Abstract
Background
Children born from mothers who underwent bariatric surgery were found to have an improved lipid profile and lower CRP levels compared to siblings born before surgery. We hypothesized that surgery before pregnancy might also influence endothelial function in the offspring.
Methods
Blood sample analysis, blood pressure (BP) measurement, and peripheral arterial tonometry (PAT) were performed in 142 children (median age 10.5 years), either born from mothers who underwent bariatric surgery (BS) before pregnancy (n = 36) from mothers with overweight/obesity (OW/OB) (n = 71) or from normal weight (NW) mothers (n = 35), allowing the determination of the Reactive Hyperemia Index (RHI) in 111 children.
Results
Children of the BS group had a higher diastolic blood pressure SDS and a lower RHI compared to the children of the OW/OB and NW group (1.32 versus 1.37 in OW/OB and 1.70 in NW; p = 0.004). After log transformation and correction for age, weight SDS, BMI SDS, body fat percentage, and diastolic BP SDS, RHI was comparable between the groups.
Conclusions
Children of mothers who underwent bariatric surgery before pregnancy do not have a disturbed endothelial function before puberty, when their increased diastolic BP and degree of adiposity is taken into account.
Impact
-
Children born after maternal bariatric surgery have a higher diastolic blood pressure without impaired endothelial function.
-
To our knowledge, this is the first study that investigates the vascular function of children based on maternal characteristics during pregnancy.
-
Adult offspring of mothers with obesity during pregnancy have an increased cardiovascular mortality. Since we cannot demonstrate a childhood-onset primary vascular dysfunction, this cardiovascular vulnerability might be more related to the hypertension and body adiposity. Thus, more emphasis should be made on the prevention of obesity and hypertension in the offspring at risk for development of obesity.
Similar content being viewed by others
Introduction
Maternal obesity is associated with dysregulated metabolic, inflammatory, and vascular pathways in the mother.1 Maternal obesity is also considered a risk factor for premature vascular dysfunction in the adult offspring, putting them at risk for coronary events.1,2,3 Higher pre-pregnancy maternal weight is associated with an adverse metabolic profile in the offspring, including higher blood pressure and adverse lipid profile both in newborns and in young adulthood.4 On the other hand, children of mothers who underwent bariatric surgery before pregnancy as treatment for morbid obesity were found to have an improved lipid profile and lower degree of inflammation compared to their siblings born before the surgery.5
Peripheral arterial tonometry (PAT) is an easy non-invasive and reliable method for the evaluation of the endothelial function in children and adolescents.6 The PAT assesses the changes in digital pulse amplitude during a hyperemic reaction after an arterial occlusion of the upper-arm, represented as the Reactive Hyperemia Index (RHI), with a lower index showing a more impaired endothelial function.7,8,9 In comparison with flow-mediated dilatation (FMD), the PAT has the advantage to be not operator dependent, but the experience with this technique in children is still limited. In children and adults, RHI has been found to be inversely related with saturated fatty acids of plasma phospholipids, total cholesterol, and diastolic blood pressure percentiles, at least in healthy male adolescents.10,11
We hypothesized that children born after bariatric surgery might have a less impaired endothelial function than children born from mothers suffering from overweight or obesity during pregnancy. We therefore analyzed the RHI by PAT in children born from operated mothers to the offspring of mothers with overweight/obesity during pregnancy and mothers with a normal weight and studied potential determinants as adiposity, lipid profile, and blood pressure.
Materials and methods
Study design
The EFFECTOR study is a cross-sectional follow-up cohort study, investigating the vascular function of the offspring of mothers who underwent bariatric surgery before pregnancy (see Table 2 for maternal characteristics).12,13,14 The Ethics Committees of the University Hospitals of Brussels and Leuven (Belgium) approved the study, which was registered at ClinicalTrials.gov (NCT02992106). A written informed consent was obtained from the parents and each child received age-adjusted information through an assent. Investigations were performed during a single home visit by the same trained pediatric fellow (K.V.D.M.). Home visits were performed in the morning, after an overnight of fasting.
Study procedures and outcome parameters
Blood pressure and PAT (EndoPAT) measurements
First, a blood pressure measurement was performed after a rest of 5 min, using a commercial oscillometer (Mindray VS-900). These blood pressure values were expressed as SD score (SDS) for sex and age according to international reference data.15 Subsequently, a non-invasive PAT measurement was performed, using a commercial device (EndoPAT 2000®, Itamar Medical Ltd, Israel).12,16 For this measurement, both index fingers of the patient were placed in pneumatic probes. After a baseline measurement for 5 min, an arterial occlusion of the right arm was performed with a blood pressure cuff. The target duration of this occlusion was 5 min and was only aborted prematurely at the request of the child. After the arterial occlusion, a rapid deflation of the cuff caused a reactive hyperemia. The concomitant software package was used to calculate the RHI as main vascular outcome. The RHI is calculated by an automated algorithm and is based on the time span from 90 to 150 s of the post-occlusion period. The within-subject variance of the RHI is 0.16 in children.6
Lipid profile and high-sensitive C-reactive protein analysis
After the EndoPAT measurement, a blood sample was obtained for the measurement of high-sensitive C-reactive protein (hsCRP), uric acid, and lipid profile (total cholesterol, low-density lipoprotein, high-density lipoprotein, and triglycerides). All laboratory analyses were performed by using the Vitros 4600 laboratory analyzer by Ortho Clinical Diagnostics. Error percentages are 30.6% for hsCRP, 3.0% for uric acid, and 5.3% for total cholesterol.
Body size and body composition assessment
Height was measured with a stadiometer (seca 2017) in standing position and weight and body fat percentage were assessed by using a bioelectrical impedance analysis (BIA) (Tanita MC-780U). The within-subject variance of the fat percentage assessment by BIA is 0.92% in children.17 The BMI was calculated (in kg/m2) and all anthropometric values were expressed as SDS for sex and age according to national reference data.18 BMI SDS between 1.3 and 2.3 was considered as overweight and above 2.3 as obesity. Excess in fat percentage by BIA was calculated to the 50th percentile for age- and sex-specific reference values.19
Questionnaire data analysis
Information on lifestyle (caffeine usage and physical activities) was obtained through a parental questionnaire, which was especially designed for this study.
Statistical analysis
All statistical analysis were performed using SPSS IBM version 25. Descriptive statistics were used to describe the population characteristics. Pearson χ2 tests were used for the comparison of categorical variables when the assumptions were met. Whenever the assumptions for the Pearson χ2 were not met (cell counts <5), we used Fisher exact tests. Since the reported RHI scores had a non-normal distribution, non-parametric statistical analyses were used throughout the analysis including the RHI as main studied outcome (Mann Whitney U analysis/Wilcoxon rank test). Correlations between continuous variables were calculated with the use of the Spearman rank correlation coefficient. A stepwise multivariate regression analysis with log RHI as a dependent factor and maternal cohort, diastolic blood pressure, and HDL cholesterol as independent factors was performed. Because of limited sample size and a combination of multiple risk factors, exploratory univariate analyses were performed uncorrected for each correlated risk factor. P values below 0.05 were considered statistically significant. Based on the observed values of the RHI in the BS and NW groups and the standard deviation of 0.4, the observed power for a significance level of 0.05 and (a minimum of) 26 subjects per group is 87.8%.
Results
Population description
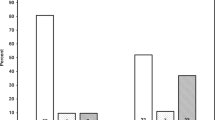
A total of 294 eligible mothers could be contacted by mail with an information leaflet and letter. In total, 143 agreed to participate in a follow-up study on their child outcome. One hundred and seven eligible subjects were lost-to-follow-up (107/294; 36.4%) and only 15% (44/294) refused to participate in the follow-up study (Fig. 1). Of the 143 included children, all but one (who had fever at the moment of the study visit) underwent the EndoPAT measurement. Out of these 142 EndoPAT measurements, 111 (78.2%) were of sufficient quality to calculate a RHI. Children without RHI did not differ in age, BMI z-score, or blood pressure readings from those with RHI results (data not shown).
The 111 included children had a median age of 10.5 years (range 4.7–13.2 years) and consisted of 56 males (50.5%) and 55 females (49.5%). The median (range) weight SDS, height SDS, and BMI SDS were respectively 0.24 SDS (−2.00 to 2.98), 0.51 SDS (−1.30 to 3.32), and −0.02 SDS (−2.56 to 2.49) (Table 1). In total, 88 children (79.3%) had a normal adiposity status (BMI SD < 1.3); 19 children (17.1%) were suffering from overweight (BMI SD 1.3–2.3); and 4 children (3.6%) were suffering from obesity (BMI SD > 2.3). Median excess in fat percentage was 1.6% (range −8.4 to 20.7%).
The children born after maternal bariatric surgery (BS group) were the youngest, but had the highest body weight SDS, BMI SDS, and fat percentage (Table 1). The BS mothers had the lowest education level and the highest prevalence of smoking. They had a comparable pre-pregnancy BMI to the women of the overweight/obesity group and lower prevalence of hypertension during pregnancy (Table 2).
Lipid profile and low-grade inflammation
No differences were found in lipid profile and low-grade inflammation between the groups (Table 3). Six children (5.4%) had an LDL cholesterol above 135 mg/dL; three children (2.7%) had an HDL cholesterol below 35 mg/dL, and 4 children (3.6%) had a hsCRP above 5 mg/L.
Vascular function
The median (range) systolic blood pressure SDS was 0.34 (−1.46 to 2.84) and diastolic blood pressure SDS was 0.07 (−1.59 to 1.58). The children in the BS group showed a significantly higher diastolic blood pressure SDS (0.23 versus 0.11 in overweight/obesity and versus −0.34 in NW; p = 0.002). None of the children had a diastolic blood pressure above 2 SDS.
The median value of the RHI was 1.44; ranging from 0.77 to 2.82. The children in the BS group showed the lowest RHI (1.32 versus 1.37 in overweight/obesity and versus 1.70 in NW; p = 0.004). (Fig. 2). When the log RHI was corrected for all the observed clinical differences between the groups (age, weight SDS, BMI SDS, body fat percentage, and diastolic blood pressure SDS), there no significant difference between the studied groups (p = 0.24).
Correlations
RHI did not correlate significantly with age, weight SDS, height SDS, BMI SDS, waist SDS, and excess in fat percentage. Males had a similar RHI than girls in the different groups (data not shown). No significant differences in median RHI was observed between overweight/obese children and normal weight children (1.30 versus 1.47; p = 0.30). The RHI tended to be higher in pubertal children (1.49 versus 1.39; p = 0.39).
RHI correlated significantly with diastolic blood pressure SDS (Rho = −0.33; p < 0.001), but not with the systolic blood pressure SDS. There was also a significant correlation with serum HDL-cholesterol level (Rho = 0.25; p = 0.016), but no significant correlations with the total cholesterol level, LDL-cholesterol level (Rho = 0.18, p = 0.08 and Rho = 0.04, p = 0.70, respectively) or hSCRP and uric acid (Rho = −0.02, p = 0.87 and Rho = −0.03, p = 0.80, respectively) were found. Caffeine use and hours of sports activity were not related to RHI.
RHI was unrelated to maternal smoking, arterial hypertension during pregnancy, pre-pregancy BMI, gestational weight gain.
In explanatory univariate assessments, diastolic blood pressure SDS, and HDL cholesterol were significant contributors to the Log RHI (respectively, r2 = 0.105; p = 0.001 and r2 = 0.087; p = 0.004).
In a multivariate stepwise regression analysis including maternal cohort, diastolic blood pressure SDS, and HDL cholesterol level as independent variables and Log RHI as outcome, all parameters were withheld with a total prediction of 23.9% in data variance.
Discussion
This is the first study investigating endothelial function in children born after maternal bariatric surgery. RHI of children born from mothers after bariatric surgery was found to be lower compared to children of mothers with overweight/obesity or a normal weight during pregnancy. However, after correction for adiposity and diastolic blood pressure, which were higher in the BS group, no significant difference in RHI was present.
Sex and age were found not to influence the RHI in our mainly pre-pubertal pediatric population, in accordance with previously reports in normal children.10,20,21,22,23,24,25 Despite observing a slightly higher RHI with progressing Tanner stages, this increase was not significant, probably due to small numbers, since progressing puberty is known to result in increased RHI values.11,26 We did observe a slightly lower RHI in the children suffering from overweight or obesity; however, the difference was not significant. Previous studies on the effect of adiposity on the RHI have been conflicting. Some authors have described an inverse relationship between the RHI and increasing adiposity,22,26,27,28,29,30 while others did not find this relationship.10,20,21,23,25,31,32 Since most of these lower RHI values in children suffering from overweight and obesity are reported by studying this specific population, one might speculate that these children already have a more pronounced underlying pathogenic damage to the vasculature. Unfortunately, we have no data on the onset of overweight or obesity in our population.
In accordance with a previous study in healthy children, a negative effect of diastolic blood pressure on RHI was found in our study.21 On the other hand, a positive correlation between the RHI and HDL level was seen. Since HDL cholesterol is considered as a protector for diverse atherogenic influences, this correlation might reflect the upregulation of the endothelial nitric oxide synthase (eNOS) as being one of the important functions of the HDL.33 Consistent with studies in healthy children, no convincing correlation with the amount of physical activity was found.34 A beneficial influence of physical activity has mainly been reported in trials in children or adolescents suffering from overweight or obesity.1
This study enlarges the experience with PAT in school-aged children who underwent testing by the EndoPAT 2000® device in the context of a non-therapeutic study. For the first time, data on endothelial function in children born after bariatric surgery are presented. Major limitations of our study are the small number of children studied in a cross-sectional study design and the lack of biomarkers of endothelial function. However, metabolic risk factors, such as HDL cholesterol and hSCRP, were studied.
In conclusion, our results show that offspring of mothers suffering from overweight or obesity during their pregnancy, whether or not they underwent bariatric surgery, do not have an impaired vascular function at childhood age compared to the offspring of normal weight mothers, when their increased diastolic blood pressure and degree of adiposity is taken into account. The increased cardiovascular mortality observed in adult offspring of obese mothers might thus be more related to the accompanying obesity and hypertension than a childhood-onset primary vascular dysfunction.
References
Van De Maele, K., Devlieger, R. & Gies, I. In utero programming and early detection of cardiovascular disease in the offspring of mothers with obesity. Atherosclerosis 275, 182–195 (2018).
Reynolds, R. M. et al. Maternal obesity during pregnancy and premature mortality from cardiovascular event in adult offspring: follow-up of 1 323 275 person years. BMJ 347, f4539 (2013).
Eriksson, J. G., Sandboge, S., Salonen, M. K., Kajantie, E. & Osmond, C. Long-term consequences of maternal overweight in pregnancy on offspring later health: findings from the Helsinki Birth Cohort Study. Ann. Med. 46, 434–438 (2014).
Devlieger, R. et al. Maternal obesity in Europe: where do we stand and how to move forward?: a scientific paper commissioned by the European Board and College of Obstetrics and Gynaecology (EBCOG). Eur. J. Obstet. Gynecol. Reprod. Biol. 201, 203–208 (2016).
Smith, J. et al. Effects of maternal surgical weight loss in mothers on intergenerational transmission of obesity. J. Clin. Endocrinol. Metab. 94, 4275–4283 (2009).
Selamet Tierney ES et al. Endothelial pulse amplitude testing: feasibility and reproducibility in adolescents. J. Pediatr. 154, 901–905 (2009).
Bruyndonckx, L. et al. Assessment of endothelial dysfunction in childhood obesity and clinical use. Oxid. Med. Cell Longev. 2013, 174782 (2013).
Donald, A. E. et al. Non-invasive assessment of endothelial function: which technique? J. Am. Coll. Cardiol. 48, 1846–1850 (2006).
Kuvin, J. T. et al. Assessment of peripheral vascular endothelial function with finger arterial pulse wave amplitude. Am. Heart J. 146, 168–174 (2003).
Chen, Y. et al. High levels of soluble intercellular adhesion molecule-1, insulin resistance and saturated fatty acids are associated with endothelial dysfunction in healthy adolescents. Atherosclerosis 211, 638–642 (2010).
Bhangoo, A., Sinha, S., Rosenbaum, M., Shelov, S. & Ten, S. Endothelial function as measured by peripheral arterial tonometry increases during pubertal advancement. Horm. Res. Paediatr. 76, 226–233 (2011).
Van De Maele, K., Gies, I. & Devlieger, R. Effect of bariatric surgery before pregnancy on the vascular function in the offspring: protocol of a cross-sectional follow-up study. BMJ Paediatr. Open 3, e000405 (2019).
Van De Maele, K. et al. Adiposity, psychomotor and behaviour outcomes of children born after maternal bariatric surgery. Pediatr Obes. 16, e12749 (2021).
Van De Maele, K., De Geyter, C., Vandenplas, Y., Gies I. & Devlieger, R. Eating habits of children born after maternal bariatric surgery. Nutrients 12, 2577. https://doi.org/10.3390/nu12092577 (2020).
National High Blood Pressure Education Program Working Group on High Blood Pressure in Children and Adolescents. The fourth report on the diagnosis, evaluation, and treatment of high blood pressure in children and adolescents. Pediatrics 114, 555 (2004).
Bruyndonckx, L. et al. Methodological considerations and practical recommendations for the application of peripheral arterial tonometry in children and adolescents. Int J. Cardiol. 168, 3183–3190 (2013).
Kettaneh, A. et al. Reliability of bioimpedance analysis compared with other adiposity measurements in children: the FLVS II Study. Diabetes Metab. 31, 534–541 (2005).
Roelants, M. & Hauspie, R. Flemish growth charts https://www.vub.be/groeicurven/groeicurven.html (2004).
McCarthy, H. D., Cole, T. J., Fry, T., Jebb, S. A. & Prentice, A. M. Body fat reference curves for children. Int J. Obes. (Lond.) 30, 598–602 (2006).
Osika, W. et al. Anger, depression and anxiety associated with endothelial function in childhood and adolescence. Arch. Dis. Child 96, 38–43 (2011).
Odanaka, Y. et al. Microvascular endothelial function in Japanese early adolescents. J. Clin. Biochem. Nutr. 61, 228–232 (2017).
Mahmud, F. H., Hill, D. J., Cuerden, M. S. & Clarson, C. L. Impaired vascular function in obese adolescents with insulin resistance. J. Pediatr. 155, 678–682 (2009).
Mahmud, F. H. et al. Altered endothelial function in asymptomatic male adolescents with type 1 diabetes. Congenit. Heart Dis. 1, 98–103 (2006).
Yano, S. et al. Glycosaminoglycan metabolism defects and atherosclerosis: frequent association of endothelial dysfunction in patients with Mucopolysaccharidosis. J. Inherit. Metab. Dis. 37, 255–261 (2014).
Scaramuzza, A. E. et al. Adolescents and young adults with type 1 diabetes display a high prevalence of endothelial dysfunction. Acta Paediatr. 104, 192–197 (2015).
Radtke, T. et al. Puberty and microvascular function in healthy children and adolescents. J. Pediatr. 161, 887–891 (2012).
Agarwal, C. et al. Obesity, hyperglycemia and endothelial function in inner city Bronx adolescents: a cross-sectional study. Int J. Pediatr. Endocrinol. 2013, 18 (2013).
Pareyn, A., Allegaert, K., Verhamme, P., Vinckx, J. & Casteels, K. Impaired endothelial function in adolescents with overweight or obesity measured by peripheral artery tonometry. Pediatr. Diabetes 16, 98–103 (2015).
Bartz, S. K., Caldas, M. C., Tomsa, A., Krishnamurthy, R. & Bacha, F. Urine albumin-to-creatinine ratio: a marker of early endothelial dysfunction in youth. J. Clin. Endocrinol. Metab. 100, 3393–3399 (2015).
Tomsa, A., Klinepeter Bartz, S., Krishnamurthy, R. & Bacha, F. Endothelial function in youth: a biomarker modulated by adiposity-related insulin resistance. J. Pediatr. 178, 171–177 (2016).
Tryggestad, J. B., Thompson, D. M., Copeland, K. C. & Short, K. R. Obese children have higher arterial elasticity without a difference in endothelial function: the role of body composition. Obesity 20, 165–171 (2012).
Czippelova, B. et al. Arterial stiffness and endothelial function in young obese patients—vascular resistance matters. J. Atheroscler. Thromb. 26, 1015–1025 (2019).
Allard-Ratick, M. P. et al. HDL: Fact, fiction, or function? HDL cholesterol and cardiovascular risk. Eur. J. Prev. Cardiol. https://doi.org/10.1177/2047487319848214 (2019).
Radtke, T., Kriemler, S., Eser, P., Saner, H. & Wilhelm, M. Physical activity intensity and surrogate markers for cardiovascular health in adolescents. Eur. J. Appl Physiol. 113, 1213–1222 (2013).
Acknowledgements
The research activities of K.VDM. are funded by a doctoral research grant from the Belgian Society for Paediatric Endocrinology and Diabetology (BESPEED) and additional research funding from Wetenschappelijk Fonds Willy Gepts of the UZ Brussel. R.D. is a recipient of a fundamental clinical researcher’s grant, 1803311N, of the “Fonds Wetenschappelijk Onderzoek” (Flemish Research Foundation), Flanders, Belgium.
Author information
Authors and Affiliations
Contributions
K.V.D.M., R.D., and I.G. designed the study and conceptualized the study protocol. Data acquisition was performed by K.V.D.M. K.V.D.M. drafted the initial blueprint, which was reviewed extensively for content and methods by all other contributing authors. All authors approved the final version of the manuscript as submitted for publication.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Van De Maele, K., Devlieger, R., De Schepper, J. et al. Endothelial function and its determinants in children born after maternal bariatric surgery. Pediatr Res 91, 699–704 (2022). https://doi.org/10.1038/s41390-021-01500-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41390-021-01500-y
This article is cited by
-
The impact of maternal bariatric surgery on long-term health of offspring: a scoping review
Pediatric Research (2023)