Abstract
Spontaneous preterm birth is a serious and common pregnancy complication associated with hormonal dysregulation, infection, inflammation, immunity, rupture of fetal membranes, stress, bleeding, and uterine distention. Heredity is 25–40% and mostly involves the maternal genome, with contribution of the fetal genome. Significant discoveries of candidate genes by genome-wide studies and confirmation in independent replicate populations serve as signposts for further research. The main task is to define the candidate genes, their roles, localization, regulation, and the associated pathways that influence the onset of human labor. Genomic research has identified some candidate genes that involve growth, differentiation, endocrine function, immunity, and other defense functions. For example, selenocysteine-specific elongation factor (EEFSEC) influences synthesis of selenoproteins. WNT4 regulates decidualization, while a heat-shock protein family A (HSP70) member 1 like, HSPAIL, influences expression of glucocorticoid receptor and WNT4. Programming of pregnancy duration starts before pregnancy and during placentation. Future goals are to understand the interactive regulation of the pathways in order to define the clocks that influence the risk of prematurity and the duration of pregnancy. Premature birth has a great impact on the duration and the quality of life. Intensification of focused research on causes, prediction and prevention of prematurity is justified.
Similar content being viewed by others
Approximately 15 million preterm live births (PTB) contribute to around 1 million infant deaths annually. According to estimates, each year the consequences of PTBs cause a loss of 80–90 million quality-adjusted life-years (QALY).1,2 QALY ranges between 1 (good health) and 0 (death). Despite the increased survival of preterm infants, the lifelong adverse consequences of very preterm birth remain high. Preterm birth rates are 5.5–10% in Europe, 10–11% in the USA, and 11–13% globally. Spontaneous preterm labor and delivery (SPTD) accounts for ~70% of all cases of PTB. About 50% of spontaneous preterm live births (SPTB) occur in apparently low-risk pregnancies. A number of acquired factors may increase the risk of SPTB, and African-American ethnicity increases the adjusted risk of SPTB. The most significant risk factor in singleton births is previous preterm delivery, which increases the risk of preterm birth 3–6-fold in subsequent pregnancies.3,4
The synthetic nonsteroidal estrogen diethylstilbestrol (DES) was administered to prevent miscarriages and premature births during 1943–1972 but had no beneficial effects. However, it increased the risk of squamous genital neoplasia in female offspring as well as infertility and hypospadias in male offspring.5 Other serious drug side effects raised concerns about the use of medication in early pregnancy.6 Tocolytic therapies administered later in pregnancy also failed to prevent SPTD/SPTB.7,8
Reproduction is exceptionally species-specific. According to epidemiological family studies, the risk of prematurity depends mostly on genetic predisposition of the mother. Genome of the fetus has also influence on the risk of premature birth.9,10 Both maternal and fetal genomes harbor variants that increase the risk of SPTD and SPTB. In live births, these two phenotypes converge. In present review we link the susceptibility of SPTD to maternal genetic factors and susceptibility of SPTB phenotype to fetal genetic factors. However, maternal and fetal genes likely interactively influence the risk of SPTD/SPTB. As the genetic predisposition in SPTD/SPTB ranges from 25 to 40%,9,10 the Human Genome Project provides tools that are useful in identifying genes that influence the risk of SPTD/SPTB. This brief review summarizes the new genomic findings about susceptibility to SPTD/SPTB. We envision that these and further genomic and epigenetic discoveries will improve prediction of risk in early pregnancy and eventually provide new tools for decreasing the risk of SPTD/SPTB.
Factors associated with onset of preterm labor and delivery
Prostaglandin E2 (PGE2), PGF2α, and oxytocin mediate the induction of cervical maturity and uterine contractions leading to delivery. Inflammation is involved in preterm, term, and post-term labor. The obstetric data that define the factors that trigger SPTD are presented below in brief.11
Uterine distention
The pressure induced by intrauterine mass in relation to uterus size leads to stretching and contraction of smooth muscle. This increases expression of contraction-associated proteins (e.g., prostaglandins, gap junction proteins, and oxytocin) and is associated with a 6–10-fold increase in the prematurity rate of twin pregnancies. The distending pressure and fetal movements generate shear forces and inflammation in fetal membranes, which may be transmitted to the uterus and cervix with consequent cervical maturation and labor.12,13
Preterm prelabor rupture of fetal membranes (PPROM)
PPROM is an initiating event in 20–30% of all SPTB cases. It is associated with increased risk of ascending infection. Not all PPROM cases lead to SPTD; many cases remain silent until term. PPROM is more common and often recurrent in African Americans.14
Infection and activated inflammation
Infection and activated inflammation are associated with 25–40% of SPTD cases. Ascending infection is the most common. Intrauterine (endometriosis), salpinx, and extrauterine infections from various sources are also risk factors for SPTB. Aside from clinical chorioamnionitis (CA), CA is mostly silent (i.e., histological CA). Cell wall components of microbes (lipopolysaccharides and other toxins) bind to specific receptors, inducing synthesis of inflammatory cytokines (e.g., IL-1 and TNF) and PGE2.3,15
Loss of immune tolerance
In early pregnancy, endometrial tissue transforms into the decidua. The decidua contains maternal Th2 and regulatory T (Treg) cells, CD56-positive lymphocytes, natural killer cells, antigen presenting cells (APC), fibroblasts, stromal cells, and fetal decidual trophoblasts. Syncytiotrophoblasts and a portion of the cytotrophoblasts are in direct contact with maternal blood cells. To suppress maternal immune activation, trophoblasts express tolerogenic HLA-G molecule. Aberrant activation of the silenced host immune system may induce SPTD.16,17
Stress
Reaction to stress may perturb production of the hormones necessary to maintain pregnancy. For example, stress is known to increase placental production of corticotropin-releasing hormone (CRH).17
Intrauterine bleeding
Intrauterine bleeding that occurs in the absence of evidence of placenta previa increases the risk of SPTB. The decidua is a common source of bleeding.18,19
Placental and fetal growth
Placenta and fetal membranes likely play roles in regulating the duration of pregnancy. Critical early events include implantation and vascularization. Fetuses that undergo SPTB tend to have slow intrauterine growth. However, the causes and consequences of growth delay remain poorly understood with respect to SPTD. Elective preterm birth is indicated for serious intrauterine growth restriction.20
Fetal maturity
Immune proteins, including surfactant proteins (SP) A and D, have been suggested to promote labor.21 In human pregnancy, the appearance of SP-A in amniotic fluid and the onset of labor may not coincide.22 Antenatal glucocorticoid has no effect on human pregnancy duration, either. Although inflammatory activation induces surfactant maturity and preterm labor, these two events may not be of consequence.23
Endocrine system
Human progesterone (P4) synthesis takes place in trophoblasts after early involution of the corpus luteum.24 P4 is an anti-inflammatory hormone that promotes immune tolerance, and maintains myometrium quiescence. P4 levels increase throughout pregnancy and even during labor. However, toward the end of human pregnancy, the responsiveness of progesterone receptor (PR) decreases as levels of PR-A (the truncated form of PR) increase and levels of PR-B (the active, full-length form of PR) decrease.25,26 Near the time of term delivery, synthesis of CRH increases in the placenta, boosting the synthesis of corticotrophin. This increases synthesis of cortisol and dehydroepiandrosterone (DHEA), a precursor of estradiol that antagonizes P4.27,28,29
Brief comment on current pharmacological approaches to prevent or delay SPTD/SPTB
Current practice recognizes primary, secondary and tertiary interventions in prevention of SPTD.30 Primary interventions target to whole population before or during pregnancy. Secondary prevention targets women with established risk of preterm birth. Tertiary conditions include early detection of imminent preterm labor and treatments that delay or prevent early birth, or accelerate fetal maturity. The focus has been on tertiary prevention.
Antibiotics and probiotics
Antibiotics may prolong the duration of pregnancy after PPROM.31 In general, however, antibiotics do not decrease the risk of SPTD/SPTB. Further investigations of the types of microbes present in maternal and intrauterine tissues, as well as prophylactic trials with probiotics, are required to substantiate the potential effects of microbial colonization on the rate of SPTD.32
Tocolytic agents
Tocolytics silence uterine contractions and may delay SPTB for several days, allowing time for antenatal steroids to take effect.33 Nonsteroidal anti-inflammatory agents are effective but may cause serious adverse effects. Oxytocin antagonist and calcium channel blockers are preferred for their safety. According to a meta-analysis, calcium channel blockers delay preterm labor.33
Progesterone
According to meta-analysis, in rare pregnancies with short cervix syndrome (cervix <0.25 cm before mid-gestation), administration of vaginal P4 increases the length of pregnancy and decreases prematurity rate and morbidity of the newborn.34 Recent meta-analysis confirmed the beneficial effect of P4 in short-cervix syndrome,35 despite in a large recent trial P4 had no beneficial effect on preterm births or postnatal outcomes.36,37 Uncertainty remains concerning the indications, dosage, route, onset, and duration of administration of P4.
Genetic predisposition to SPTD/SPTB
Studies of the duration of pregnancy in twin sisters and in families with recurrent SPTB/SPTD demonstrate that both maternal and fetal genetic factors contribute to the variance in gestation length.38,39 Hereditary factors contribute an estimated 25–40% toward the risk of premature birth. In one study, the maternal and fetal genetic contributions were ~60% and 40%, respectively, toward the variance in pregnancy duration.9 According to Wilcox10 pregnancy duration is transmitted as a matrilineal trait. However, there is no consistent evidence to indicate whether either mitochondrial genome40,41,42 or parental imprinting43 influence the SPTD/SPTB rate or duration of pregnancy.
Ethnicity influences the predisposition to preterm birth. Ethnic differences in the risk of SPTB may be due to genetic or environmental factors or a combination of both.44 In African American populations, the prematurity rate is high and the influence of environmental factors is prominent.45 Acquired risk factors include very young, and also old age of the mother; nutrition; extremes in body mass index (BMI); heavy exercise or severe stress; and exposure to infection, drugs, smoking, and toxins.38
Expressivity is likely low for common alleles associated with complex traits like SPTD that occur at an early age. Rare variants that are dominant and have a high expressivity influencing the pregnancy duration are likely damaging, disease-causing variants identifiable in families with repeated SPTD/SPTBs. Pathways that influence the duration of pregnancy may contain a large number of variants and multiple SPTD-predisposing candidate genes.
Large population sizes based on power analysis, an accurate definition of phenotypes, and minimization of ethnic and acquired differences between SPTD/SPTB and control populations will influence the success of genomic analysis. Accuracy of diagnostics is important with exclusions of multiple pregnancies, elective preterm deliveries due to maternal/fetal complications, and cases of severe clinical CA.
Populations that underwent a recent bottleneck followed by population growth without significant population admixture are attractive for genetic studies. They may have unique single-gene diseases and lower allelic variation, which decrease the population size requirements for genetic discoveries. Northeastern Finland, which has a typical population history and a unique pattern of single gene diseases, is one such example.46
Candidate gene studies
Conventional candidate gene studies have focused on genes presumed to influence SPTD/SPTB-triggering events. These genes influence the inflammatory response or resistance against infection. Another set of candidate genes influence uterine muscle contractions, tensile strength of fetal membranes, or quantity of cervical connective tissue. Still others influence placental function, differentiation, and stress; response to environmental toxins; and activity of hormones necessary to maintain pregnancy. Currently, variants representing several hundred genes have been evaluated and may show significant trends.47,48,49,50 However, these findings have either not been replicated or they have had a nominal association in genomic studies. These types of approaches, however, are still justified especially when addressing the significance of suggestive associations from the genome-wide studies in more detail.
Genome-wide approaches
The whole genome consists of ~3.1 billion base pairs, while the whole exome comprises ~180,000 exons, 35 million base pairs and 22,000 genes (about 1.2% of the whole-genome sequence). Thus far, the most common approach taken by genomic studies to investigate complex diseases has been genome-wide association study (GWAS) analyses. Typically, chips contain 0.2–2 million single nucleotide variants (SNV). Less than 0.2% of all SNVs are located in exons, and these SNVs are enriched in many chips. The expanding approaches to genomic studies include whole exome sequence (WES) and whole-genome sequence (WGS) analyses. In multifactorial phenotypes, like SPTB, disease associating genetic alterations are often outside the coding areas and thus WGS approaches have increasing importance. Table 1 shows some practical aspects of genomic studies on SPTD/SPTB.
The linkage disequilibrium of individual variants within haploblocks enables fairly accurate estimation of a very large number of SNVs. This imputation method yields an accurate estimate of 2–15 million SNVs. Variants that remain unaccounted for are mostly rare SNVs (frequency < 0.01), particularly those located in intergenic regions. Copy number variations (CMV), insertion or deletion (InDel) variants and mitochondrial DNA (mtDNA) are often included in genome analysis. Other studies have a focus on specific metabolic pathways or pharmacogenomics. Thus far, genomic studies on SPTD/SPTB have been limited.
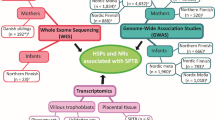
Genomic discovery requires replication. Further molecular and translational studies are essential to understand the specific function of a gene, its variants, and the functional pathways that contribute to its expression. Bioinformatics tools have become increasingly accurate as the data on molecular cellular processes in various species, tissues, cells, and diseases continue to accumulate. These data are helpful in planning and interpretation of experiments that address the molecular aspects of specific genes and their influences on individual targets51 (Fig. 1).
RNA molecules include messenger RNA (mRNA), ribosomal RNA (rRNA), transfer RNA (tRNA), signal recognition particle RNA (SRP RNA), and others. Several epigenetic systems participate in gene expression. In addition to DNA methylation (known as the methylome; i.e., the pattern of genome-wide DNA methylation), a number of proteins (particularly histones) and regulatory RNA species, e.g., microRNA (miRNA), long noncoding RNA (lncRNA), and antisense RNA (asRNA) are involved in regulation of DNA replication as well as transcription or translation. Indeed, binding of transcription factors to their target sites as well as interactions among transcription factors play critical roles in gene expression. The proteome consists of ~170,000 individual proteins with multiple splice variants, intracellular processing, distribution, structure, function, and pathway involvement. Post-transcriptional and post-translational genomic entities include the proteome, lipidome, and metabolome, to mention a few. However, these areas are outside of the scope of the current review.
The current studies on association of microbiome and SPTD/SPTB have been excluded from the present review.52,53,54
Genome-wide linkage analysis
In the past, linkage analysis was the principal tool used to define single-gene diseases. In the first genomic analysis of SPTB/SPTD, seven large families from Northern Finland were analyzed using autosomal markers for either SPTB or SPTD outcomes.55 The study found that the gene encoding type 1 insulin-like growth factor receptor (IGFR1) was associated with SPTB. The study also identified joint haplotypes shared by several SPTB-affected families, and these results were replicated in a case–control study (Table 2). IGF1R influences growth, differentiation, inflammation, immunity, and susceptibility to infections. IGF1 may influence susceptibility to preterm birth.56 IGFbp1 binds IGF1 and IGF2, thereby altering their half-life and function. Cervical-phosphorylated IGFbp1 has value in predicting SPTD.57,58
X-chromosome linkage analysis revealed an association between SPTB and an exonic variant of androgen receptor (AR). This SPTB-associating variant comprises long CAG repeats on the exon that decrease AR activity and are associated with susceptibility to SPTB.59 There is evidence that androgen activity throughout pregnancy and androgen withdrawal in late pregnancy are of importance.60,61 An intron variant of CXCR3 in X-chromosome was also associated with SPTB.62 This chemokine receptor is abundant in trophoblasts. In cord blood in SPTB cases, the SPTB-predisposing CXCR3-SNV on rs220964 was highly expressed, and this increased expression is associated with high cord blood levels of CXCL9, a CXCR3 ligand. Cxcr3 deletion modified the maternal LPS-induced cytokine response in the fetal compartments62 (Table 2).
Whole-exome sequencing
WES analyses involve families with high prevalence of SPTB and SPTD. The analysis provides a very high number of variants in comparison to mostly a limited study population. According to feasible assumption, many WES studies focus on rare (frequency < 0.01), possibly damaging SNVs (Table 1).
A WES analysis followed by pathway analysis of ten mothers, including two mother–daughter pairs from families highly affected by SPTD/SPTB, revealed the complement and coagulation cascade among the top pathways. This feasible finding was further tested in case–control setting of 565 Finnish mothers and involving 67 promising coding region SNVs of CFH, CR1, F13B, F5, CR2 and C4BPA. A missense variant of CR1 was most significantly associated with SPTD63 (Table 2).
A genome-wide targeted screening identified IGF1 as candidate of preterm birth.56 Subsequent study involved sequencing of haplotype blocks and selected exomes with the aim to define pathways and candidate genes. Comparison of preterm birth-affected families and final analysis identified IGF1, ATM, and IQGAP2 as the most promising and connected.64
In African Americans prone to PPROM-SPTB, selective WES analysis identified potentially damaging missense variants in genes that encode fibrillar collagens and proteins involved in collagen synthesis. The SMV data were further studied in a SPTB-PPROM population and controls born at term.65 Another WES analysis identified genes involved in innate immunity. A targeted case–control replication study confirmed associations between SPTB and several rare alleles. Of the likely damaging rare alleles in the innate immunity and host defense genes, CARD6, DEFB1, FUT2, MBL2, NLRP10, and NOD2 were only detected in the PPROM-SPTB cases66 (Table 2).
GWAS studies
In GWAS analyses, because there are about 106 binomial comparisons to be made, the p value must be <5 × 10−8 to reach statistical significance. Since a consistent large difference in the risk ratio of the common allele distribution between cases and controls of identical ethnicity is practically unattainable, a large population of cases and controls is required. A lack of homogeneity, particularly in multiethnic populations, is a limitation that causes population stratification, which complicates analyses. Independent replications are required to confirm the results. Large and accurate GWASs and meta-analyses will likely identify the most robust candidate genes for future research. Rare SNVs identified among families with recurrent phenotype by WES/GWS studies may nominally replicate in large GWAS studies (Table 1).
The first GWAS of SPTB included 2072 pregnant singleton mothers and their newborns in the USA. The racial distribution was mixed in this study, with 59% of Caucasian ethnicity, and the ethnic distribution was similar between cases and controls. The team validated the 96 best SNVs in 493 mothers and their children: variants associated with CCDC25 and DCP1A yielded the lowest p values (1 × 10−6). However, these results were not replicated. The authors stated that the heterogeneity of the SPTB/SPTD phenotype may be a serious limitation67 (Table 2).
Another GWAS included 1921 mothers and 1199 children from a Norwegian birth register cohort.68 The outcomes included labor-initiated and PPROM-initiated SPTD and SPTB. These phenotypes were analyzed separately and together. The top candidate gene, DPP6, was associated with PPROM-SPTD (p = 5.1 × 10−7). Several replicated loci from the maternal GWAS and a literature-informed top candidate gene set showed a nominal association with the phenotypes.
A recent GWAS study of SPTB included 1349 SPTB cases and 12,595 ancestry-matched controls classified into population cohorts by ethnicity (Table 2).69 Two intergenic loci, each in one specific ancestry, were associated with SPTB at a genome-wide level of significance. The investigators were unable to identify a replication cohort for these findings. Despite the negative result of their large study, the authors stressed the possibility that ancestry-based environmental selective pressure may contribute to genetic susceptibility to SPTD.
Advanced genomic studies
Midway between genomic and candidate gene studies is the selective genomic approach. Plunkett70 conducted a representative study of this type based on the hypothesis that human gestation time has been shortened relative to other primates. Accordingly, many genes involved in parturition would display accelerated evolution in human lineages. This study tested the association between SPTD and 150 genes and around 8400 SNVs with rapid phylogeny. Of these, FSHR contained eight of the ten SNVs associated with SPTD in Caucasian (including Finnish) populations. An African American population also had a haplotype of the FSHR gene that nominally associated with preterm delivery.70 FSH/FSHR influences contractility of the uterus and expression of progesterone receptor (PR).
In another approach genetic analysis is combined with placental proteome analysis. A recent analysis investigated SNVs (n = 77) in ten fetal genes encoding placental proteins associated with the duration of pregnancy. Of these, only a SNV within CPPED1 associated with pregnancy duration.71 Both CPPED1 protein and CPPED1 mRNA in the basal plate of the placenta were associated with labor and length of pregnancy. CPPED1 is a phosphatase that inactivates AKT1 in the PI3K/AKT1 signaling pathway in bladder cancer cells.72 A pathway analysis in which siRNA was used to inhibit CPPED1 expression in trophoblast-derived cells revealed that CPPED1 affects expression of genes involved in angiogenesis and proximal PI3K/AKT1 signaling pathways.71 Potential phosphorylation targets of AKT1 include inactivation of hypo phosphorylated FOXO1, which participates in the progesterone receptor B (PR-B) transcriptional complex.73
Thus far, only one GWAS study has identified a significant association with the risk of SPTD. This study also found significant associations with duration of pregnancy.43 This landmark study described the GWAS findings from a total of 43,568 mothers of European ancestry. The discovery population represents self-reported data regarding the first pregnancy, including length of gestation, and apparently excludes indicated preterm births. The replication studies included three Nordic data sets with a total of 8643 mothers. Overall, 14 genomic loci achieved significant (p < 5 × 10−8, four genes) or suggestive (p < 1 × 10−6, ten genes) associations (Table 1). EBF1, EEFSEC, AGTR2, and WNT4 were significantly associated with at least one of the outcomes in the discovery stage. Remarkably, all of these findings were replicated in the Nordic data sets. In addition, ADCY5 and RAP2C, which had suggestive significance in the discovery stage, achieved genome-wide significance in the joint analysis with the replicates. All associations were of maternal origin, as suggested by the analysis of mother–infant dyads.
The top SNVs in both the discovery and replication stages for SPTD and for gestational length were associated with early B-cell factor 1 (EBF1), which is essential for normal B cell development. Previous GWAS studies have identified associations of EBF1 with cardiovascular, metabolic, and endocrine risk, as well as with low birth weight.74 Likewise, SPTD-associating variants near angiotensin II receptor, type 2 (AGTR2) on the X chromosome may modulate uteroplacental circulation and contribute to the risk of preeclampsia.75 The renin–angiotensin system and oxidative stress are potential mechanisms for both preeclampsia and SPTD. Identification of an association between SPTD and AGTR2 is consistent with the possibility that insufficient oxygen and nutrient uptake by the placenta are not exclusively associated with fetal growth restriction and elective preterm delivery. The combination of placental perfusion defect and CA is associated with SPTD and poor neurological outcome;76 a modest decrease in fetal growth is also associated with SPTB.77,78
Zhang et al. also found that variants of eukaryotic elongation factor selenocysteine-tRNA-specific (EEFSEC) are associated with SPTD and length of pregnancy.43 The SPTD-associated SNV rs9998765 is in linkage disequilibrium with hypospadias- and menarche age-associated SNVs. EEFSEC participates in the incorporation of selenocysteine (Sec) into Sec-proteins. Phosphorylated selenium serves as a substrate when phosphoserine-tRNA is converted to Sec-tRNA (Fig. 2a–c). Transfer of Sec-tRNA to Sec-protein requires both a cis-acting Sec insertion sequence (SECIS) element in the 3′ untranslated region (3′ UTR) of each Sec-protein and several specific trans-acting factors. These may include EEFSEC, SECISBP2, L30, nucleolin, and EIF4A3 (Fig. 2d).
Intake, metabolism, and incorporation of selenium into selenoproteins. a List of selenium-containing compounds in the diet. b Metabolism of Se-compounds. Selenomethionine may replace methionine in proteins. According to what is currently known, it does not alter protein function and therefore is likely part of selenium storage. c Incorporation of selenide into selenocysteine (Sec) tRNA(Sec)Ser. The pathway consists of transformations catalyzed by the enzymes shown in the figure. SPS2 is a Sec-protein. d Synthesis of Sec-proteins. Sec-tRNA(Sec)Ser uniquely has a UGA in the open reading frame. In the 3′ UTR of the genes encoding Sec-proteins, the stem-loop structure known as Sec insertion sequence (SECIS) designates the UGA triplet that otherwise is a termination codon. Gene-specific differences in the SECIS elements affect how well trans-acting factors influence translation efficiency. EEFSEC and SECISPB2 are major trans-acting factors; additional factors may include nucleolin, EIF4A3, and L30. e List of 25 Sec-proteins identified in the human genome and transcriptome and brief statement about the function. Alone among Sec-proteins, SelenoP contains more than one Sec amino acid
The human genome contains 25 Sec-proteins, including glutathione peroxidases (GPX) (n = 5), thioredoxin reductases (n = 3), thyroid hormone deiodinases (n = 3), proteins involved in endoplasmic reticulum functions (n = 3), and others (Fig. 2e). SelenoP contains 10 Sec amino acids and it transports selenium to tissues.79 Apolipoprotein E receptor-2 (apoER2) facilitates uptake of SEPP1 in the nervous system, testis, and placenta. In addition, protein-bound methionine binds selenium to selenomethionine (Semet).
Selenium protects against acute pro-oxidant injury. An in vitro study revealed that knockdown of SECISBP2 reduces levels of selenoproteins GPX1, SelK, and DiO2 and increases oxidative stress in trophoblast cells. When SECISBP2 expression is inhibited by siRNA, the synthesis of human chorion gonadotropin (hGC) and progesterone decreased, and the functions of trophoblasts deteriorated.80 The recommended daily intake of selenium ranges from 55 to 90 μg. Bioavailability of selenium may depend on the quality of Se-nutrient and on intracellular uptake of SelenoP79 or selenite81 (Fig. 2a). Both severe Se-excess and Se-deficiency cause serious symptoms.82,83 Inborn errors of Sec-proteins cause protein-specific traits.84 Male genitalia, thyroid, brain, and placenta have high Se-content and their Se-contents are resistant to acute Se-deficiency.79 Serum selenium concentration in early pregnancy does not consistently associate with duration of pregnancy.85 In Malawi, which has a high prematurity rate, the selenium content of the soil is low.86 In Finland, selenium supplementation of the fertilizers started in 1984, because of the low Se-content in soil. In Northern Finland, the singleton SPTB-rate was ~50% lower in 1987 than in 1966.87 Multiple factors could influence on such a large difference, starting from the difference in the calculation of the pregnancy length.
Zhang et al. showed that WNT4 is associated with pregnancy duration, and they discussed one feasible mechanism behind this phenotype.43 The WNT gene family consists of structurally related genes that encode secreted signaling proteins. These proteins have been implicated in several developmental processes. During placentation, WNT4 is a downstream target of bone morphogenetic protein 2 (BMP2), a member of the TGFβ superfamily; activation of WNT4 by BMP2 induces endometrial stroma cell proliferation and differentiation into decidua stroma cells.88 Zhang et al. showed that WNT4 variants influence the binding affinity of estrogen receptor-1 (ESR1) during decidualization, which includes differentiation of endothelial fibroblasts into secretory decidua cells. Estrogen plays a major role in vascularization during formation of the decidua. Analysis of expression of human endothelial stromal cells revealed a striking difference in the WNT4 allele-dependent binding affinity of electrophoretic mobility shift assay(EMSA)-validated ESR1 to WNT4. These findings imply that differentiation of the endometrium into the decidua in early pregnancy sets the stage for placentation and may influence the duration of pregnancy.
The most recent study concerns the WES analysis of families with predisposition to SPTD.89 The aim was to identify rare frequency (<0.01), possibly damaging nucleotide variants in complex families with recurrent SPTD. WES analyses included 17 mothers of the northern Finnish origin. The replication population included 93 Danish sister pairs and two triads, all with history of preterm delivery (Table 1). The glucocorticoid receptor (GR) signaling pathway was significantly affected by these rare variants (p < 1.7e−8). This pathway was replicated in the Danish sister pairs. A gene in this pathway, heat shock protein family A (Hsp70) member 1 like (HSPA1L), contained two likely damaging missense alleles, identified in four different Finnish families. One of them (rs34620296) was also more frequent in cases than in controls (0.0025 vs. 0.0010, p = 0.002) of the large GWAS study.43 In silico analysis predicted an additional phosphorylation site generated by the rs34620296 variant. Finally, decidualization experiment revealed that HSPA1L Ala268Thr (rs34620296) variants influenced the protein level of GR and expression of WNT4; the latter was previously shown to associate with SPTD.43
Comment
Currently, in about 50% of preterm births, there are no identified risk factors. Mendelian randomization analysis based on genomic data suggest that the association between maternal height and length of pregnancy is causal, which indicates that anthropometry influences the duration of pregnancy.90 Human evolution, characterized by prone position, a narrow birth canal, and a large head, may have favored a decrease in the duration of pregnancy that may even associate with SPTD, leading to fetal demise, which was less harmful than a too large fetus that additionally threatened the mother.70 Animal models are useful for investigating the induction of SPTD but understanding the variance in the duration of pregnancy requires species-specific approaches.
Genomic candidate genes for SPTD/SPTB identified thus far indicate that development, growth and immunity are important for pregnancy duration, particularly formation of the decidua and vascularization of the placenta. The cervix and uterine corpus respond to signals from both the mother and fetus. Further genomic studies are required. The generally low-expressivity alleles have a higher impact on the prematurity rate than the rare high-penetrance alleles associated with recurrent SPTD. However, the latter likely reveal SPTD-predisposing pathways that contain additional disease genes and variants.
Investigation of the candidate genes, pathways, gene–environment interactions and the underlying epigenetic regulation will further explain why some individuals are more sensitive to SPTD-triggering events than the other. Prevention of SPTD/SPTB requires multiple approaches and individualized prevention strategies.
Conclusion
Current research on prevention of SPTB has been concentrating on prevention of the imminent preterm birth. Shifting the target towards early detection of the risk, may be a more promising approach that eventually can lead to effective prophylaxis or early treatment of pregnancies with established risk of SPTD. Identifying mothers that are at the highest genetic risk for SPTD may allow individualized strategies for prevention.
Optimal duration of pregnancy that involves appropriate early development, growth, and differentiation, is among the most critical events influencing both the duration and the quality of postnatal life and therefore require considerable intensification of research.
References
Blencowe, H. et al. Preterm birth-associated neurodevelopmental impairment estimates at regional and global levels for 2010. Pediatr. Res. 74(Suppl 1), 17–34 (2013).
Yoshida, S. et al. Setting research priorities to improve global newborn health and prevent stillbirths by 2025. J. Glob. Health 6, 010508 (2016).
Goldenberg, R. L., Culhane, J. F., Iams, J. D. & Romero, R. Epidemiology and causes of preterm birth. Lancet 371, 75–84 (2008).
Muglia, L. J. & Katz, M. The enigma of spontaneous preterm birth. N. Engl. J. Med. 362, 529–535 (2010).
Al Jishi, T. & Sergi, C. Current perspective of diethylstilbestrol (DES) exposure in mothers and offspring. Reprod. Toxicol. 71, 71–77 (2017).
Troisi, R., Hatch, E. E. & Titus, L. The diethylstilbestrol legacy: a powerful case against intervention in uncomplicated pregnancy. Pediatrics 138(Suppl 1), S42–S44 (2016).
Stewart, L. A. et al. Evaluating progestogens for prevention of preterm birth international collaborative (EPPPIC) individual participant data (IPD) meta-analysis: protocol. Syst. Rev. 6, 235 (2017).
Smid, M. C., Stringer, E. M. & Stringer, J. S. A worldwide epidemic: the problem and challenges of preterm birth in low- and middle-income countries. Am. J. Perinatol. 33, 276–289 (2016).
Lunde, A., Melve, K. K., Gjessing, H. K., Skjaerven, R. & Irgens, L. M. Genetic and environmental influences on birth weight, birth length, head circumference, and gestational age by use of population-based parent-offspring data. Am. J. Epidemiol. 165, 734–774 (2007).
Wilcox, A. J., Skjaerven, R. & Lie, R. T. Familial patterns of preterm delivery: maternal and fetal contributions. Am. J. Epidemiol. 167, 474–479 (2008).
Romero, R., Dey, S. K. & Fisher, S. J. Preterm labor: one syndrome, many causes. Science 345, 760–765 (2014).
Adams Waldorf, K. M. et al. Uterine overdistention induces preterm labor mediated by inflammation: observations in pregnant women and nonhuman primates. Am. J. Obstet. Gynecol. 213, 830.e1–830.e19 (2015).
Renthal, N. E., Williams, K. C. & Mendelson, C. R. MicroRNAs--mediators of myometrial contractility during pregnancy and labour. Nat. Rev. Endocrinol. 9, 391–401 (2013).
Hadley, C. B. 1, Main, D. M. & Gabbe, S. G. Risk factors for preterm premature rupture of the fetal membranes. Am. J. Perinatol. 7, 374–379 (1990).
Kim, C. J. et al. Chronic inflammation of the placenta: definition, classification, pathogenesis, and clinical significance. Am. J. Obstet. Gynecol. 213, S53–S69 (2015).
Bonney, E. A. Alternative theories: Pregnancy and immune tolerance. J. Reprod. Immunol. 123, 65–71 (2017).
Hillhouse, E. W. & Grammatopoulos, D. K. Role of stress peptides during human pregnancy and labour. Reproduction 124, 323–329 (2002).
Velez Edwards, D. R., Baird, D. D., Hasan, R., Savitz, D. A. & Hartmann, K. E. First-trimester bleeding characteristics associate with increased risk of preterm birth: data from a prospective pregnancy cohort. Hum. Reprod. 27, 54–60 (2012).
Schatz, F., Guzeloglu-Kayisli, O., Arlier, S., Kayisli, U. A. & Lockwood, C. J. The role of decidual cells in uterine hemostasis, menstruation, inflammation, adverse pregnancy outcomes and abnormal uterine bleeding. Hum. Reprod. Update 22, 497–515 (2016).
Morgan, T. K. Role of the placenta in preterm birth: a review. Am. J. Perinatol. 33, 258–266 (2016).
Mendelson, C. R., Montalbano, A. P. & Gao, L. Fetal-to-maternal signaling in the timing of birth. J. Steroid Biochem. Mol. Biol. 170, 19–27 (2017).
Hallman, M., Arjomaa, P., Mizumoto, M. & Akino, T. Surfactant proteins in the diagnosis of fetal lung maturity. I. Predictive accuracy of the 35 kD protein, the lecithin/sphingomyelin ratio, and phosphatidylglycerol. Am. J. Obstet. Gynecol. 158, 531–535 (1988).
Hallman, M. The surfactant system protects both fetus and newborn. Neonatology 103, 320–326 (2013).
Byrns, M. C. Regulation of progesterone signaling during pregnancy: implications for the use of progestins for the prevention of preterm birth. J. Steroid Biochem. Mol. Biol. 139, 173–181 (2014).
Mesiano, S. et al. Progesterone withdrawal and estrogen activation in human parturition are coordinated by progesterone receptor A expression in the myometrium. J. Clin. Endocrinol. Metab. 87, 2924–2930 (2002).
Patel, B. et al. Role of nuclear progesterone receptor isoforms in uterine pathophysiology. Hum. Reprod. Update 21, 155–173 (2015).
Petraglia, F., Imperatore, A. & Challis, J. R. Neuroendocrine mechanisms in pregnancy and parturition. Endocr. Rev. 31, 783–816 (2010).
Renthal N. E., et al. Molecular regulation of parturition: a myometrial perspective. Cold Spring Harb. Perspect. Med. 5, pii: a023069 (2015).
Menon, R., Bonney, E. A., Condon, J., Mesiano, S. & Taylor, R. N. Novel concepts on pregnancy clocks and alarms: redundancy and synergy in human parturition. Hum. Reprod. Update 22, 535–560 (2016).
Iams, J. D., Romero, R., Culhane, J. F. & Goldenberg, R. L. Primary, secondary, and tertiary interventions to reduce the morbidity and mortality of preterm birth. Lancet 371, 164–175 (2008).
Kenyon, S., Boulvain, M. & Neilson, J. P. Antibiotics for preterm rupture of membranes. Cochrane Database Syst. Rev. 12, CD001058 (2013).
Ruiz, L., Moles, L., Gueimonde, M. & Rodriguez, J. M. Perinatal microbiomes’ influence on preterm birth and preterms’ health: influencing factors and modulation strategies. J. Pediatr. Gastroenterol. Nutr. 63, e193–e203 (2016).
Haas, D. M., Caldwell, D. M., Kirkpatrick, P., McIntosh, J. J. & Welton, N. J. Tocolytic therapy for preterm delivery: systematic review and network meta-analysis. Brit. Med. J. 345, e6226 (2012).
Romero, R. et al. Vaginal progesterone decreases preterm birth and neonatal morbidity and mortality in women with a twin gestation and a short cervix: an updated meta‐analysis of individual patient data. Ultrasound Obstet. Gynecol. 49, 303–314 (2017).
Romero, R. et al. Vaginal progesterone for preventing preterm birth and adverse perinatal outcomes in singleton gestations with a short cervix: meta-analysis of individual patient data. Am. J. Obstet. Gynecol. 218, 161–180 (2018).
Norman, J. E. et al. Vaginal progesterone prophylaxis for preterm birth (the OPPTIMUM study): a multicentre, randomised, double-blind trial. Lancet 387, 2106–2116 (2016).
Norman, J. E. et al. Does progesterone prophylaxis to prevent preterm labour improve outcome? A randomized double-blind placebo-controlled trial (OPPTIMUM). Health Technol. Assess. 22, 1–304 (2018).
Bezold, K. Y., Karjalainen, M. K., Hallman, M., Teramo, K. & Muglia, L. J. The genomics of preterm birth: from animal models to human studies. Genome Med. 5, 34 (2013).
York, T. P., Strauss, J. F. 3rd, Neale, M. C. & Eaves, L. J. Estimating fetal and maternal genetic contributions to premature birth from multiparous pregnancy histories of twins using MCMC and maximum-likelihood approaches. Twin Res Hum. Genet 12, 333–342 (2009).
Alleman, B. W. et al. No observed association for mitochondrial SNPs with preterm delivery and related outcomes. Pediatr. Res. 72, 539–544 (2012).
Manuck, T. A. et al. Absence of mitochondrial progesterone receptor polymorphisms in women with spontaneous preterm birth. Reprod. Sci. 17, 913–916 (2010).
Velez, D. R. et al. Mitochondrial DNA variant A4917G, smoking and spontaneous preterm birth. Mitochondrion 8, 130–135 (2008).
Zhang, G. et al. Genetic associations with gestational duration and spontaneous preterm birth. N. Engl. J. Med. 377, 1156–1167 (2017).
Rosenberg, N. A. et al. Genome-wide association studies in diverse populations. Nat. Rev. Genet. 11, 356–366 (2010).
Parets, S. E., Conneely, K. N., Kilaru, V., Menon, R. & Smith, A. K. DNA methylation provides insight into intergenerational risk for preterm birth in African Americans. Epigenetics 10, 784–792 (2015).
Palo, J. U., Ulmanen, I., Lukka, M., Ellonen, P. & Sajantila, A. Genetic markers and population history: Finland revisited. Eur. J. Hum. Genet. 17, 1336–1346 (2009).
Sheikh, I. A. et al. Spontaneous preterm birth and single nucleotide gene polymorphisms: a recent update. BMC Genom. 17(Suppl 9), 759 (2016).
Plunkett, J. & Muglia, L. J. Genetic contributions to preterm birth: implications from epidemiological and genetic association studies. Ann. Med. 40, 167–195 (2008).
Rood, K. M. & Buhimschi, C. S. Genetics, hormonal influences, and preterm birth. Semin. Perinatol. 41, 401–408 (2017).
Strauss, J. F. et al. Spontaneous preterm birth: advances toward the discovery of genetic predisposition. Am. J. Obstet. Gynecol. 218, 294–314 (2018).
Singh, A. J., Ramsey, S. A., Filtz, T. M. & Kioussi, C. Differential gene regulatory networks in development and disease. Cell. Mol. Life Sci. 75, 1013–1025 (2017).
Prince, A. L. et al. The placental membrane microbiome is altered among subjects with spontaneous preterm birth with and without chorioamnionitis. Am. J. Obstet. Gynecol. 214, 627.e16 (2016).
Nelson, D. B., Shin, H., Wu, J. & Dominguez-Bello, M. G. The gestational vaginal microbiome and spontaneous preterm birth among Nulliparous African American women. Am. J. Perinatol. 33, 887–893 (2016).
Dahl, C. et al. Gut microbiome of mothers delivering prematurely shows reduced diversity and lower relative abundance of Bifidobacterium and Streptococcus. PLoS ONE 12, e0184336 (2017).
Haataja, R. et al. Mapping a new spontaneous preterm birth susceptibility gene, IGF1R, using linkage, haplotype sharing, and association analysis. PLoS Genet. 7, e1001293 (2011).
Uzun, A., Dewan, A. T., Istrail, S. & Padbury, J. F. Pathway-based genetic analysis of preterm birth. Genomics 101, 163–170 (2013).
Rahkonen, L. et al. Elevated levels of decidual insulin-like growth factor binding protein-1 in cervical fluid in early and mid-pregnancy are associated with an increased risk of spontaneous preterm delivery. BJOG 117, 701–710 (2010).
Conde-Agudelo, A. & Romero, R. Cervical phosphorylated insulin-like growth factor binding protein-1 test for the prediction of preterm birth: a systematic review and meta-analysis. Am. J. Obstet. Gynecol. 214, 57–73 (2016).
Karjalainen, M. K. et al. A potential novel spontaneous preterm birth gene, AR, identified by linkage and association analysis of X chromosomal markers. PLoS ONE 7, e51378 (2012).
Bethin, K. E. et al. Microarray analysis of uterine gene expression in mouse and human pregnancy. Mol. Endocrinol. 17, 1454–1469 (2003).
Makieva, S., Saunders, P. T. & Norman, J. E. Androgens in pregnancy: roles in parturition. Hum. Reprod. Update 20, 542–559 (2014).
Karjalainen, M. K. et al. CXCR3 polymorphism and expression associate with spontaneous preterm birth. J. Immunol. 195, 2187–2198 (2015).
McElroy, J. J. et al. Maternal coding variants in complement receptor 1 and spontaneous idiopathic preterm birth. Hum. Genet. 132, 935–942 (2013).
Uzun, A. et al. Targeted sequencing and meta-analysis of preterm birth. PLoS ONE 11, e0155021 (2016).
Modi, B. P. et al. Rare mutations and potentially damaging missense variants in genes encoding fibrillar collagens and proteins involved in their production are candidates for risk for preterm premature rupture of membranes. PLoS ONE 12, e0174356 (2017).
Modi, B. P. et al. Mutations in fetal genes involved in innate immunity and host defense against microbes increase risk of preterm premature rupture of membranes (PPROM). Mol. Genet. Genom. Med. 5, 720–729 (2017).
Zhang, H. et al. A genome-wide association study of early spontaneous preterm delivery. Genet. Epidemiol. 39, 217–226 (2015).
Bacelis, J. et al. Literature-informed analysis of a genome-wide association study of gestational age in Norwegian women and children suggests involvement of inflammatory pathways. PLoS ONE 11, e0160335 (2016).
Rappoport, N. et al. A genome-wide association study identifies only two ancestry specific variants associated with spontaneous preterm birth. Sci. Rep. 8, 226 (2018).
Plunkett, J. et al. An evolutionary genomic approach to identify genes involved in human birth timing. PLoS Genet. 7, e1001365 (2011).
Haapalainen, A. M. et al. Expression of CPPED1 in human trophoblasts is associated with timing of term birth. J. Cell. Mol. Med. 22, 968–981 (2018).
Zhuo, D. X. et al. CSTP1, a novel protein phosphatase, blocks cell cycle, promotes cell apoptosis, and suppresses tumor growth of bladder cancer by directly dephosphorylating Akt at Ser473 site. PLoS ONE 8, e65679 (2013).
Diep, C. H. et al. Progesterone receptors induce FOXO1-dependent senescence in ovarian cancer cells. Cell Cycle 12, 1433–1449 (2013).
Beaumont, R. N. et al. Genome-wide association study of offspring birth weight in 86577 women identifies five novel loci and highlights maternal genetic effects that are independent of fetal genetics. Hum. Mol. Genet. 27, 742–756 (2018).
GOPEC Consortium. Disentangling fetal and maternal susceptibility for pre-eclampsia: a British multicenter candidate-gene study. Am. J. Hum. Genet. 77, 127–131 (2005).
Kaukola, T. et al. Population cohort associating chorioamnionitis, cord inflammatory cytokines and neurologic outcome in very preterm, extremely low birth weight infants. Pediatr. Res. 59, 478–483 (2006).
Marsál, K. Intrauterine growth restriction. Curr. Opin. Obstet. Gynecol. 14, 127–135 (2002).
Zeitlin, J., Ancel, P. Y., Saurel-Cubizolles, M. J. & Papiernik, E. The relationship between intrauterine growth restriction and preterm delivery: an empirical approach using data from a European case-control study. Br. J. Obstet. Gynecol. 107, 750–758 (2000).
Burk, R. F. & Hill, K. E. Regulation of Selenium metabolism and transport. Annu. Rev. Nutr. 35, 109–134 (2015).
Li, M. et al. Loss of selenocysteine insertion sequence binding protein 2 suppresses the proliferation, migration/invasion and hormone secretion of human trophoblast cells via the PI3K/Akt and ERK signaling pathway. Placenta 55, 81–89 (2017).
McDermott, J. R. et al. Zinc- and bicarbonate-dependent ZIP8 trasporter mediates selenite uptake. Oncotarget 7, 35327–35340 (2016).
MacFarquhar, J. K. et al. Acute selenium toxicity associated with a dietary supplement. Arch. Intern. Med. 170, 256–261 (2010).
Zhou, H., Wang, T., Li, Q. & Li, D. Prevention of Keshan disease by selenium supplementation: a systematic review and meta-analysis. Biol. Trace Elem. Res. https://doi.org/10.1007/s12011-018-1302-5 (2018).
Schweizer, U. & Fradejas-Villar, N. Why 21? The significance of selenoproteins for human health revealed by inborn errors of metabolism. FASEB J. 30, 3669–3681 (2016).
Rayman, M. P., Wijnen, H., Vader, H., Kooistra, L. & Pop, V. Maternal selenium status during early gestation and risk for preterm birth. Can. Med. Assoc. J. 183, 549–555 (2011).
Hurst, R. et al. Soil-type influences human selenium status and underlies widespread selenium deficiency risks in Malawi. Sci. Rep. 3, 1425 (2013).
Olsen, P. et al. Epidemiology of preterm delivery in two birth cohorts with an interval of 20 years. Am. J. Epidemiol. 142, 1184–1193 (1995).
Knöfler, M. & Pollheimer, J. Human placental trophoblast invasion and differentiation: a particular focus on Wnt signaling. Front. Genet. 4, 190 (2013).
Huusko, J. M. et al. Whole exome sequencing reveals HSPA1L as a genetic risk factor for spontaneous preterm birth. PLoS Genet. 14, e1007394 (2018).
Zhang, G. et al. Assessing the causal relationship of maternal height on birth size and gestational age at birth: a mendelian randomization analysis. PLoS Med. 12, e1001865 (2015).
Acknowledgements
Supported by the Jane and Aatos Erkko Foundation (M.H., M.R.), and the Sigrid Jusélius Foundation (M.H.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hallman, M., Haapalainen, A., Huusko, J.M. et al. Spontaneous premature birth as a target of genomic research. Pediatr Res 85, 422–431 (2019). https://doi.org/10.1038/s41390-018-0180-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41390-018-0180-z
This article is cited by
-
Elevated human placental heat shock protein 5 is associated with spontaneous preterm birth
Pediatric Research (2023)
-
Integrative genetic, genomic and transcriptomic analysis of heat shock protein and nuclear hormone receptor gene associations with spontaneous preterm birth
Scientific Reports (2021)
-
Periodontal Disease and Birth Outcomes: Are We Missing Something?
Current Oral Health Reports (2020)
-
Preterm neonatal immunology at the intestinal interface
Cellular and Molecular Life Sciences (2020)
-
The contributions of genetics to premature birth
Pediatric Research (2019)