Abstract
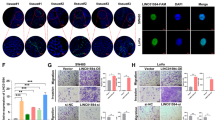
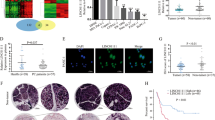
Long noncoding RNAs (lncRNAs) are implicated in human cancer, but their mechanisms of action are largely unknown. In this study, we investigated lncRNA alterations that contribute to colorectal cancer (CRC) through microarray expression profiling in CRC patient samples. Here, we report that the CRC-associated lncRNA PVT1-214 is a key regulator of CRC development and progression; patients with high PVT1-214 expression had a shorter survival and poorer prognosis. In vitro and in vivo investigation of the role of PVT1-214 revealed a complex integrated phenotype affecting cell growth, stem-like properties, migration, and invasion. Furthermore, using RNA pull-down and mass spectrometry, we found that Lin28 (also known as Lin28A), a highly conserved RNA-binding protein, is associated with PVT1-214. Strikingly, we found that PVT1-214 not only upregulated Lin28 protein expression in CRC cells by stabilizing Lin28, but also participated in crosstalk with Lin28 mRNA through competition for miR-128 binding, imposing an additional level of post-transcriptional regulation. In addition, we further show that PVT1-214 repressed expression of let-7 family miRNAs, which was abrogated by Lin28 knockdown. Taken together, our findings support a model in which the PVT1-214/Lin28/let-7 axis serves as a critical regulator of CRC pathogenesis, which may simulate a new direction for CRC therapeutic development.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Change history
09 November 2021
A Correction to this paper has been published: https://doi.org/10.1038/s41388-021-02093-w
References
Djebali S, Davis CA, Merkel A, Dobin A, Lassmann T, Mortazavi A, et al. Landscape of transcription in human cells. Nature. 2012;489:101–8.
Tay Y, Rinn J, Pandolfi PP. The multilayered complexity of ceRNA crosstalk and competition. Nature. 2014;505:344–52.
Rupaimoole R, Slack FJ. MicroRNA therapeutics: towards a new era for the management of cancer and otherdiseases. Nat Rev Drug Discov. 2017;16:203–22.
Batista PJ, Chang HY. Long noncoding RNAs: cellular address codes in development and disease. Cell. 2013;152:1298–307.
Dhamija S, Diederichs S. From junk to master regulators of invasion: lncRNA functions in migration, EMTand metastasis. Int J Cancer. 2016;139:269–80.
Malone CD, Hannon GJ. Small RNAs as guardians of the genome. Cell. 2009;136:656–68.
Volders PJ, Helsens K, Wang X, Menten B, Martens L, Gevaert K, et al. LNCipedia: a database for annotated human lncRNA transcript sequences and structures. Nucl Acids Res. 2013;41:D246–51.
Chen FC, Pan CL, Lin HY. Functional implications of RNA splicing for human long intergenic noncoding RNAs. Evol Bioinform Online. 2014;10:219–28.
Cao C, Zhang T, Zhang D, Xie L, Zou X, Lei L, et al. The long non-coding RNA, SNHG6-003, functions as a competing endogenous RNA to promote the progression of hepatocellular carcinoma. Oncogene. 2017;36:1112–22.
Xu MD, Wang Y, Weng W, Wei P, Qi P, Zhang Q, et al. A Positive feedback loop of lncRNA-PVT1 and FOXM1 facilitates gastric cancergrowth and invasion. Clin Cancer Res. 2017;23:2071–80.
Kong R, Zhang EB, Yin DD, You LH, Xu TP, Chen WM, et al. Long noncoding RNA PVT1 indicates a poor prognosis of gastric cancer and promotes cell proliferation through epigenetically regulating p15 and p16. Mol Cancer. 2015;14:82–96.
Wang F, Yuan JH, Wang SB, Yang F, Yuan SX, Ye C, et al. Oncofetal long noncoding RNA PVT1 promotes proliferation and stem cell-likeproperty of hepatocellular carcinoma cells by stabilizing NOP2. Hepatology. 2014;60:1278–90.
Wan L, Sun M, Liu GJ, Wei CC, Zhang EB, Kong R, et al. Long Noncoding RNA PVT1 promotes non-small cell lung cancer cell proliferation through epigenetically regulating LATS2 expression. Mol Cancer Ther. 2016;15:1082–94.
Li PD, Hu JL, Ma C, Ma H, Yao J, Chen LL, et al. Upregulation of the long non-coding RNA PVT1 promotes esophageal squamous cellcarcinoma progression by acting as a molecular sponge of miR-203 and LASP1. Oncotarget. 2017;8:34164–176.
Takahashi Y, Sawada G, Kurashige J, Uchi R, Matsumura T, Ueo H, et al. Amplification of PVT-1 is involved in poor prognosis via apoptosis inhibition in colorectal cancers. Br J Cancer. 2014;110:164–171.
Zhou Q, Chen F, Zhao J, Li B, Liang Y, Pan W, et al. Long non-coding RNA PVT1 promotes osteosarcoma development by acting as a molecular sponge to regulate miR-195. Oncotarget. 2016;7:82620–633.
Tseng YY, Moriarity BS, Gong W, Akiyama R, Tiwari A, Kawakami H, et al. PVT1 dependence in cancer with MYC copy-number increase. Nature. 2014;512:82–96.
Wang Y, Zhou J, Wang Z, Wang P, Li S. Upregulation of SOX2 activated LncRNA PVT1 expression promotes breast cancer cell growth and invasion. Biochem Biophys Res Commun. 2017;493:429–36.
Guo K, Yao J, Yu Q, Li Z, Huang H, Cheng J, et al. The expression pattern of long non-coding RNA PVT1 in tumor tissues and inextracellular vesicles of colorectal cancer correlates with cancer progression. Tumour Biol. 2017;39:9122–32.
Zheng J, Yu F, Dong P, Wu L, Zhang Y, Hu Y, et al. Long non-coding RNA PVT1 activates hepatic stellate cells through competitivelybinding microRNA-152. Oncotarget. 2016;7:62886–897.
Izaurralde E. Elucidating the temporal order of silencing. EMBO Rep. 2012;13:662–73.
Filipowicz W, Bhattacharyya SN, Sonenberg N. Mechanisms of post-transcriptional regulation by microRNAs: are the answers insight. Nat Rev Genet. 2008;9:102–14.
Heo I, Joo C, Cho J, Ha M, Han J, Kim VN. Lin28 mediates the terminal uridylation of let-7 precursor MicroRNA. Mol Cell. 2008;32:276–84.
Newman MA, Thomson JM, Hammond SM. Lin-28 interaction with the Let-7 precursor loop mediates regulated microRNAprocessing. RNA. 2008;14:1539–49.
Mattick JS, Makunin IV. Non-coding RNA. Hum Mol Genet. 2006;15:R17–29.
Viswanathan SR, Powers JT, Einhorn W, Hoshida Y, Ng TL, Toffanin S, et al. Lin28 promotes transformation and is associated with advanced human malignancies. Nat Genet. 2009;41:843–58.
Cao D, Allan RW, Cheng L, Peng Y, Guo CC, Dahiya N, et al. RNA-binding protein LIN28 is a marker for testicular germ cell tumors. Hum Pathol. 2011;42:710–18.
Wang T, He Y, Zhu Y, Chen M, Weng M, Yang C, et al. Comparison of the expression and function of Lin28A and Lin28B in colon cancer. Oncotarget. 2016;7:79605–16.
Salmena L, Poliseno L, Tay Y, Kats L, Pandolfi PP. A ceRNA hypothesis: the Rosetta Stone of a hidden RNA language. Cell. 2011;146:353–68.
Song YX, Sun JX, Zhao JH, Yang YC, Shi JX, Wu ZH, et al. Non-coding RNAs participate in the regulatory network of CLDN4 via ceRNA mediatedmiRNA evasion. Nat Commun. 2017;8:289–302.
Ma MZ, Zhang Y, Weng MZ, Wang SH, Hu Y, Hou ZY, et al. Long noncoding RNA GCASPC, a target of miR-17-3p, negatively regulates pyruvate carboxylase-dependent cell proliferation in gallbladder cancer. Cancer Res. 2016;76:5361–71.
Lu W, Wang J, Yang G, Yu N, Huang Z, Xu H, et al. Posttranscriptional regulation of Galectin-3 by miR-128 contributes to colorectal cancer progression. Oncotarget. 2017;8:15242–351.
Powers JT, Tsanov KM, Pearson DS, Roels F, Spina CS, Ebright R, et al. Multiple mechanisms disrupt the let-7 microRNA family in neuroblastoma. Nature. 2016;535:246–51.
Gokbuget D, Pereira JA, Bachofner S, Marchais A, Ciaudo C, Stoffel M, et al. The Lin28/let-7 axis is critical for myelination in the peripheral nervoussystem. Nat Commun. 2015;6:8584–93.
Cho SW, Xu J, Sun R, Mumbach MR, Carter AC, Chen YG, et al. Promoter of lncRNA gene PVT1 is a tumor-suppressor dna boundary element. Cell. 2018;173:1398–412.
Di CS, Zhang F, Sancho A, Li S, Aguilo F, Sun Y, et al. RBM5-AS1 is critical for self-renewal of colon cancer stem-like cells. Cancer Res. 2016;76:5615–27.
He F, Chen H, Yang P, Wu Q, Zhang T, Wang C, et al. Gankyrin sustains PI3K/GSK-3beta/beta-catenin signal activation and promotescolorectal cancer aggressiveness and progression. Oncotarget. 2016;7:81156–71.
He F, Peng F, Xia X, Zhao C, Luo Q, Guan W, et al. MiR-135a promotes renal fibrosis in diabetic nephropathy by regulating TRPC1. Diabetologia. 2014;57:1726–36.
Acknowledgements
This work was supported by grants from the National Natural Science Foundation of China (Grant no. 8187101464, 81600654 and 81600862); the Guangdong Natural Science Foundation (Grant no. 2017A030311035, 2016A030313490 and 2016A030310227); the Fundamental Research Funds for the Central Universities (Grant no. XZYXD2175080); and the Guangdong Provincial Department of Science and Technology (Grant no. 2017A020215146); Guangzhou Science Technology and Innovation Commission (Grant no. 201805010003); and US National Institutes of Health (NIH) grant R01 HL136507.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Accession numbers: Microarray data have been deposited in the Gene Expression Omnibus database (accession number GSE109454).
Electronic supplementary material
Rights and permissions
About this article
Cite this article
He, F., Song, Z., Chen, H. et al. Long noncoding RNA PVT1-214 promotes proliferation and invasion of colorectal cancer by stabilizing Lin28 and interacting with miR-128. Oncogene 38, 164–179 (2019). https://doi.org/10.1038/s41388-018-0432-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41388-018-0432-8
This article is cited by
-
Long non-coding RNA signature in colorectal cancer: research progression and clinical application
Cancer Cell International (2023)
-
AC010883.5 promotes cell proliferation, invasion, migration, and epithelial-to-mesenchymal transition in cervical cancer by modulating the MAPK signaling pathway
BMC Cancer (2023)
-
Identification of LncPVT1 and CircPVT1 as prognostic biomarkers in human colorectal polyps
Scientific Reports (2023)
-
LncRNA PVT1 influences breast cancer cells glycolysis through sponging miR-145-5p
Genes & Genomics (2023)
-
Long Non-coding RNA Involved in the Pathophysiology of Atrial Fibrillation
Cardiovascular Drugs and Therapy (2023)