Abstract
Background
We aimed to evaluate if lung mechanics measured by forced oscillatory technique (FOT) during the first day of life help identify extremely low gestational age newborns (ELGANs) at risk of prolonged mechanical ventilation (MV) and oxygen dependency.
Methods
Positive end-expiratory pressure (PEEP) was increased 2 cmH2O above the clinically set PEEP, then decreased by four 5-min steps of 1 cmH2O, and restored at the clinical value. At each PEEP, FOT measurements were performed bedside during MV. Changes in respiratory mechanics with PEEP, clinical parameters, and chest radiographs were evaluated.
Results
Twenty-two newborns (24+4 ± 1+4 wks gestational age (GA); birth weight 653 ± 166 g) on assist/control ventilation were studied. Infants were ventilated for 40 ± 36 d (range 1–155 d), 11 developed severe bronchopulmonary dysplasia (BPD) and one died before 28 d. Early lung mechanics correlated with days on MV, days of respiratory support, and BPD grade. Effects of increasing PEEP on oscillatory reactance assessed by FOT together with GA and radiographic score predicted days on MV (multilinear model, r2 = 0.73). A logistic model considering the same FOT parameter together with GA predicts BPD development.
Conclusions
FOT can be applied bedside in ELGANs, where early changes in lung mechanics with PEEP improve clinical prediction of respiratory outcomes.
Similar content being viewed by others
Introduction
Despite improvements in the management of extremely low gestational age newborn (ELGAN) infants, they are at high risk of requiring prolonged mechanical ventilation, prolonged oxygen dependency, and developing bronchopulmonary dysplasia (BPD).1,2
Many studies have tried to identify early risk factors and prediction models for respiratory outcomes3,4,5 in order to personalize treatment in infants at high risk of developing respiratory disease. Despite several efforts, the prediction models based on available clinical data show inadequate accuracy, suggesting the need of identifying other variables to improve their performance.3
Among the functional parameters proposed as additional risk factors, studies have shown that altered lung mechanics in the first days of life identify infants at risk of needing prolonged respiratory support or developing BPD.6,7,8,9,10,11,12,13,14,15,16,17,18 Unfortunately, the techniques used for assessing lung function in these studies are often time and effort demanding, require trained personal, interfere with ventilation, and cause discomfort to the infant.19 These limitations prevent lung function tests being applied for screening preterm infants in clinical practice.
The Forced Oscillation Technique (FOT) is a non-invasive method suitable for monitoring lung mechanics without interfering with the breathing activity of the patient or ongoing ventilatory modes.20 It is based on the application of high-frequency low-amplitude pressure oscillations at the airway opening while measuring the resulting flow. FOT has been successfully applied in studying changes in lung mechanics in ventilated preterm newborns21,22 and, as it can be easily integrated into a mechanical ventilator without modifying the hardware of the device, it can be performed without additional external equipment.23
The aim of this study is to assess if lung mechanical parameters obtained by FOT during the first day of life provide useful, independent information from other easily available clinical parameters for identifying ELGANs at risk of prolonged mechanical ventilation (MV), oxygen need and development of BPD.
Materials and methods
This study was performed in the Neonatal Intensive Care Unit of Uppsala University Children Hospital, Uppsala, Sweden, between 2011 and 2014. The study was approved by the local Ethics Committee and informed parental consent was obtained for each infant (D:nr 99092).
Study population and protocol
Preterm infants born before 28 weeks gestational age (GA) and assisted by invasive MV during the first day of life were eligible for the study. Exclusion criteria were the presence of major congenital anomalies or hemodynamic instability.
All infants were ventilated (Stephanie ventilator, Fritz Stephan GmbH, Germany) with assist control (A/C) mode.
During the first day of life, lung mechanics were measured in supine position by FOT at different PEEPs. Prior to study measurements, all infants had received surfactant and chosen PEEP (“clinical PEEP”) was set according to oxygenation and auscultation criteria by the attending physician. Therefore, our FOT PEEP trial was not designed for lung recruitment but rather for probing changes in lung mechanics over a small range of PEEP values, in order to quantify lung distensibility (pressure dependence of lung compliance) and tendency to derecruit (instability of lung periphery).
The measurements were performed by starting from the clinically set PEEP value. PEEP was initially increased by 2 cmH2O and then gradually decreased by four steps of 1 cmH2O. At the end of the decreasing trial, PEEP was restored to clinically set value (Supplemental Figure S1 (online)). Each step lasted 5 min and FOT was applied for 2 min after 3 min of stabilization. FiO2 was adjusted by the attending clinician when needed for maintaining SpO2 within clinically set range. All the other ventilatory parameters were kept constant.
Measurements
SpO2, heart rate, and blood pressure were monitored continuously with the patient data management system (IntelliVue Clinical Information Portfolio, ICIP, Philips Healthcare, Eindhoven, Netherlands) during the study.
The FOT signal was generated by the ventilator by superimposing a sinusoidal HFOV waveform (Inspiratory/Expiratory time = 1:1) with an amplitude of 2 cmH2O to the A/C one without interfering with settings made by the attending clinician. Measurements were performed after setting HFOV frequency at 10 Hz and then at 5 Hz for 1 min each. PEEP was manually adjusted to match the preset one as the ventilator could not control it properly when set in this modality.
Pressure was measured at the inlet of the ETT by a differential pressure transducer (PXLA0075DN, Sensym, Milpitas, CA) and the airflow signal at the airway opening was provided by the ventilator as analog output. This set-up did not increase dead space nor equipment resistance sensed by the patient.
Flow and pressure signals were sampled at 300 Hz and stored on a personal computer. The frequency response of the measurement set-up was computed and digitally compensated. The accuracy of the measurements was verified in vitro (errors < 8%).
Clinical data were collected from both medical records and computerized surveillance sheet (detailed list reported in Table 1 and Supplemental Table S1 (online)). Chest radiographs, routinely performed during the first hours after birth in patients in need of MV, were also collected.
Data analysis
Clinical data
Oxygenation index (OI) and ventilation index (VI) were computed as FiO2 × MAP × 100/PaO2 and PaCO2 × respiratory rate × (PIP-PEEP)/1000 [mmHg × bpm × cmH2O], respectively. FiO2 is the fraction of inspired oxygen, MAP is the mean airway pressure, PaO2 and PaCO2 are the partial pressure of O2 and CO2 in arterial blood, and PIP is peak inspiratory pressure.
Severity of infant respiratory distress syndrome (IRDS) was determined from chest radiograph evaluation according to the grading scale (1–4) described by Paetzel24 (grading description in Supplemental Figure S2 (online)). Two radiologists, masked to patient identity and clinical data, graded independently and twice each (separated by at least 2 weeks) the chest radiograph of the subjects. Rib level of the diaphragm were also evaluated considering the right side of the frontal chest radiograph. The average of the four gradings was divided by the rib level of the diaphragm (IRDS/DH) to account for differences in inspired air at the instant when the X ray was taken.
BPD was defined as oxygen dependency at 36 weeks post-menstrual age (PMA) and was graded as 1–3 (mild, moderate, and severe, respectively).25,26
FOT data
Respiratory system input impedance was computed using the least squared method14 at the two frequencies considered and expressed as resistance (R) and reactance (X), accounting for the resistive and the elastic/inertial properties, respectively.23
The impedance of the ETTs used by patients was measured in vitro and subtracted from the impedance data measured on infants.27 Ten breaths were automatically selected at each frequency and PEEP and their end-expiratory R and X values were averaged to provide one data point per patient per PEEP. We selected the end-expiratory impedance values for two reasons: (1) X values at this volume reflect the amount of alveoli open throughout the whole respiratory cycle (therefore excluding the contribution from intra-tidal recruitment) and (2) R values are not affected by the possible increase due to the development of turbulent flow within the airways as in this moment the respiratory flow is close to zero.
Definition of FOT-based lung function parameters
As X is related to dynamic compliance, it accounts for both lung volume derecruitment and lung (over) distension. In order to specifically characterize the elastic properties of lung parenchyma and the stability of lung periphery we defined a set of FOT-based parameters measured at different PEEPs. X values were considered at the following PEEPs: (1) the clinical PEEP before the trial (XPcl_b), (2) the highest PEEP of the trial (XPM), and (3) the PEEP providing the maximal X value during the decreasing limb of the trial (XM).
Difference in X between the PEEP levels of the trial were computed to describe: (1) the possible lung volume recruitment occurred as consequence of rising PEEP by the difference between X measured at the clinical PEEP during the decremental trial and XPcl_b (ΔXRec) (X measured at the same distending pressure but with different volume history); (2) the possible lung volume derecruitment occurred as consequence of lowering PEEP by the difference between X at the clinical PEEP during the decremental trial and X at the clinical PEEP after the trial (ΔXdeRec) (X measured at the same distending pressure but with different volume history), (3) the distensibility of the lung parenchyma by the difference between XPcl_b and XPM (ΔXdis) (X measured at different distending pressures).
Graphical representation of these parameters is reported in Fig. 1. To avoid possible bias, the clinicians involved in this study were blinded to these data.
Graphical representation of FOT-based lung function parameters on the X vs PEEP curve of a representative subject. Arrows indicate the temporal sequence of the measurements. XPcl_b = X at the clinical PEEP before the trial; XPM = X at the maximal PEEP; XM = maximal X value; ΔXRec = difference between X at the clinical PEEP during the decremental trial and XPcl_b; ΔXdeRec = difference between X at the clinical PEEP during the decremental trial and X at the clinical PEEP after the trial; ΔXdis = difference between XPcl_b and XPM
Statistical analysis
Data were tested for normality using the Kolmogorov–Smirnov test. One-way ANOVA for repeated measurements was used to test the significance of differences in impedance among PEEP levels.
Spearman’s correlation was performed to test whether there was a statistical dependence between respiratory outcomes, lung mechanics and clinical parameters and ρ was used to test the correlation strength.
Differences between infants that developed BPD and the ones without BPD were tested by t-test.
Significant independent variables associated with days of MV before discharge, days of oxygen dependence, and BPD development were identified by multilinear regression and logistic regression analysis, respectively. Complete-case analysis was performed. Demographical data that are commonly considered risk factors for BPD (GA, Z-score of the birth weight [z_BW] and sex) perinatal factors (type of delivery, chorioamnionitis, administration, and duration of prenatal steroids treatment, prolonged premature rupture of the membranes [PPROM], time and dose of surfactant) together with parameters describing respiratory condition (APGAR at 5 min, OI, VI, IRDS/DH) and lung mechanics (RPcl_b, XPcl_b, XM, XPM, ΔXRec, ΔXdeRec, ΔXdis) were considered as input variables. Ventilator settings were not used as input variables because they are related to OI and VI and dependent on local clinical practice. Multilinear models with statistically significant variable coefficients were compared on r2. Models with r2 < 0.70 were excluded. Logistic models with statistically significant variable coefficients and able to correctly predict BPD development were compared on pseudo-R2 and on the areas below the receiver operator characteristic (ROC) curves. Differences were considered statistically significant for p < 0.05.
The desired sample size was computed in G × Power (linear multiple regression: fixed model, single regression coefficient, one tail). To assess effects of adding a FOT parameter in a 3-inputs multiple regression models with alpha = 0.05, power = 0.80, and a large effect size (0.35), 20 patients are needed.
Results
Twenty-two newborn infants were studied during the first day of life (16 ± 8 h from birth) (Table 1).
All infants received early surfactant. Six infants (27%) had PPROM, and all but one received prenatal steroids (further clinical characteristics in Supplemental Table S1 (online) and Supplemental Table S2 (online)).
Infants were ventilated for 40 ± 37d (range 1–155 d) before discharge, 4 and 11 developed BPD grade 2 and 3, respectively, and one died before 28 days.
Measurements were well tolerated by all infants indicating that FOT is feasible even in the first hours after birth. Quality of measurements was good for all patients.
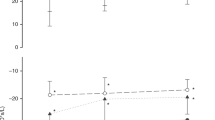
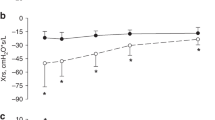
Figure 2 shows changes in respiratory mechanics with PEEP. As the clinical PEEP range was 4–5 cmH2O, the decremental PEEP trial range was 6–2 or 7–3 cmH2O. The difference between absolute X values at the different frequencies was expected, as X depends not only on lung mechanics but also on the oscillation frequency.23 R and X presented similar trends with PEEP at 5 and 10 Hz, with 5 Hz showing the largest changes but also higher variability due to interference of spontaneous breathing activity. Therefore, measurements at 10 Hz were chosen for the analysis. R presents a tendency to decrease with PEEP. X shows marked dependence on PEEP. R and X at the clinical PEEP at 10 Hz ranged from 19 to 72 and from −23 to −88 cmH2O × s/l, respectively. No significant differences were found in SpO2/FiO2 with different PEEPs.
Median (IQR) of ΔXDeRec resulted in 3.90 (1.83;8.39), indicating that lung volume derecruitment occurred in some infants during the trial. Median (IQR) of ΔXRec resulted in 0.02 (–2.20;2.89), and indicated that increasing PEEP during the trial did not recruit lung volume.
GA correlated with R and X values, showing the highest correlation with XM, but not with ΔX parameters. z-BW didn’t correlate with FOT parameters. Correlation between respiratory outcomes and measured parameters are shown in Table 2. Days on MV and days on MV + CPAP correlated with GA, z_BW, XPcl_b, XM, and XPM. Days on MV correlated also with ΔXdis. BPD grade (0–3, where 0 = no BPD) correlated with z_BW and ΔXdis. Hospital days with oxygen dependence correlated only with GA and z_BW.
We did not find any multilinear models for predicting days on MV with statistically significant coefficients and r2 > 0.70 when using one or two inputs among the considered ones (see methods). We found one multilinear model using three-inputs: GA, the radiographic parameter IRDS/DH and the functional parameter ΔXdis (days on MV = 0.002 – 14.2 × GA + 86.7 × IRDS/DH + 3.0*ΔXdis). Normalized coefficients indicated that GA followed by ΔXdis were the parameters that had the strongest influence on days on MV (Fig. 3). Using four inputs did not improve the prediction.
We did not find any multilinear models with statistically significant coefficients able to predict the total hospital days of oxygen dependency even when using up to 4 inputs.
Infants that did or did not develop BPD differed in GA (24.0 ± 1.5 vs 26.1 ± 1.0 wks; p < 0.01), z_BW (−0.94 ± 0.52 vs −0.33 ± 0.74; p = 0.04) and ΔXdis (12.1 ± 5.4 vs 5.8 ± 7.0 cmH2O × s/l; p = 0.04).
One logistic model using one input (GA; logitP = −27.4 + 1.1 × GA) and one logistic model using two inputs were identified for BPD prediction (logitP = −2682.7 + 119.8 × GA-44.4 × ΔXdis). X-normalized coefficient, pseudo-R2, and ROC curves of these models are presented in Fig. 4. Both Pseudo-R2 and ROC curves did not improve adding a third input to the model.
Receiver operating characteristic (ROC) of the developed BPD prediction models. Input parameters with the respective partial-normalized coefficients (as proposed by Agresti29), the area under the ROC curves, and pseudo-r2 are reported
Perinatal factors, such as prenatal steroids, PPROM, chorioamnionitis, and surfactant administration and doses, together with APGAR at 5 min, OI, and VI did not affect days on MV and BPD severity in our material.
Discussion
The main results of this study are: (1) lung function assessment by FOT in clinical practice in mechanically ventilated ELGANs during the first day of life is feasible, (2) X parameters and ΔXdis correlate with respiratory outcomes, (3) ΔXdis in the first day of life in combination with a demographic and a radiographic parameter allow predicting days on MV, (4) considering ΔXdis together with GA improved prediction of BPD development compared to models obtained with standard clinical data in the first day of life.
Our results confirm that lung mechanics in the first day of life is related to respiratory outcomes, as reported earlier using the interrupter or occlusion techniques,6,7,9,11,12,13,15,18 or the equation of motion of the respiratory system, which implies either measuring esophageal pressure or sedation of the infant.8,9,14,16 The use of FOT allowed us to successfully measure lung mechanics at bedside, non-invasively and without interfering with ventilation nor requiring disconnection of the patient from the ventilator at any time. This is not only relevant for clinical usability, comfort, and safety of the patients but also for avoiding possible changes in lung function caused by disconnection.28
Even if FOT is not yet widely available (to our knowledge it is currently implemented in only one commercial neonatal ventilator), it can be applied in clinical practice by using any commercial ventilator able to combine CMV and HFOV, and that allows exporting flow and pressure data to a computer for data analysis.
In our patient population, respiratory outcomes correlated with GA, z-BW, X values and ΔXdis but not with R or other clinical parameters. While R mainly reflects the condition of the central airways, X is related to the elasticity of the lung that is influenced by lung volume, alveolarization, recruitment, and distention. Therefore, it depends on the development of the lung periphery and disease condition. For these reasons, X values correlated highly with GA and depended on PEEP level.
While XPcl_b has the advantage of being measured without requiring any intervention, XM is more independent from the clinical PEEP as it reflects the best mechanical condition of the respiratory system achievable over a wide range of PEEPs, explaining why XM had the strongest correlation to GA.
Compared to previous studies, we considered not only single assessments of mechanical properties but also how lung mechanics change with PEEP.
Measuring X at a single PEEP value has two limitations: (i) it requires the availability of reference equations for the absolute values to correct for anthropometric characteristics of the subjects; (ii) it is impossible to distinguish if low X values are mainly due to lung volume derecruitment or (over) distention.
The PEEP trial we used was designed to facilitate the interpretation of X measurements through the evaluation of the relative changes as a consequence of modifying the PEEP instead of evaluating absolute values. Considering that there are several physiological/pathophysiological mechanisms that strongly influence the relationship between X and PEEP value, it is important to carefully identify the appropriate PEEP range for performing the trial in order to properly interpret the meaning of these data. We decided therefore to perform the test starting at the clinically set PEEP, i.e., the minimal PEEP able to provide optimal SpO2, which means a PEEP value which may be different for each infant but provides a standardized condition (best recruitment) for each patient.
We assessed lung mechanics three times at the clinical PEEP: at baseline, after higher PEEPs and after lower PEEPs. As these measurements are performed at the same distending level (same PEEP), differences in X are mostly due to the lung volume recruitment/derecruitment occurring during the trial. As expected, increasing PEEP by 2 cmH2O did not result in lung recruitment (ΔXRec close to zero) while decreasing PEEP by 2 cmH2O resulted in derecruitment in some infants (ΔXDeRec > 0). We did not find any correlation between the parameters quantifying this (ΔXRec, ΔXDeRec) and respiratory outcomes.
Lung volume derecruitment occurring during the trial prevented distinguishing the relative contribution of decreasing lung distention at low PEEP and lung volume derecruitment to changes in X. On the contrary, as the lungs were recruited before the trial and ΔXRec confirmed that minimal or no lung recruitment occurred during the maneuver, we considered that recruitment had no or minor impact on the X changes with increasing PEEPs that we quantify by ΔXdis. Therefore, ΔXdis reflects a reduction in compliance due to tissue distention and is related to the degree of lung mechanical stress around the operating volumes. An increased ΔXdis suggests a less compliant lung that can be easily overdistended by PEEP. Healthy lungs show almost no decrease in compliance with PEEPs within the clinical range of PEEPs.21
Interestingly ΔXdis did not correlate with GA, but did correlate with days on MV and BPD grade and, therefore, it was included in the prediction models for both days on MV and BPD development. On the contrary, despite the higher correlation of absolute X values with total days on MV in comparison with ΔXdis, they were not included in the prediction models because they add little information to the prediction model containing GA.
Limitations of the study
Even though the sample size of our population could identify parameters related to days on MV and BPD development, the prediction models need to be tested on a larger population. Our findings are applicable to this particular population of steroid and surfactant treated ELGANs and need to be tested in other populations. Also, our population was relatively homogeneous, and the high incidence of BPD requires further studies to better evaluate the sensitivity and specificity of this method in predicting BPD. However, the good prediction of the days of MV (which ranged between 1 and 155 days) suggests that the characterization of lung mechanics by FOT provides relevant independent information for characterizing the severity of the disease.
Conclusion
In mechanically ventilated ELGANs, changes in lung mechanics with PEEP on the first day of life combined with other commonly available clinical variables identify infants at risk of negative respiratory outcomes. Future studies are required to evaluate the prognostic value of this parameter in a larger population.
References
Stoll, B. J. et al. Neonatal outcomes of extremely preterm infants from the NICHD Neonatal Research Network. Pediatrics 126, 443–456 (2010).
Shah, P. S. et al. Neonatal outcomes of very low birth weight and very preterm neonates: an international comparison. J. Pediatr. 177, 144–152 (2016). e6.
Onland, W. et al. Clinical prediction models for bronchopulmonary dysplasia: a systematic review and external validation study. BMC Pediatr. 13, 207 (2013).
Wilson, A. et al. Neonatal assisted ventilation: predictors, frequency, and duration in a mature managed care organization. Pediatrics 105, 822–830 (2000).
Hintz, S. R., Bann, C. M., & AmbalavananN. Predicting time to hospital discharge for extremely preterm infants. Pediatrics 125, e146–e154 (2010).
Lui, K. et al. Early changes in respiratory compliance and resistance during the development of bronchopulmonary dysplasia in the era of surfactant therapy. Pediatr. Pulmonol. 30, 282–290 (2000).
Kavvadia, V., Greenough, A. & Dimitriou, G. Early prediction of chronic oxygen dependency by lung function test results. Pediatr. Pulmonol. 29, 19–26 (2000).
Van Lierde, S., Smith, J. & Devlieger, H. Pulmonary mechanics during respiratory distress syndrome in the prediction of outcome and differentiation of mild and severe bronchopulmonary dysplasia. Pediatr. Pulmonol. 224, 218–224 (1994).
Freezer, N. J. & Sly, P. D. Predictive value of measurements of respiratory mechanics in preterm infants with HMD. Pediatr. Pulmonol. 16, 116–123 (1993).
Choukroun, M. L. et al. Early respiratory system mechanics and the prediction of chronic lung disease in ventilated preterm neonates requiring surfactant treatment. Biol. Neonate 83, 30–35 (2003).
Snepvangers, Y. et al. Respiratory outcome in preterm ventilated infants: importance of early respiratory system resistance. Eur. J. Pediatr. 163, 378–384 (2004).
Williams, O. et al. Lung function and exhaled nitric oxide levels in infants developing chronic lung disease. Pediatr. Pulmonol. 42, 107–113 (2007).
May, C. et al. Prediction of bronchopulmonary dysplasia. Arch. Dis. Child. Fetal Neonatal Ed. 96, F410–F416 (2011).
Graff, M. A. et al. Compliance measurement in respiratory distress syndrome: the prediction of outcome. Pediatr. Pulmonol. 2, 332–336 (1986).
Simbruner, G. et al. Respiratory compliance of newborns after birth and its prognostic value for the course and outcome of respiratory disease. Respiration 43, 414–423 (1982).
Bhutani, V. K. & Abbasi, S. Relative likelihood of bronchopulmonary dysplasia based on pulmonary mechanics measured in preterm neonates during the first week of life. J. Pediatr. 120, 605–613 (1992).
Charitharth Lal, V., & Ambalavanan, N. Biomarkers, early diagnosis, and clinical predictors of BPD. Clin. Perinatol. 42, 739–754 (2015).
Lischka, A. et al. Comparison of chest radiography and static respiratory compliance in the assessment of the severity of pulmonary diseases in newborns with respiratory distress. Pediatr. Radiol. 14, 369–372 (1984).
Gappa, M. et al. Lung function tests in neonates and infants with chronic lung disease: lung and chest-wall mechanics. Pediatr. Pulmonol. 41, 291–317 (2006).
Bates, J. H. T. et al. Oscillation mechanics of the respiratory system. Compr. Physiol. 1, 1233–1272 (2011).
Dellacà, R. L. et al. Relationship between respiratory impedance and positive end-expiratory pressure in mechanically ventilated neonates. Intensive Care Med. 39, 511–519 (2013).
Vendettuoli, V. et al. Positional effects on lung mechanics of ventilated preterm infants with acute and chronic lung disease. Pediatr. Pulmonol. 50, 798–804 (2014).
Kaczka, D. W. & Dellacà, R. L. Oscillation mechanics of the respiratory system: applications to lung disease. Crit. Rev. Biomed. Eng. 39, 337–359 (2011).
Paetzel, M. Respiratory distress syndrome (grade 1–4) of the premature and newborn (IRDS). PedRad 2, 11 (2002).
Jobe, A. H. & Bancalari, E. Bronchopulmonary dysplasia. Am. J. Respir. Crit. Care Med. 163, 1723–1729 (2001).
Ryan, R. M. A new look at bronchopulmonary dysplasia classification. J. Perinatol. 26, 207–209 (2006).
Dorkin, H. L. et al. Respiratory system impedance from 4 to 40 Hz in paralyzed intubated infants with respiratory disease. J. Clin. Invest. 72, 903–910 (1983).
Schibler, A. & Frey, U. Role of lung function testing in the management of mechanically ventilated infants. Arch. Dis. Child. Fetal Neonatal Ed. 87, F7–F10 (2002).
Menard, S. Six approaches to calculating standardized logistic regression coefficients. Am. Stat. 58, 218–223 (2004).
Acknowledgements
The authors would like to thank Drs. Eva Penno, MD PhD, and Peter Pech, MD PhD, at the Department of Radiology, Uppsala University Children’s Hospital, Uppsala, for assistance in the evaluation of radiographs. The authors gratefully acknowledge MarJanna Dahl of the University of Utah for revising the manuscript. This study was supported by grants from the Her Royal Highness Crown Princess Lovisa’s Foundation for Children’s Health Care, Stockholm and the Gillbergska Foundation, Uppsala. The study sponsors had no role in the design of the study, the collection, analysis, or interpretation of data, the writing of the report or the decision to submit the manuscript for publication.
Author information
Authors and Affiliations
Contributions
C.V. contributed to the study design and to the experimental activity, carried out data processing and analysis, and drafted the manuscript. L.W. contributed to the study design, took part to the experimental activity, carried out data analysis, and contributed to drafting the manuscript. R.S. contributed to study design, data interpretation, and critically revised the manuscript. R.L.D. contributed to the study design, data interpretation, and in writing the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors have no conflicts of interest. Politecnico di Milano University, institution of C.V. and R.L.D., received research grants from Chiesi Farmaceutici SpA, Acutronic Medical Systems AG, and Philips. Politecnico di Milano also licensed patents on diagnostic application of the forced oscillation technique to Acutronic Medical Systems AG and Philips.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Veneroni, C., Wallström, L., Sindelar, R. et al. Oscillatory respiratory mechanics on the first day of life improves prediction of respiratory outcomes in extremely preterm newborns. Pediatr Res 85, 312–317 (2019). https://doi.org/10.1038/s41390-018-0133-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41390-018-0133-6
This article is cited by
-
Oscillometry for personalizing continuous distending pressure maneuvers: an observational study in extremely preterm infants
Respiratory Research (2024)
-
Combining lung ultrasound and oscillatory mechanics for assessing lung disease in very preterm infants
Pediatric Research (2024)
-
The best of both worlds: Refining respiratory phenotypes through combined non-invasive lung monitoring
Pediatric Research (2024)
-
Oscillatory mechanics trajectory in very preterm infants: a cohort study
Pediatric Research (2023)
-
Oscillatory mechanics at birth for identifying infants requiring surfactant: a prospective, observational trial
Respiratory Research (2021)