. 315, H1759–H1764 (2018).
van Elmpt, W. J., Nijsen, T. M., Griep, P. A. & Arends, J. B. A model of heart rate changes to detect seizures in severe epilepsy. Seizure 15, 366–375 (2006).
Greene, B. R. et al. Heart and respiration rate changes in the neonate during electroencephalographic seizure. Med. Biol. Eng. Comput. 44, 27–34 (2006).
Shao, L. R. & Stafstrom, C. E. Pediatric epileptic encephalopathies: pathophysiology and animal models. Semin. Pediatr. Neurol. 23, 98–107 (2016).
Abend, N. S. & Wusthoff, C. J. Neonatal seizures and status epilepticus. J. Clin. Neurophysiol. 29, 441–448 (2012).
Chapman, K. E., Raol, Y. H. & Brooks-Kayal, A. Neonatal seizures: controversies and challenges in translating new therapies from the lab to the isolette. Eur. J. Neurosci. 35, 1857–1865 (2012).
Lombroso, C. T. Neonatal seizures: gaps between the laboratory and the clinic. Epilepsia 48, 83–106 (2007).
Pestana, R. R., Kinjo, E. R., Hernandes, M. S. & Britto, L. R. Reactive oxygen species generated by NADPH oxidase are involved in neurodegeneration in the pilocarpine model of temporal lobe epilepsy. Neurosci. Lett. 484, 187–191 (2010).
Waldbaum, S. & Patel, M. Mitochondria, oxidative stress, and temporal lobe epilepsy. Epilepsy Res. 88, 23–45 (2010).
Nam, B. et al. In vivo detection of hydrogen sulfide in the brain of live mouse: application in neuroinflammation models. Eur. J. Nucl. Med. Mol. Imaging 49, 4073–4087 (2022).
Funding
The works were supported by awards NS101717 (to H.P.) and NS105655 (to H.P.) from the National Institutes of Health.
Author information
Authors and Affiliations
Contributions
H.P., M.P., and J.L. conceived and designed research; J.L. and A.F. performed experiments; H.P., J.L., and A.F. analyzed data; H.P., M.P., and J.L. interpreted results of experiments; H.P., M.P., and J.L. drafted, edited, and revised manuscript; all authors approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, J., Pourcyrous, M., Fedinec, A.L. et al. Cerebroprotective actions of hydrogen sulfide in the epileptic brain in newborn pigs. Pediatr Res 94, 495–502 (2023). https://doi.org/10.1038/s41390-023-02486-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41390-023-02486-5