Abstract
Background
Studies in newborns with mild neonatal encephalopathy (mNE) demonstrated normal outcomes, but recent literature suggests otherwise.
Methods
This retrospective cohort study examined inborn infants between 2014 and 2017. Biochemical and clinical characteristics determined the presence of NE and an encephalopathy score categorized infants as Definite or Possible mNE. An Unexposed control group consisted of newborns not meeting the inclusion criteria. Long-term outcomes assessed included cerebral palsy, seizures, developmental disorder, and motor and speech delay. The association of mNE with seizure disorder by 3 years of age was assessed with logistic regression and developmental disorders with Cox proportional hazards models.
Results
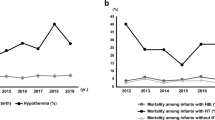
Of the 156,501 births, we identified 130 with Definite mNE and 445 with Possible mNE (0.8 and 2.8 per 1000 births, respectively). Both groups had significantly higher rates of any developmental disorder and motor and speech delay when compared to the Unexposed (p < 0.05, except for p = 0.07 for motor delay in the Possible NE group). The Definite mNE group had higher rates of developmental disorder and motor and speech delay when compared to the Unexposed with hazard ratios (95% CI) 2.0 (1.2–3.2), 3.7 (1.5–8.8), and 2.1 (1.3–3.5), respectively.
Conclusions
An estimate of short- and long-term consequences of mNE suggests that there may be a higher risk of adverse outcome.
Impact
-
Infants with mild NE are at significant risk for adverse short- and long-term outcomes. The risk of having an abnormal long-term outcome at 3 years of age were doubled in the mild NE group compared to the Unexposed group.
-
Randomized clinical trials are needed as neuroprotective strategies may mitigate these.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 14 print issues and online access
$259.00 per year
only $18.50 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
A deidentified analytic dataset with an accompanying data dictionary used in this study could be shared with qualified researchers who request the data to replicate the results shown in this article given approval by the Kaiser Foundation Research Institute Human Subjects Committee and by the Human Subjects Committee at the institutions requesting the data and a signed data sharing agreement.
Change history
28 November 2022
A Correction to this paper has been published: https://doi.org/10.1038/s41390-022-02389-x
References
Kurinczuk, J. J., White-Koning, M. & Badawi, N. Epidemiology of neonatal encephalopathy and hypoxic-ischaemic encephalopathy. Early Hum. Dev. 86, 329–338 (2010).
Lawn, J. E. et al. 1 Year after the Lancet Neonatal Survival Series–was the call for action heard? Lancet 367, 1541–1547 (2006).
Sarnat, H. B. & Sarnat, M. S. Neonatal encephalopathy following fetal distress. A clinical and electroencephalographic study. Arch. Neurol. 33, 696–705 (1976).
Jacobs, S. E. et al. Cooling for newborns with hypoxic ischaemic encephalopathy. Cochrane Database Syst. Rev. CD003311 (2013).
Robertson, C. M. & Finer, N. N. Long-term follow-up of term neonates with perinatal asphyxia. Clin. Perinatol. 20, 483–500 (1993).
Robertson, C. M., Finer, N. N. & Grace, M. G. School performance of survivors of neonatal encephalopathy associated with birth asphyxia at term. J. Pediatr. 114, 753–760 (1989).
Chalak, L. F. et al. Prospective research in infants with mild encephalopathy identified in the first six hours of life: neurodevelopmental outcomes at 18-22 months. Pediatr. Res. 84, 861–868 (2018).
Finder, M. et al. Two-year neurodevelopmental outcomes after mild hypoxic ischemic encephalopathy in the era of therapeutic hypothermia. JAMA Pediatr. 174, 48–55 (2020).
Murray, D. M., O’Connor, C. M., Ryan, C. A., Korotchikova, I. & Boylan, G. B. Early EEG grade and outcome at 5 years after mild neonatal hypoxic ischemic encephalopathy. Pediatrics 138, e20160659 (2016).
Odd, D. E., Lewis, G., Whitelaw, A. & Gunnell, D. Resuscitation at birth and cognition at 8 years of age: a cohort study. Lancet 373, 1615–1622 (2009).
van Handel, M., Swaab, H., de Vries, L. S. & Jongmans, M. J. Behavioral outcome in children with a history of neonatal encephalopathy following perinatal asphyxia. J. Pediatr. Psychol. 35, 286–295 (2010).
DuPont, T. L. et al. Short-term outcomes of newborns with perinatal acidemia who are not eligible for systemic hypothermia therapy. J. Pediatr. 162, 35–41 (2013).
Prempunpong, C. et al. Prospective research on infants with mild encephalopathy: the PRIME study. J. Perinatol. 38, 80–85 (2018).
Reiss, J., Sinha, M., Gold, J., Bykowski, J. & Lawrence, S. M. Outcomes of infants with mild hypoxic ischemic encephalopathy who did not receive therapeutic hypothermia. Biomed. Hub. 4, 1–9 (2019).
El-Dib, M. et al. Should therapeutic hypothermia be offered to babies with mild neonatal encephalopathy in the first 6 h after birth? Pediatr. Res. 85, 442–448 (2019).
Muniraman, H. et al. Biomarkers of hepatic injury and function in neonatal hypoxic ischemic encephalopathy and with therapeutic hypothermia. Eur. J. Pediatr. 176, 1295–1303 (2017).
Jetton, J. G. & Askenazi, D. J. Update on acute kidney injury in the neonate. Curr. Opin. Pediatr. 24, 191–196 (2012).
Robins, D. L., Fein, D., Barton, M. L. & Green, J. A. The modified checklist for autism in toddlers: an initial study investigating the early detection of autism and pervasive developmental disorders. J. Autism Dev. Disord. 31, 131–144 (2001).
Radecki, L., Sand-Loud, N., O’Connor, K. G., Sharp, S. & Olson, L. M. Trends in the use of standardized tools for developmental screening in early childhood: 2002-2009. Pediatrics 128, 14–19 (2011).
Lee, A. C. et al. Intrapartum-related neonatal encephalopathy incidence and impairment at regional and global levels for 2010 with trends from 1990. Pediatr. Res. 74(Suppl. 1), 50–72 (2013).
Azzopardi, D. V. et al. Moderate hypothermia to treat perinatal asphyxial encephalopathy. N. Engl. J. Med. 361, 1349–1358 (2009).
Eicher, D. J. et al. Moderate hypothermia in neonatal encephalopathy: safety outcomes. Pediatr. Neurol. 32, 18–24 (2005).
Gluckman, P. D. et al. Selective head cooling with mild systemic hypothermia after neonatal encephalopathy: multicentre randomised trial. Lancet 365, 663–670 (2005).
Shankaran, S. et al. Whole-body hypothermia for neonates with hypoxic-ischemic encephalopathy. N. Engl. J. Med. 353, 1574–1584 (2005).
Pin, T. W., Eldridge, B. & Galea, M. P. A review of developmental outcomes of term infants with post-asphyxia neonatal encephalopathy. Eur. J. Paediatr. Neurol. 13, 224–234 (2009).
Committee on Fetus and Newborn et al. Hypothermia and neonatal encephalopathy. Pediatrics 133, 1146–1150 (2014).
Chawla, S., Bates, S. V. & Shankaran, S. Is it time for a randomized controlled trial of hypothermia for mild hypoxic-ischemic encephalopathy? J. Pediatr. 220, 241–244 (2020).
Saw, C. L., Rakshasbhuvankar, A., Rao, S., Bulsara, M. & Patole, S. Current practice of therapeutic hypothermia for mild hypoxic ischemic encephalopathy. J. Child Neurol. 34, 402–409 (2019).
Acknowledgements
We would like to acknowledge the important contributions of Abhinav Kareddy, University of California, Los Angeles (Redcap Database set-up, statistical analysis, and review of manuscript). We would like to acknowledge the important contributions of William E Benitz MD Stanford University, Palo Alto.
Funding
This study was supported by Kaiser Permanente Northern California (KPNC) Central Research Committee Community Benefit Grant.
Author information
Authors and Affiliations
Contributions
V.-P.A. designed the study, designed the data collection tool, and prepared and revised the manuscript. A.S. collected data, carried out the initial analyses, and reviewed the manuscript. E.W. study design and coordinated data collection. S.X. data analysis and collection tool. K.V.M. designed the study, reviewed, and revised the manuscript. M.C. data collection, analysis, reviewed and revised the manuscript. M.K. designed the study, data analysis, and reviewed and revised the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
Consent was not required for this.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised: The author’s name Michael W. Kuzniewicz was incorrectly given as ‘Michael Kuzniwiecz’.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Akula, VP., Sriram, A., Xu, S. et al. Adverse short- and long-term outcomes among infants with mild neonatal encephalopathy. Pediatr Res 94, 1003–1010 (2023). https://doi.org/10.1038/s41390-022-02249-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41390-022-02249-8