Abstract
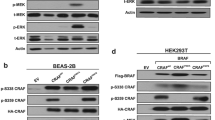
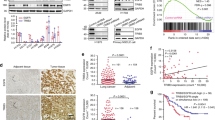
Mutational activation of the epidermal growth factor receptor (EGFR) is a major player in the pathogenesis of non-small cell lung cancer (NSCLC). NSCLC patients with constitutively active EGFR mutations eventually develop drug resistance against EGFR tyrosine-kinase inhibitors; therefore, better understandings of key components of mutant EGFR (mtEGFR) signaling are required. Here, we initially observed aberrantly high expression of protein kinase Cα (PKCα) in lung adenocarcinomas, especially those with EGFR mutations, and proceeded to examine the role of PKCα in the regulation of the signaling pathways downstream of mtEGFR. The results showed that NSCLC cell lines with constitutively active EGFR mutations tend to have very or moderately high PKCα levels. Furthermore, PKCα was constitutively activated in HCC827 and H4006 cells which have an EGFR deletion mutation in exon 19. Interestingly, mtEGFR was not required for the induction of PKCα at protein and message levels suggesting that the increased levels of PKCα are due to independent selection. On the other hand, mtEGFR activity was required for robust activation of PKCα. Loss of functions studies revealed that the NSCLC cells rely heavily on PKCα for the activation of the mTORC1 signaling pathway. Unexpectedly, the results demonstrated that PKCα was required for activation of Akt upstream of mTOR but only in cells with the mtEGFR and with the increased expression of PKCα. Functionally, inhibition of PKCα in HCC827 led to caspase-3-dependent apoptosis and a significant decrease in cell survival in response to cellular stress induced by serum starvation. In summary, the results identified important roles of PKCα in regulating mTORC1 activity in lung cancer cells, whereby a primary switching occurs from PKCα-independent to PKCα-dependent signaling in the presence of EGFR mutations. The results present PKCα as a potential synergistic target of personalized treatment for NSCLC with constitutively active mutant forms of EGFR and constitutively active PKCα.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout










Similar content being viewed by others
Change history
30 September 2019
The original HTML version of this Article was updated after publication to include the Supplementary file, as it was previously omitted.
01 October 2019
An amendment to this paper has been published and can be accessed via a link at the top of the paper.
References
Kanne JP. Screening for lung cancer: what have we learned? Am J Roentgenol. 2014;202:530–5.
Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M, et al. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer. 2015;136:E359–86.
Molina JR, Yang P, Cassivi SD, Schild SE, Adjei AA (eds). Non-small cell lung cancer: epidemiology, risk factors, treatment, and survivorship. Mayo Clin Proc. 2008;83:584–94.
Shames DS, Wistuba II. The evolving genomic classification of lung cancer. J Pathol. 2014;232:121–33.
Hollstein M, Sidransky D, Vogelstein B, Harris CC. p53 mutations in human cancers. Science. 1991;253:49–53.
Reissmann PT, Koga H, Takahashi R, Figlin RA, Holmes EC, Piantadosi S, et al. Inactivation of the retinoblastoma susceptibility gene in non-small-cell lung cancer. The Lung Cancer Study Group. Oncogene. 1993;8:1913–9.
Jin G, Kim MJ, Jeon H-S, Choi JE, Kim DS, Lee EB, et al. PTEN mutations and relationship to EGFR, ERBB2, KRAS, and TP53 mutations in non-small cell lung cancers. Lung Cancer. 2010;69:279–83.
Sordella R, Bell DW, Haber DA, Settleman J. Gefitinib-sensitizing EGFR mutations in lung cancer activate anti-apoptotic pathways. Science. 2004;305:1163–7.
Gazdar A. Activating and resistance mutations of EGFR in non-small-cell lung cancer: role in clinical response to EGFR tyrosine kinase inhibitors. Oncogene. 2009;28:S24–31.
Cooper WA, Lam DC, O'Toole SA, Minna JD. Molecular biology of lung cancer. J Thorac Dis. 2013;5:S479–90.
Antonicelli A, Cafarotti S, Indini A, Galli A, Russo A, Cesario A, et al. EGFR-targeted therapy for non-small cell lung cancer: focus on EGFR oncogenic mutation. Int J Med Sci. 2013;10:320–30.
Steins M, Thomas M.Geissler M, Recent results in cancer research Fortschritte der Krebsforschung Progres dans les recherches sur le cancer. Erlotinib. 2014;201:109–23.
Steinberg SF. Structural basis of protein kinase C isoform function. Physiol Rev. 2008;88:1341–78.
Griner EM, Kazanietz MG. Protein kinase C and other diacylglycerol effectors in cancer. Nat Rev Cancer. 2007;7:281–94.
Kong C, Zhu Y, Liu D, Yu M, Li S, Li Z, et al. Role of protein kinase C-alpha in superficial bladder carcinoma recurrence. Urology. 2005;65:1228–32.
Gupta A, Galoforo S, Berns C, Martinez A, Corry P, Guan KL, et al. Elevated levels of ERK2 in human breast carcinoma MCF‐7 cells transfected with protein kinase Cα. Cell Prolif. 1996;29:655–63.
Kim S, Han J, Lee SK, Choi M-Y, Kim J, Lee J, et al. Berberine suppresses the TPA-induced MMP-1 and MMP-9 expressions through the inhibition of PKC-α in breast cancer cells. J Surg Res. 2012;176:e21–9.
Oster H, Leitges M. Protein kinase C α but not PKCζ suppresses intestinal tumor formation in ApcMin/+ mice. Cancer Res. 2006;66:6955–63.
Jiang X-H, Tu S-P, Cui J-T, Lin MC, Xia HH, Wong WM, et al. Antisense targeting protein kinase C α and β1 inhibits gastric carcinogenesis. Cancer Res. 2004;64:5787–94.
Fan Q-W, Cheng C, Knight ZA, Haas-Kogan D, Stokoe D, James CD, et al. EGFR signals to mTOR through PKC and independently of Akt in glioma. Sci Signal. 2009;2:ra4.
Smith SD, Enge M, Bao W, Thullberg M, Costa TD, Olofsson H, et al. Protein kinase Calpha (PKCalpha) regulates p53 localization and melanoma cell survival downstream of integrin alphav in three-dimensional collagen and in vivo. J Biol Chem. 2012;287:29336–47.
Hsieh Y-H, Wu T-T, Huang C-Y, Hsieh Y-S, Hwang J-M, Liu J-Y. p38 mitogen-activated protein kinase pathway is involved in protein kinase Cα–regulated invasion in human hepatocellular carcinoma cells. Cancer Res. 2007;67:4320–7.
Singhal SS, Yadav S, Singhal J, Drake K, Awasthi YC, Awasthi S. The role of PKCα and RLIP76 in transport‐mediated doxorubicin‐resistance in lung cancer. FEBS Lett. 2005;579:4635–41.
O'Neill AK, Gallegos LL, Justilien V, Garcia EL, Leitges M, Fields AP, et al. Protein kinase Cα promotes cell migration through a PDZ-dependent interaction with its novel substrate discs large homolog 1 (DLG1). J Biol Chem. 2011;286:43559–68.
La Porta CA, Tessitore L, Comolli R. Changes in protein kinase C alpha, delta and in nuclear beta isoform expression in tumour and lung metastatic nodules induced by diethylnitrosamine in the rat. Carcinogenesis. 1997;18:715–9.
Cheng CY, Hsieh HL, Sun CC, Lin CC, Luo SF, Yang CM. IL‐1β induces urokinse‐plasminogen activator expression and cell migration through PKCα, JNK1/2, and NF‐κB in A549 cells. J Cell Physiol. 2009;219:183–93.
Wang C, Wang X, Liang H, Wang T, Yan X, Cao M, et al. miR-203 inhibits cell proliferation and migration of lung cancer cells by targeting PKCα. PLoS ONE. 2013;8:e73985.
Abera MB, Kazanietz MG. Protein kinase Cα mediates erlotinib resistance in lung cancer cells. Mol Pharm. 2015;87:832–41.
Lahn M, Su C, Li S, Chedid M, Hanna KR, Graff JR, et al. Expression levels of protein kinase C-α in non–small-cell lung cancer. Clin Lung Cancer. 2004;6:184–9.
Barretina J, Caponigro G, Stransky N, Venkatesan K, Margolin AA, Kim S, et al. The Cancer Cell Line Encyclopedia enables predictive modelling of anticancer drug sensitivity. Nature. 2012;483:603–7.
El Osta M, Liu M, Adada M, Senkal CE, Idkowiak-Baldys J, Obeid LM, et al. Sustained PKCβII activity confers oncogenic properties in a phospholipase D-and mTOR-dependent manner. FASEB J. 2014;28:495–505.
Vincent E, Elder D, Thomas E, Phillips L, Morgan C, Pawade J, et al. Akt phosphorylation on Thr308 but not on Ser473 correlates with Akt protein kinase activity in human non-small cell lung cancer. Br J Cancer. 2011;104:1755–61.
Vansteenkiste J, Ramlau R, Von Pawel J, San Antonio B, Eschbach C, Szczesna A, et al. A phase II randomized study of cisplatin-pemetrexed plus either enzastaurin or placebo in chemonaive patients with advanced non-small cell lung cancer. Oncology. 2012;82:25–9.
Willey CD, Xiao D, Tu T, Kim KW, Moretti L, Niermann KJ, et al. Enzastaurin (LY317615), a protein kinase C beta selective inhibitor, enhances antiangiogenic effect of radiation. Int J Radiat Oncol* Biol* Phys. 2010;77:1518–26.
Caino MC, Lopez-Haber C, Kim J, Mochly-Rosen D, Kazanietz MG. Proteins kinase Cϵ is required for non-small cell lung carcinoma growth and regulates the expression of apoptotic genes. Oncogene. 2012;31:2593–2600.
Hill K, Erdogan E, Khoor A, Walsh M, Leitges M, Murray NR, et al. Protein kinase Cα suppresses Kras-mediated lung tumor formation through activation of a p38 MAPK-TGFβ signaling axis. Oncogene. 2014;33:2134–44.
Antal CE, Hudson AM, Kang E, Zanca C, Wirth C, Stephenson NL, et al. Cancer-associated protein kinase C mutations reveal kinase’s role as tumor suppressor. Cell. 2015;160:489–502.
Alessi DR, James SR, Downes CP, Holmes AB, Gaffney PR, Reese CB, et al. Characterization of a 3-phosphoinositide-dependent protein kinase which phosphorylates and activates protein kinase Bα. Curr Biol. 1997;7:261–9.
Vanhaesebroeck B, Alessi DR. The PI3K–PDK1 connection: more than just a road to PKB. Biochem J. 2000;346:561–76.
Sarbassov DD, Guertin DA, Ali SM, Sabatini DM. Phosphorylation and regulation of Akt/PKB by the rictor-mTOR complex. Science. 2005;307:1098–101.
Hanada M, Feng J, Hemmings BA. Structure, regulation and function of PKB/AKT—a major therapeutic target. Biochim Biophys Acta (BBA)-Proteins Proteom. 2004;1697:3–16.
Tsurutani J, Fukuoka J, Tsurutani H, Shih JH, Hewitt SM, Travis WD, et al. Evaluation of two phosphorylation sites improves the prognostic significance of Akt activation in non-small-cell lung cancer tumors. J Clin Oncol. 2006;24:306–14.
Gallay N, Dos Santos C, Cuzin L, Bousquet M, Gouy VS, Chaussade C, et al. The level of AKT phosphorylation on threonine 308 but not on serine 473 is associated with high-risk cytogenetics and predicts poor overall survival in acute myeloid leukaemia. Leukemia. 2009;23:1029–38.
Rodrigues GA, Falasca M, Zhang Z, Ong SH, Schlessinger J. A novel positive feedback loop mediated by the docking protein Gab1 and phosphatidylinositol 3-kinase in epidermal growth factor receptor signaling. Mol Cell Biol. 2000;20:1448–59.
Mattoon DR, Lamothe B, Lax I, Schlessinger J. The docking protein Gab1 is the primary mediator of EGF-stimulated activation of the PI-3K/Akt cell survival pathway. BMC Biol. 2004;2:24.
Song X, Fan P-D, Bantikassegn A, Guha U, Threadgill DW, Varmus H, et al. ERBB3-independent activation of the PI3K pathway in EGFR-mutant lung adenocarcinomas. Cancer Res. 2015;75:1035–45.
Feng X, Hannun YA. An essential role for autophosphorylation in the dissociation of activated protein kinase C from the plasma membrane. J Biol Chem. 1998;273:26870–4.
Idkowiak-Baldys J, Becker KP, Kitatani K, Hannun YA. Dynamic sequestration of the recycling compartment by classical protein kinase C. J Biol Chem. 2006;281:22321–31.
Liu M, Idkowiak-Baldys J, Roddy PL, Baldys A, Raymond J, Clarke CJ, et al. Sustained activation of protein kinase C induces delayed phosphorylation and regulates the fate of epidermal growth factor receptor. PLoS ONE. 2013;8:e80721.
Sanjana NE, Shalem O, Zhang F. Improved vectors and genome-wide libraries for CRISPR screening. Nat Methods. 2014;11:783.
Pulkoski-Gross MJ, Jenkins ML, Truman J-P, Salama MF, Clarke CJ, Burke JE, et al. An intrinsic lipid-binding interface controls sphingosine kinase 1 function. J Lipid Res. 2018;59:462–74.
Idkowiak-Baldys J, Baldys A, Raymond JR, Hannun YA. Sustained receptor stimulation leads to sequestration of recycling endosomes in a classical protein kinase C- and phospholipase D-dependent manner. J Biol Chem. 2009;284:22322–31.
Acknowledgements
This work was supported by NCI grant CA97132.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Salama, M.F., Liu, M., Clarke, C.J. et al. PKCα is required for Akt-mTORC1 activation in non-small cell lung carcinoma (NSCLC) with EGFR mutation. Oncogene 38, 7311–7328 (2019). https://doi.org/10.1038/s41388-019-0950-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41388-019-0950-z
This article is cited by
-
A cuproptosis score model and prognostic score model can evaluate clinical characteristics and immune microenvironment in NSCLC
Cancer Cell International (2024)