Abstract
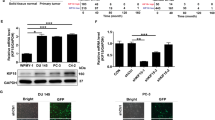
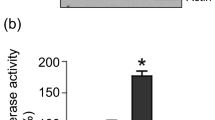
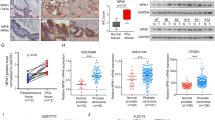
Prostate cancer (PCa) initiation and progression requires activation of numerous oncogenic signaling pathways. Nuclear-cytoplasmic transport of oncogenic factors is mediated by Karyopherin proteins during cell transformation. However, the role of nuclear transporter proteins in PCa progression has not been well defined. Here, we report that the KPNB1, a key member of Karyopherin beta subunits, is highly expressed in advanced prostate cancers. Further study showed that targeting KPNB1 suppressed the proliferation of prostate cancer cells. The knockdown of KPNB1 reduced nuclear translocation of c-Myc, the expression of downstream cell cycle modulators, and phosphorylation of regulator of chromatin condensation 1 (RCC1), a key protein for spindle assembly during mitosis. Meanwhile, CHIP assay demonstrated the binding of c-Myc to KPNB1 promoter region, which indicated a positive feedback regulation of KPNB1 expression mediated by the c-Myc. In addition, NF-κB subunit p50 translocation to nuclei was blocked by KPNB1 inhibition, which led to an increase in apoptosis and a decrease in tumor sphere formation of PCa cells. Furthermore, subcutaneous xenograft tumor models with a stable knockdown of KPNB1 in C42B PCa cells validated that the inhibition of KPNB1 could suppress the growth of prostate tumor in vivo. Moreover, the intravenously administration of importazole, a specific inhibitor for KPNB1, effectively reduced PCa tumor size and weight in mice inoculated with PC3 PCa cells. In summary, our data established the functional link between KPNB1 and PCa prone c-Myc, NF-kB, and cell cycle modulators. More importantly, inhibition of KPNB1 could be a new therapeutic target for PCa treatment.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Ghildyal R, Ho A, Wagstaff KM, Dias MM, Barton CL, Jans P, et al. Nuclear import of the respiratory syncytial virus matrix protein is mediated by importin beta1 independent of importin alpha. Biochemistry. 2005;44:12887–95.
van der Watt PJ, Ngarande E, Leaner VD. Overexpression of Kpnbeta1 and Kpnalpha2 importin proteins in cancer derives from deregulated E2F activity. PLoS One. 2011;6:e27723.
Yang J, Lu C, Wei J, Guo Y, Liu W, Luo L, et al. Inhibition of KPNA4 attenuates prostate cancer metastasis. Oncogene. 2017;36:2868–78.
Huang L, Wang HY, Li JD, Wang JH, Zhou Y, Luo RZ et al. KPNA2 promotes cell proliferation and tumorigenicity in epithelial ovarian carcinoma through upregulation of c-Myc and downregulation of FOXO3a. Cell Death Dis. 2013;4:e745.
Huang L, Zhou Y, Cao XP, Lin JX, Zhang L, Huang ST, et al. KPNA2 promotes migration and invasion in epithelial ovarian cancer cells by inducing epithelial-mesenchymal transition via Akt/GSK-3beta/Snail activation. J Cancer. 2018;9:157–65.
Aggarwal A, Agrawal DK. Importins and exportins regulating allergic immune responses. Mediat Inflamm. 2014, Volume 2014, Article ID 476357.
Fagerlund R, Kinnunen L, Kohler M, Julkunen I, Melen K. NF-{kappa}B is transported into the nucleus by importin {alpha}3 and importin {alpha}4. J Biol Chem. 2005;280:15942–51.
Cai Y, Shen Y, Gao L, Chen M, Xiao M, Huang Z, et al. Karyopherin Alpha 2 promotes the inflammatory response in rat pancreatic acinar cells via facilitating NF-kappaB activation. Dig Dis Sci. 2016;61:747–57.
Liang P, Zhang H, Wang G, Li S, Cong S, Luo Y, et al. KPNB1, XPO7 and IPO8 mediate the translocation ofNF-kappaB/p65 into the nucleus. Traffic. 2013;14:1132–43.
Yan W, Li R, He J, Du J, Hou J. Importin beta1 mediates nuclear factor-kappaB signal transduction into the nuclei of myeloma cells and affects their proliferation and apoptosis. Cell Signal. 2015;27:851–9.
Wang S, Zhao Y, Xia N, Zhang W, Tang Z, Wang C, et al. KPNbeta1 promotes palmitate-induced insulin resistance via NF-kappaB signaling in hepatocytes. J Physiol Biochem. 2015;71:763–72.
Ha S, Jeong J, Oh J, Rhee S, Ham SW. A small organic molecule blocks EGFR transport into the nucleus by the nonclassical pathway resulting in repression of cancer invasion. Chembiochem. 2018;19:131–35.
Angus L, van der Watt PJ, Leaner VD. Inhibition of the nuclear transporter, Kpnbeta1, results in prolonged mitotic arrest and activation of the intrinsic apoptotic pathway in cervical cancer cells. Carcinogenesis. 2014;35:1121–31.
Martens-de Kemp SR, Nagel R, Stigter-van Walsum M, van der Meulen IH, van Beusechem VW, Braakhuis BJ, et al. Functional genetic screens identify genes essential for tumor cell survival in head and neck and lung cancer. Clin Cancer Res. 2013;19:1994–2003.
Kuusisto HV, Jans DA. Hyper-dependence of breast cancer cell types on the nuclear transporter Importin beta1. Biochim Biophys Acta. 2015;1853:1870–8.
Kodama M, Kodama T, Newberg JY, Katayama H, Kobayashi M, Hanash SM, et al. In vivo loss-of-function screens identify KPNB1 as a new druggable oncogene in epithelial ovarian cancer. Proc Natl Acad Sci USA. 2017;114:E7301–E7310.
Soderholm JF, Bird SL, Kalab P, Sampathkumar Y, Hasegawa K, Uehara-Bingen M, et al. Importazole, a small molecule inhibitor of the transport receptor importin-beta. ACS Chem Biol. 2011;6:700–8.
Akinyeke T, Matsumura S, Wang X, Wu Y, Schalfer ED, Saxena A, et al. Metformin targets c-MYC oncogene to prevent prostate cancer. Carcinogenesis. 2013;34:2823–32.
Koch HB, Zhang R, Verdoodt B, Bailey A, Zhang CD, Yates JR 3rd, et al. Large-scale identification of c-MYC-associated proteins using a combined TAP/MudPIT approach. Cell Cycle. 2007;6:205–17.
Qin ZH, Chen RW, Wang Y, Nakai M, Chuang DM, Chase TN. Nuclear factor kappaB nuclear translocation upregulates c-Myc and p53 expression during NMDA receptor-mediated apoptosis in rat striatum. J Neurosci. 1999;19:4023–33.
Ellwood-Yen K, Graeber TG, Wongvipat J, Iruela-Arispe ML, Zhang J, Matusik R, et al. Myc-driven murine prostate cancer shares molecular features with human prostate tumors. Cancer Cell. 2003;4:223–38.
Karin M, Cao Y, Greten FR, Li ZW. NF-kappaB in cancer: from innocent bystander to major culprit. Nat Rev Cancer. 2002;2:301–10.
Laurila E, Vuorinen E, Savinainen K, Rauhala H, Kallioniemi A. KPNA7, a nuclear transport receptor, promotes malignant properties of pancreatic cancer cells in vitro. Exp Cell Res. 2014;322:159–67.
Winkler J, Ori A, Holzer K, Sticht C, Dauch D, Eiteneuer EM, et al. Prosurvival function of the cellular apoptosis susceptibility/importin-alpha1 transport cycle is repressed by p53 in liver cancer. Hepatology. 2014;60:884–95.
Yang LL, Hu BY, Zhang YX, Qiang SL, Cai J, Huang W et al. Suppression of the nuclear transporter-KPN beta 1 expression inhibits tumor proliferation in hepatocellular carcinoma. Med Oncol. 2015;32:128.
Alshareeda AT, Negm OH, Green AR, Nolan CC, Tighe P, Albarakati N, et al. KPNA2 is a nuclear export protein that contributes to aberrant localisation of key proteins and poor prognosis of breast cancer. Brit J Cancer. 2015;112:1929–37.
Altan B, Yokobori T, Mochiki E, Ohno T, Ogata K, Ogawa A, et al. Nuclear karyopherin-2 expression in primary lesions and metastatic lymph nodes was associated with poor prognosis and progression in gastric cancer. Carcinogenesis. 2013;34:2314–21.
Wang HJ, Tao T, Yan W, Feng Y, Wang YZ, Cai JQ et al. Upregulation of miR-181s reverses mesenchymal transition by targeting KPNA4 in glioblastoma. Sci Rep. 2015;5:13072.
Vuorinen EM, Rajala NK, Rauhala HE, Nurminen AT, Hytonen VP, Kallioniemi A. Search for KPNA7 cargo proteins in human cells reveals MVP and ZNF414 as novel regulators of cancer cell growth. Bba-Mol Basis Dis. 2017;1863:211–9.
Shen SH, Gui TT, Ma CC. Identification of molecular biomarkers for pancreatic cancer with mRMR shortest path method. Oncotarget. 2017;8:41432–9.
Stelma T, Chi A, van der Watt PJ, Verrico A, Lavia P, Leaner VD. Targeting nuclear transporters in cancer: diagnostic, prognostic and therapeutic potential. IUBMB Life. 2016;68:268–80.
Mahipal A, Malafa M. Importins and exportins as therapeutic targets in cancer. Pharmacol Ther. 2016;164:135–43.
Lu T, Bao Z, Wang Y, Yang L, Lu B, Yan K, et al. Karyopherinbeta1 regulates proliferation of human glioma cells via Wnt/beta-catenin pathway. Biochem Biophys Res Commun. 2016;478:1189–97.
Zhang P, Garnett J, Creighton CJ, Al Sannaa GA, Igram DR, Lazar A, et al. EZH2-miR-30d-KPNB1 pathway regulates malignant peripheral nerve sheath tumour cell survival and tumourigenesis. J Pathol. 2014;232:308–18.
Schmitz MH, Held M, Janssens V, Hutchins JR, Hudecz O, Ivanova E, et al. Live-cell imaging RNAi screen identifies PP2A-B55alpha and importin-beta1 as key mitotic exit regulators in human cells. Nat Cell Biol. 2010;12:886–93.
Chen T, Muratore TL, Schaner-Tooley CE, Shabanowitz J, Hunt DF, Macara IG. N-terminal alpha-methylation of RCC1 is necessary for stable chromatin association and normal mitosis. Nat Cell Biol. 2007;9:596–U203.
Moore WJ, Zhang CM, Clarke PR. Targeting of RCC1 to chromosomes is required for proper mitotic spindle assembly in human cells. Curr Biol. 2002;12:1442–7.
Wang J, Wan XY, Gao YF, Zhong MT, Sha L, Liu B, et al. Latcripin-13 domain induces apoptosis and cell cycle arrest at the G1 phase in human lung carcinoma A549 cells. Oncol Rep. 2016;36:441–7.
Hood FE, Clarke PR. RCC1 isoforms differ in their affinity for chromatin, molecular interactions and regulation by phosphorylation. J Cell Sci. 2007;120:3436–45.
Hutchins JR, Moore WJ, Hood FE, Wilson JS, Andrews PD, Swedlow JR, et al. Phosphorylation regulates the dynamic interaction of RCC1 with chromosomes during mitosis. Curr Biol. 2004;14:1099–104.
Gurel B, Iwata T, Koh CM, Jenkins RB, Lan FS, Van Dang C, et al. Nuclear MYC protein overexpression is an early alteration in human prostate carcinogenesis. Mod Pathol. 2008;21:1156–67.
Koh CM, Bieberich CJ, Dang CV, Nelson WG, Yegnasubramanian S, De Marzo AM. MYC and prostate cancer. Genes Cancer. 2010;1:617–28.
Bretones G, Delgado MD, Leon J. Myc and cell cycle control. Biochim Biophys Acta. 2015;1849:506–16.
Nakanishi C, Toi M. Nuclear factor-kappa B inhibitors as sensitizers to anticancer drugs. Nat Rev Cancer. 2005;5:297–309.
Gagne B, Tremblay N, Park AY, Baril M, Lamarre D. Importin beta 1 targeting by hepatitis C virus NS3/4A protein restricts IRF3 and NF-kappa B signaling of IFNB1 antiviral response. Traffic. 2017;18:362–77.
Lundberg L, Pinkham C, Baer A, Amaya M, Narayanan A, Wagstaff KM, et al. Nuclear import and export inhibitors alter capsid protein distribution in mammalian cells and reduce Venezuelan Equine Encephalitis Virus replication. Antivir Res. 2013;100:662–72.
Lo HW, Ali-Seyed M, Wu Y, Bartholomeusz G, Hsu SC, Hung MC. Nuclear-cytoplasmic transport of EGFR involves receptor endocytosis, importin beta1 and CRM1. J Cell Biochem. 2006;98:1570–83.
van der Watt PJ, Chi A, Stelma T, Stowell C, Strydom E, Carden S, et al. Targeting the nuclear import receptor Kpnbeta1 as an anticancer therapeutic. Mol Cancer Ther. 2016;15:560–73.
Zhang PY, Yang XB, Ma XY, Ingram DR, Lazar AJ, Torres KE et al. Antitumor effects of pharmacological EZH2 inhibition on malignant peripheral nerve sheath tumor through the miR-30a and KPNB1 pathway. Mol Cancer 2015;14:1.
Hadjebi O, Casas-Terradellas E, Garcia-Gonzalo FR, Rosa JL. The RCC1 superfamily: from genes, to function, to disease. Biochim Biophys Acta. 2008;1783:1467–79.
Thalmann GN, Anezinis PE, Chang SM, Zhau HE, Kim EE, Hopwood VL, et al. Androgen-independent cancer progression and bone metastasis in the LNCaP model of human prostate cancer. Cancer Res. 1994;54:2577–81.
Wu HC, Hsieh JT, Gleave ME, Brown NM, Pathak S, Chung LW. Derivation of androgen-independent human LNCaP prostatic cancer cell sublines: role of bone stromal cells. Int J Cancer. 1994;57:406–12.
Watson PA, Ellwood-Yen K, King JC, Wongvipat J, Lebeau MM, Sawyers CL. Context-dependent hormone-refractory progression revealed through characterization of a novel murine prostate cancer cell line. Cancer Res. 2005;65:11565–71.
Elgogary A, Xu Q, Poore B, Alt J, Zimmermann SC, Zhao L, et al. Combination therapy with BPTES nanoparticles and metformin targets the metabolic heterogeneity of pancreatic cancer. Proc Natl Acad Sci USA. 2016;113:E5328–5336.
Acknowledgements
This study was partly supported by NYU CSCB Pilot Study Award to JY, National Institutes of Health grants R01CA180277, R01DE025992 and R01DE027074 to XL. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author contributions
JY and YG conducted most of the experiments, data acquisition, and manuscript preparation. LL, RZ, YZ, and YW conducted some of the experiments, CL and WY conducted bioinformatics analyses, CJW, CHC, and SO did blind analysis of the IHC images. XL contributes to the conceptual design and manuscript revision.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Yang, J., Guo, Y., Lu, C. et al. Inhibition of Karyopherin beta 1 suppresses prostate cancer growth. Oncogene 38, 4700–4714 (2019). https://doi.org/10.1038/s41388-019-0745-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41388-019-0745-2
This article is cited by
-
MicroRNA-130a-3p impedes the progression of papillary thyroid carcinoma through downregulation of KPNB1 by targeting PSME3
Endocrine (2023)
-
Mannose: a potential saccharide candidate in disease management
Medicinal Chemistry Research (2023)
-
Tumor-promoting properties of karyopherin β1 in melanoma by stabilizing Ras-GTPase-activating protein SH3 domain-binding protein 1
Cancer Gene Therapy (2022)
-
A mass spectrometry-based approach for the identification of Kpnβ1 binding partners in cancer cells
Scientific Reports (2022)
-
Inhibition of Kpnβ1 mediated nuclear import enhances cisplatin chemosensitivity in cervical cancer
BMC Cancer (2021)