Abstract
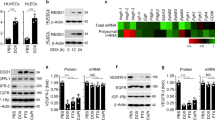
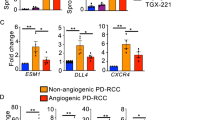
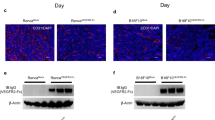
Inhibitors of Vascular Endothelial Growth Factor target both tumor vasculature and cancer cells that have hijacked VEGF Receptors (VEGFRs) signaling for tumor growth-promoting activities. It is important to get precise insight in the specificity of cell responses to these antiangiogenic drugs to maximize their efficiency and minimize off-target systemic toxicity. Here we report that Axitinib, an inhibitor of VEGFRs currently in use as a second line treatment for advanced renal cell carcinoma, promotes senescence of human endothelial cells in vitro. A one-hour pulse of Axitinib is sufficient for triggering cell senescence. Mechanistically, this requires oxidative stress-dependent activation of the Ataxia Telangiectasia Mutated (ATM) kinase. Axitinib-mediated senescence promoting action is prevented by short-term treatment with antioxidants or ATM inhibitors, which conversely fail to prevent senescence induced by the DNA-damaging drug doxorubicin. Coherently, induction of oxidative stress-related genes distinguishes the response of endothelial cells to Axitinib from that to doxorubicin. Importantly, an Axitinib pulse causes cell senescence in glioblastoma cells. However, neither antioxidants nor ATM inhibitors can reverse this phenotype. Thus, antioxidants may selectively protect endothelial cells from Axitinib by decreasing systemic toxicity and maintaining a functional vascularization necessary for efficient delivery of chemotherapeutic drugs within the tumor mass.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Hayflick L, Moorhead PS. The serial cultivation of human diploid cell strains. Exp Cell Res. 1961;25:585–621.
Pawlikowski JS, Adams PD, Nelson DM. Senescence at a glance. J Cell Sci. 2013;126:4061–7.
He S, Sharpless NE. Senescence in Health and Disease. Cell. 2017;169:1000–11.
Ferrara N. Vascular endothelial growth factor as a target for anticancer therapy. Oncologist. 2004;9(Suppl 1):2–10.
Welti J, Loges S, Dimmeler S, Carmeliet P. Recent molecular discoveries in angiogenesis and antiangiogenic therapies in cancer. J Clin Invest. 2013;123:3190–3200.
Jain RK. Normalization of tumor vasculature: an emerging concept in antiangiogenic therapy. Science. 2005;307:58–62.
Wilmes LJ, Pallavicini MG, Fleming LM, Gibbs J, Wang D, Li KL, et al. AG-013736, a novel inhibitor of VEGF receptor tyrosine kinases, inhibits breast cancer growth and decreases vascular permeability as detected by dynamic contrast-enhanced magnetic resonance imaging. Magn Reson Imaging. 2007;25:319–27.
Rugo HS, Herbst RS, Liu G, Park JW, Kies MS, Steinfeldt HM, et al. Phase I trial of the oral antiangiogenesis agent AG-013736 in patients with advanced solid tumors: Pharmacokinetic and clinical results. J Clin Oncol. 2005;23:5474–83.
Klaeger S, Heinzlmeir S, Wilhelm M, Polzer H, Vick B, Koenig PA, et al. The target landscape of clinical kinase drugs. Science. 2017;358:6367.
Bellesoeur A, Alexandre J. Axitinib in the treatment of renal cell carcinoma: design, development, and place in therapy. Drug Des Devel Ther. 2017;11:2801–11.
Hu-Lowe DD, Zou HY, Grazzini ML, Hallin ME, Wickman GR, Amundson K, et al. Nonclinical antiangiogenesis and antitumor activities of axitinib (AG-013736), an oral, potent, and selective inhibitor of vascular endothelial growth factor receptor tyrosine kinases 1, 2, 3. Clin Cancer Res. 2008;14:7272–83.
Morelli MB, Amantini C, Santoni M, Soriani A, Nabissi M, Cardinali C, et al. Axitinib induces DNA damage response leading to senescence, mitotic catastrophe, and increased NK cell recognition in human renal carcinoma cells. Oncotarget. 2015;6:36245–59.
Morelli MB, Amantini C, Nabissi M, Cardinali C, Santoni M, Bernardini G, et al. Axitinib induces senescence-associated cell death and necrosis in glioma cell lines. The proteasome inhibitor, bortezomib, potentiates axitinib-induced cytotoxicity in a p21(Waf/Cip1) dependent manner. Oncotarget. 2016;8:3380–95.
Elmore LW, Rehder CW, Di X, McChesney PA, Jackson-Cook CK, Gewirtz DA, et al. Adriamycin-induced senescence in breast tumor cells involves functional p53 and telomere dysfunction. J Biol Chem. 2002;277:35509–15.
Song YS, Lee BY, Hwang ES. Dinstinct ROS and biochemical profiles in cells undergoing DNA damage-induced senescence and apoptosis. Mech Ageing Dev. 2005;126:580–90.
Przybylska D, Janiszewska D, Goździk A, Bielak-Zmijewska A, Sunderland P, Sikora E, et al. NOX4 downregulation leads to senescence of human vascular smooth muscle cells. Oncotarget. 2016;7:66429–43.
Dimri GP, Lee X, Basile G, Acosta M, Scott G, Roskelley C, et al. A biomarker that identifies senescent human cells in culture and in aging skin in vivo. Proc Natl Acad Sci USA. 1995;92:9363–7.
Chang MW-F, Grillari J, Mayrhofer C, Fortschegger K, Allmaier G, Marzban G, et al. Comparison of early passage, senescent and hTERT immortalized endothelial cells. Exp Cell Res. 2005;309:121–36.
White RR, Vijg J. Do DNA double-strand breaks drive aging? Mol Cell. 2016;63:729–38.
Davalli P, Mitic T, Caporali A, Lauriola A, D’Arca D. ROS, cell senescence, and novel molecular mechanisms in aging and age-related diseases. Oxid Med Cell Longev. 2016;2016:3565127
Passos JF, Nelson G, Wang C, Richter T, Simillion C, Proctor CJ, et al. Feedback between p21 and reactive oxygen production is necessary for cell senescence. Mol Syst Biol. 2010;6:347.
Ewald JA, Desotelle JA, Wilding G, Jarrard DF. Therapy-induced senescence in cancer. J Natl Cancer Inst. 2010;102:1536–46.
Nair RR, Bagheri M, Saini DK. Temporally distinct roles of ATM and ROS in genotoxic-stress-dependent induction and maintenance of cellular senescence. J Cell Sci. 2015;128:342–53.
Hickson I, Zhao Y, Richardson CJ, Green SJ, Martin NMB, Orr AI, et al. Identification and characterization of a novel and specific inhibitor of the ataxia-telangiectasia mutated kinase ATM. Cancer Res. 2004;64:9152–9.
Menendez D, Inga A, Snipe J, Krysiak O, Schönfelder G, Resnick MA. A single-nucleotide polymorphism in a half-binding site creates p53 and estrogen receptor control of vascular endothelial growth factor receptor 1. Mol Cell Biol. 2007;27:2590–2600.
Freund A, Laberge R-M, Demaria M, Campisi J. Lamin B1 loss is a senescence-associated biomarker. Mol Biol Cell. 2012;23:2066–75.
Lenain C, Gusyatiner O, Douma S, van den Broek B, Peeper DS. Autophagy-mediated degradation of nuclear envelope proteins during oncogene-induced senescence. Carcinogenesis. 2015;36:1263–74.
Bonello-Palot N, Simoncini S, Robert S, Bourgeois P, Sabatier F, Levy N, et al. Prelamin A accumulation in endothelial cells induces premature senescence and functional impairment. Atherosclerosis. 2014;237:45–52.
Coppé J-P, Desprez P-Y, Krtolica A, Campisi J. The senescence-associated secretory phenotype: the dark side of tumor suppression. Annu Rev Pathol Mech Dis. 2010;5:99–118.
Hovinga KE, Stalpers LJa, van Bree C, Donker M, Verhoeff JJC, Rodermond HM, et al. Radiation-enhanced vascular endothelial growth factor (VEGF) secretion in glioblastoma multiforme cell lines—a clue to radioresistance? J Neurooncol. 2005;74:99–103.
Liang Y, Li XY, Rebar EJ, Li P, Zhou Y, Chen B, et al. Activation of vascular endothelial growth factor A transcription in tumorigenic glioblastoma cell lines by an enhancer with cell type-specific DNase I accessibility. J Biol Chem. 2002;277:20087–94.
Perrigue PM, Silva ME, Warden CD, Feng NL, Reid MA, Mota DJ, et al. The histone demethylase jumonji coordinates cellular senescence including secretion of neural stem cell–attracting cytokines. Mol Cancer Res. 2015;13:636–50.
Coppé J-P, Patil CK, Rodier F, Sun Y, Muñoz DP, Goldstein J. et al. Senescence-associated secretory phenotypes reveal cell-nonautonomous functions of oncogenic RAS and the p53 tumor suppressor. PLoS Biol. 2008;6:e301
Folkman J. Tumor angiogenesis: therapeutic implications. N Engl J Med. 1971;285:1182–6.
Goel HL, Mercurio AM. VEGF targets the tumour cell. Nat Rev Cancer. 2013;13:871–82.
Kang HT, Park JT, Choi K, Kim Y, Choi HJC, Jung CW, et al. Chemical screening identifies ATM as a target for alleviating senescence. Nat Chem Biol. 2017;13:616–23.
Guo Z, Kozlov S, Lavin MF, Person MD, Paull TT. ATM activation by oxidative stress. Science. 2010;330:517–21.
Demaria M, O’Leary MN, Chang J, Shao L, Liu S, Alimirah F, et al. Cellular senescence promotes adverse effects of chemotherapy and cancer relapse. Cancer Discov. 2017;7:165–76.
Barrett T, Wilhite SE, Ledoux P, Evangelista C, Kim IF, Tomashevsky M, et al. NCBI GEO: archive for functional genomics data sets—update. Nucleic Acids Res. 2013;41:991–5.
Acknowledgements
The study was supported by research grants from: MIUR-CNR Flagship Invecchiamento #DSB.AD009.001.004 to AL and MLF; Associazione Italiana per la Ricerca sul Cancro-AIRC IG2016-n.19069, MIUR-JPI HDHL NUTRICOG MiTyrAge, PRIN_2015LZE9944_005 and RF-2016-02362022 to DB; by RF-2016-02363460 to VS. VS has also been supported by AIRC IG2016-n.19069. We thank Dr Silvia Soddu and Prof Carlo Gaetano for their suggestions on p53 protein detection and ROS measurement, respectively.
Author information
Authors and Affiliations
Contributions
MPM, GR, MP, VS, SP and AR conducted the experiments; FF performed bioinformatics analyses; RP, DB and AL analyzed and discussed the data; AF gave conceptual advice, contributed to experimental design, critical data analysis and manuscript revision; AL and MLF conceived the study and drafted the manuscript. All authors read and approved the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mongiardi, M.P., Radice, G., Piras, M. et al. Axitinib exposure triggers endothelial cells senescence through ROS accumulation and ATM activation. Oncogene 38, 5413–5424 (2019). https://doi.org/10.1038/s41388-019-0798-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41388-019-0798-2
This article is cited by
-
Pharmacological targeting of CBP/p300 drives a redox/autophagy axis leading to senescence-induced growth arrest in non-small cell lung cancer cells
Cancer Gene Therapy (2023)
-
Mechanisms and consequences of endothelial cell senescence
Nature Reviews Cardiology (2023)
-
Tackling cellular senescence by targeting miRNAs
Biogerontology (2022)
-
Gene expression profiling of hypoxic response in different models of senescent endothelial cells
Aging Clinical and Experimental Research (2021)
-
Wogonin induces cellular senescence in breast cancer via suppressing TXNRD2 expression
Archives of Toxicology (2020)