Abstract
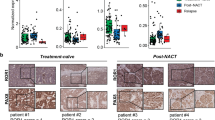
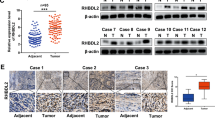
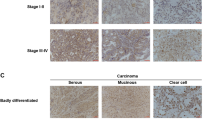
Ovarian carcinoma is the most lethal type of gynecologic malignancies. Alterations of Notch pathway are prevalent in ovarian carcinogenesis. This study investigated the expression profile and function of delta-like 1 homolog (DLK1), a non-canonical Notch ligand, during ovarian carcinogenesis. Tissue microarray (TMA) consisting of surgically resected samples from 221 patients with ovarian carcinoma was constructed for DLK1 expression. DLK1 overexpression or knockdown was achieved by adenovirus gene delivery to evaluate the effect of DLK1 on the oncogenic behaviors in ovarian cancer cells and in xenografted tumors. TMA analysis revealed that elevated DLK1 expression was correlated with stages, lymph node metastasis and E-cadherin downregulation. Despite no influence on survival among ovarian carcinoma patients, DLK1 overexpression was specially associated with overall survival and progression free survival in high-grade serous carcinoma (HGSC) patients, constituting an independent prognostic factor for these patients. By adenovirus gene delivery, it was found modulation of cellular DLK1 level regulated the tumorigenic behaviors and epithelial-mesenchymal transition (EMT) in vitro and in vivo. Immunohistochemical analysis further showed that DLK1 overexpression resulted in escalated proliferation, angiogenesis, EMT and Notch activities. Application of recombinant DLK1 extracellular domain (rDLK1-EC) recapitulated the tumorigenic behaviors of DLK1 in ovarian cancer cells. By using neutralizing antibody or pharmaceutical inhibitor, blockade of Notch signaling attenuated the tumorigenic behaviors evoked by DLK1 overexpression. The present study indicates that DLK1 overexpression participates in ovarian carcinogenesis through Notch activation and EMT induction. Moreover, DLK1 may constitute a novel diagnostic biomarker and therapeutic target for HGSC.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2017. CA Cancer J Clin. 2017;67:7–30.
Kurman RJ, Shih IeM. The dualistic model of ovarian carcinogenesis: revisited, revised, and expanded. Am J Pathol. 2016;186:733–47.
Kurman RJ, Shih IeM. The origin and pathogenesis of epithelial ovarian cancer: a proposed unifying theory. Am J Surg Pathol. 2010;34:433–43.
Shih IeM, Wang TL. Notch signaling, gamma-secretase inhibitors, and cancer therapy. Cancer Res. 2007;67:1879–82.
Bell D, Berchuck A, Birrer M, Chien J, Cramer D, Dao F. et al. Integrated genomic analyses of ovarian carcinoma. Nature. 2011;474:609–15.
Choi JH, Park JT, Davidson B, Morin PJ, Shih Ie M, Wang TL. Jagged-1 and Notch3 juxtacrine loop regulates ovarian tumor growth and adhesion. Cancer Res. 2008;68:5716–23.
Chen X, Stoeck A, Lee SJ, Shih Ie M, Wang MM, Wang TL. Jagged1 expression regulated by Notch3 and Wnt/beta-catenin signaling pathways in ovarian cancer. Oncotarget. 2010;1:210–8.
Zhang L, Volinia S, Bonome T, Calin GA, Greshock J, Yang N, et al. Genomic and epigenetic alterations deregulate microRNA expression in human epithelial ovarian cancer. Proc Natl Acad Sci Usa. 2008;105:7004–9.
Nueda ML, Garcia-Ramirez JJ, Laborda J, Baladron V. dlk1 specifically interacts with insulin-like growth factor binding protein 1 to modulate adipogenesis of 3T3-L1 cells. J Mol Biol. 2008;379:428–42.
Abdallah BM, Boissy P, Tan Q, Dahlgaard J, Traustadottir GA, Kupisiewicz K, et al. dlk1/FA1 regulates the function of human bone marrow mesenchymal stem cells by modulating gene expression of pro-inflammatory cytokines and immune response-related factors. J Biol Chem. 2007;282:7339–51.
Liu Y, Tan J, Li L, Li S, Zou S, Zhang Y, et al. Study on the molecular mechanisms of dlk1 stimulated lung cancer cell proliferation. Zhongguo Fei Ai Za Zhi. 2010;13:923–7.
Yanai H, Nakamura K, Hijioka S, Kamei A, Ikari T, Ishikawa Y, et al. Dlk-1, a cell surface antigen on foetal hepatic stem/progenitor cells, is expressed in hepatocellular, colon, pancreas and breast carcinomas at a high frequency. J Biochem. 2010;148:85–92.
Ceder JA, Jansson L, Helczynski L, Abrahamsson PA. Delta-like 1 (Dlk-1), a novel marker of prostate basal and candidate epithelial stem cells, is downregulated by notch signalling in intermediate/transit amplifying cells of the human prostate. Eur Urol. 2008;54:1344–53.
Thiery JP, Acloque H, Huang RY, Nieto MA. Epithelial-mesenchymal transitions in development and disease. Cell. 2009;139:871–90.
Klymenko Y, Kim O, Stack MS. Complex determinants of epithelial: mesenchymal phenotypic plasticity in ovarian cancer. Cancers (Basel). 2017;9:1–32.
Huang CC, Kuo HM, Wu PC, Cheng SH, Chang TT, Chang YC, et al. Correction to: soluble delta-like 1 homolog (DLK1) stimulates angiogenesis through Notch1/Akt/eNOS signaling in endothelial cells. Angiogenesis. 2018;21:901.
Ma Z, Cai H, Zhang Y, Chang L, Cui Y. MiR-129-5p inhibits non-small cell lung cancer cell stemness and chemoresistance through targeting DLK1. Biochem Biophys Res Commun. 2017;490:309–16.
Li L, Tan J, Zhang Y, Han N, Di X, Xiao T, et al. DLK1 promotes lung cancer cell invasion through upregulation of MMP9 expression depending on Notch signaling. PLoS ONE. 2014;9:e91509.
Baladron V, Ruiz-Hidalgo MJ, Gubina E, Bonvini E, Laborda J. Specific regions of the extracellular domain of dlk, an EGF-like homeotic protein involved in differentiation, participate in intramolecular interactions. Front Biosci. 2001;6:A25–32.
Laborda J. The role of the epidermal growth factor-like protein dlk in cell differentiation. Histol Histopathol. 2000;15:119–29.
Smas CM, Sul HS. Molecular mechanisms of adipocyte differentiation and inhibitory action of pref-1. Crit Rev Eukaryot Gene Expr. 1997;7:281–98.
Smas CM, Sul HS. Pref-1, a protein containing EGF-like repeats, inhibits adipocyte differentiation. Cell. 1993;73:725–34.
Smas CM, Chen L, Sul HS. Cleavage of membrane-associated pref-1 generates a soluble inhibitor of adipocyte differentiation. Mol Cell Biol. 1997;17:977–88.
Huang J, Zhang X, Zhang M, Zhu JD, Zhang YL, Lin Y, et al. Up-regulation of DLK1 as an imprinted gene could contribute to human hepatocellular carcinoma. Carcinogenesis. 2007;28:1094–103.
Yin D, Xie D, Sakajiri S, Miller CW, Zhu H, Popoviciu ML, et al. DLK1: increased expression in gliomas and associated with oncogenic activities. Oncogene. 2006;25:1852–61.
Kawakami T, Chano T, Minami K, Okabe H, Okada Y, Okamoto K. Imprinted DLK1 is a putative tumor suppressor gene and inactivated by epimutation at the region upstream of GTL2 in human renal cell carcinoma. Hum Mol Genet. 2006;15:821–30.
Wang Y, Sul HS. Ectodomain shedding of preadipocyte factor 1 (Pref-1) by tumor necrosis factor alpha converting enzyme (TACE) and inhibition of adipocyte differentiation. Mol Cell Biol. 2006;26:5421–35.
Kim YS, Kim E, Park YJ, Kim Y. Retinoic acid receptor beta enhanced the anti-cancer stem cells effect of beta-carotene by down-regulating expression of delta-like 1 homologue in human neuroblastoma cells. Biochem Biophys Res Commun. 2016;480:254–60.
Zhang SS, Tan JJ, Li L, Chen WT, Han NJ, Guo SP, et al. [Up-regulation of DLK1 in non-small cell lung cancer and the relevant molecular mechanism]. Zhonghua Zhong Liu Za Zhi. 2016;38:510–4.
Benetatos L, Hatzimichael E, Londin E, Vartholomatos G, Loher P, Rigoutsos I, et al. The microRNAs within the DLK1-DIO3 genomic region: involvement in disease pathogenesis. Cell Mol Life Sci. 2013;70:795–814.
Deng J, Wang L, Chen H, Hao J, Ni J, Chang, et al. Targeting epithelial-mesenchymal transition and cancer stem cells for chemoresistant ovarian cancer. Oncotarget. 2016;7:55771–88.
Haslehurst AM, Koti M, Dharsee M, Nuin P, Evans K, Geraci J, et al. EMT transcription factors snail and slug directly contribute to cisplatin resistance in ovarian cancer. BMC Cancer. 2012;12:91.
Haga CL, Phinney DG. MicroRNAs in the imprinted DLK1-DIO3 region repress the epithelial-to-mesenchymal transition by targeting the TWIST1 protein signaling network. J Biol Chem. 2012;287:42695–707.
Kim Y, Lin Q, Zelterman D, Yun Z. Hypoxia-regulated delta-like 1 homologue enhances cancer cell stemness and tumorigenicity. Cancer Res. 2009;69:9271–80.
Xu J, Wang M, Zhang Z, Zhao W, Wang C, Tu L, et al. Prognostic values of DLK1 for surgery and imatinib mesylate adjuvant therapy in gastrointestinal stromal tumors. Am J Cancer Res. 2016;6:2700–12.
Huang CC, Kuo HM, Wu PC, Cheng SH, Chang TT, Chang YC, et al. Soluble delta-like 1 homolog (DLK1) stimulates angiogenesis through Notch1/Akt/eNOS signaling in endothelial cells. Angiogenesis. 2018;21:299–312.
Baladron V, Ruiz-Hidalgo MJ, Nueda ML, Diaz-Guerra MJ, Garcia-Ramirez JJ, Bonvini E, et al. dlk acts as a negative regulator of Notch1 activation through interactions with specific EGF-like repeats. Exp Cell Res. 2005;303:343–59.
Rodriguez P, Higueras MA, Gonzalez-Rajal A, Alfranca A, Fierro-Fernandez M, Garcia-Fernandez RA, et al. The non-canonical NOTCH ligand DLK1 exhibits a novel vascular role as a strong inhibitor of angiogenesis. Cardiovasc Res. 2012;93:232–41.
Gasperowicz M, Otto F. The notch signalling pathway in the development of the mouse placenta. Placenta. 2008;29:651–9.
Guseh JS, Bores SA, Stanger BZ, Zhou Q, Anderson WJ, Melton DA, et al. Notch signaling promotes airway mucous metaplasia and inhibits alveolar development. Development. 2009;136:1751–9.
Nueda ML, Naranjo AI, Baladron V, Laborda J. The proteins DLK1 and DLK2 modulate NOTCH1-dependent proliferation and oncogenic potential of human SK-MEL-2 melanoma cells. Biochim Biophys Acta. 2014;1843:2674–84.
Nueda ML, Naranjo AI, Baladron V, Laborda J. Different expression levels of DLK1 inversely modulate the oncogenic potential of human MDA-MB-231 breast cancer cells through inhibition of NOTCH1 signaling. FASEB J. 2017;31:3484–96.
Falix FA, Aronson DC, Lamers WH, Gaemers IC. Possible roles of DLK1 in the Notch pathway during development and disease. Biochim Biophys Acta. 2012;1822:988–95.
Peters JM, Shah YM, Gonzalez FJ. The role of peroxisome proliferator-activated receptors in carcinogenesis and chemoprevention. Nat Rev Cancer. 2012;12:181–95.
Park JT, Li M, Nakayama K, Mao TL, Davidson B, Zhang Z, et al. Notch3 gene amplification in ovarian cancer. Cancer Res. 2006;66:6312–8.
Hu W, Liu T, Ivan C, Sun Y, Huang J, Mangala LS, et al. Notch3 pathway alterations in ovarian cancer. Cancer Res. 2014;74:3282–93.
Jung JG, Stoeck A, Guan B, Wu RC, Zhu H, Blackshaw S, et al. Notch3 interactome analysis identified WWP2 as a negative regulator of Notch3 signaling in ovarian cancer. PLoS Genet. 2014;10:e1004751.
Chen X, Thiaville MM, Chen L, Stoeck A, Xuan J, Gao M, et al. Defining NOTCH3 target genes in ovarian cancer. Cancer Res. 2012;72:2294–303.
Park JT, Shih IeM, Wang TL. Identification of Pbx1, a potential oncogene, as a Notch3 target gene in ovarian cancer. Cancer Res. 2008;68:8852–60.
Park JT, Chen X, Trope CG, Davidson B, Shih Ie M, Wang TL. Notch3 overexpression is related to the recurrence of ovarian cancer and confers resistance to carboplatin. Am J Pathol. 2010;177:1087–94.
Liu GS, Wang JH, Lee JH, Tsai PJ, Tsai HE, Sheu SJ, et al. Gene delivery by subconjunctival injection of adenovirus in rats: a study of local distribution, transgene duration and safety. PLoS ONE. 2015;10:e0143956.
Hu TH, Huang CC, Liu LF, Lin PR, Liu SY, Chang HW, et al. Expression of hepatoma-derived growth factor in hepatocellular carcinoma. Cancer. 2003;98:1444–56.
Euhus DM, Hudd C, LaRegina MC, Johnson FE. Tumor measurement in the nude mouse. J Surg Oncol. 1986;31:229–34.
Wu JC, Wang CT, Hung HC, Wu WJ, Wu DC, Chang MC, et al. Heteronemin is a novel c-Met/STAT3 inhibitor against advanced prostate cancer cells. Prostate. 2016;76:1469–83.
Author contributions
Conception and design: C.C.H., S.H.C., and M.H.T. Development of methodology: C.C.H., S.H.C., M.L.K., and T.H.U. Acquisition of data (provided animals, acquired and managed patients, provided facilities, etc.): C.C.H., C.H.W., W.Y.L., and J.S.W. Administrative, technical, or material support: C.C.H., S.H.C., C.H.W., M.L.K., T.H.C., S.T.H., C.T.F., and S.C.H. Writing, review, and/or revision of the manuscript: C.C.H., S.H.C., C.H.W., and M.H.T. Study supervision: C.C.H., and M.H.T.
Funding
This work was supported by grants from the Ministry of Science and Technology, Taiwan (NSC101-2320-B-182-009, NSC102-2320-B-182-011, MOST103-2320-B-182-005-MY3 to C.C.H., and MOST105-2325-B-110-001 to M.H.T.). We thank Chang Gung Medical Foundation Kaohsiung Chang Gung Memorial Hospital Tissue Bank (CLRPG8E0161 and CLRPG8F1701), and the Kaohsiung Armed Forces General Hospital, Taiwan (107-10) for technical support.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Huang, CC., Cheng, SH., Wu, CH. et al. Delta-like 1 homologue promotes tumorigenesis and epithelial-mesenchymal transition of ovarian high-grade serous carcinoma through activation of Notch signaling. Oncogene 38, 3201–3215 (2019). https://doi.org/10.1038/s41388-018-0658-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41388-018-0658-5
This article is cited by
-
Exploring the cellular and molecular differences between ovarian clear cell carcinoma and high-grade serous carcinoma using single-cell RNA sequencing and GEO gene expression signatures
Cell & Bioscience (2023)
-
MiR-26a-5p as a useful therapeutic target for upper tract urothelial carcinoma by regulating WNT5A/β-catenin signaling
Scientific Reports (2022)
-
Extracellular vesicles derived from ascitic fluid enhance growth and migration of ovarian cancer cells
Scientific Reports (2021)
-
Age-associated genes in human mammary gland drive human breast cancer progression
Breast Cancer Research (2020)
-
Hypoxia-induced release, nuclear translocation, and signaling activity of a DLK1 intracellular fragment in glioma
Oncogene (2020)