Abstract
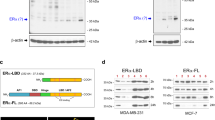
The receptor tyrosine kinase Ret, a key gain-of-function mutated oncoprotein in thyroid carcinomas, has recently been implicated in other cancer types. While Ret copy number gains and mutations have been reported at low frequencies in breast tumors, we and others have reported that Ret is overexpressed in about 40% of human tumors and this correlates with poor patient prognosis. Ret activation regulates numerous intracellular pathways related to proliferation and inflammation, but it is not known whether abnormal Ret expression is sufficient to induce mammary carcinomas. Using a novel doxycycline-inducible transgenic mouse model with the MMTV promoter controlling Ret expression, we show that overexpression of wild-type Ret in the mammary epithelium produces mammary tumors, displaying a morphology that recapitulates characteristics of human luminal breast tumors. Ret-evoked tumors are estrogen receptor positive and negative for progesterone receptor. Moreover, tumors rapidly regress after doxycycline withdrawal, indicating that Ret is the driving oncoprotein. Using next-generation sequencing, we examined the levels of transcripts in these tumors, confirming a luminal signature. Ret-evoked tumors have been passaged in mice and used to test novel therapeutic approaches. Importantly, we have determined that tumors are resistant to endocrine therapy, but respond successfully to treatment with a Ret kinase inhibitor. Our data provide the first compelling evidence for an oncogenic role of non-mutated Ret in the mammary gland and are an incentive for clinical development of Ret as a cancer biomarker and therapeutic target.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Hynes NE, Watson CJ. Mammary gland growth factors: roles in normal development and in cancer. Cold Spring Harb Perspect Biol. 2010;2:a003186.
Grieco M, Santoro M, Berlingieri MT, Melilo RM, Donghi R, Bongarzone I, et al. PTC is a novel rearranged form of the ret proto-oncogene and is frequently detected in vivo in human thyroid papillary carcinomas. Cell. 1990;60:557–63.
Kohno T, Ichikawa H, Totoki Y, Yasuda K, Hiramoto M, Nammo T. et al. KIF5B-RET fusions in lung adenocarcinoma. Nat Med. 2012;18:375–7.
Stransky N, Cerami E, Schalm S, Kim JL, Lengauer C. The landscape of kinase fusions in cancer. Nat Commun. 2014;5:4846.
Nik-Zainal S, Davies H, Staaf J, Ramakrishna M, Glodzik D, Zou X. et al. Landscape of somatic mutations in 560 breast cancer whole-genome sequences. Nature. 2016;534:47–54.
Boulay A, Breuleux M, Stephan C, Fux C, Brisken C, Fiche M, et al. The Ret receptor tyrosine kinase pathway functionally interacts with the ERalpha pathway in breast cancer. Cancer Res. 2008;68:3743–51.
Gattelli A, Nalvarte I, Boulay A, Roloff TC, Schreiber M, Carragher N, et al. Ret inhibition decreases growth and metastatic potential of estrogen receptor positive breast cancer cells. EMBO Mol Med. 2013;5:1335–50.
Esseghir S, Todd SK, Hunt T, Poulsom R, Plaza-Menacho I, Reis-Filho JS, et al. A role for glial cell derived neurotrophic factor induced expression by inflammatory cytokines and RET/GFR alpha 1 receptor up-regulation in breast cancer. Cancer Res. 2007;67:11732–41.
Plaza-Menacho I, Morandi A, Robertson D, Pancholi S, Drury S, Dowsett M, et al. Targeting the receptor tyrosine kinase RET sensitizes breast cancer cells to tamoxifen treatment and reveals a role for RET in endocrine resistance. Oncogene. 2010;29:4648–57.
Spanheimer PM, Park JM, Askeland RW, Kulak MV, Woodfield GW, De Andrade JP, et al. Inhibition of Ret increases the efficacy of antiestrogen and is a novel treatment strategy for luminal breast cancer. Clin Cancer Res. 2014;20:2115–25.
Gunther EJ, Belka GK, Wertheim GB, Wang J, Hartman JL, Boxer RB, et al. A novel doxycycline-inducible system for the transgenic analysis of mammary gland biology. FASEB J. 2002;16:283–92.
Moody SE, Sarkisian CJ, Hahn KT, Gunther EJ, Pickup S, Dugan KD, et al. Conditional activation of Neu in the mammary epithelium of transgenic mice results in reversible pulmonary metastasis. Cancer Cell. 2002;2:451–61.
Sarkisian JC, Keister BA, Stairs DB, Boxer RB, Moody SE, Chodosh LA. Dose-dependent oncogene-induced senescence in vivo and its evasion during mamary tumorigenesis. Nat Cell Biol. 2007;9:493–505.
Woodfield GW, Chen Y, Bair TB, Domann FE, Weigel J. Identification of primary gene targets of TFAP2C in hormone responsive breast carcinoma cells. Genes Chrom Cancer. 2010;49:948–62.
Morandi A, Martin LA, Gao Q, Pancholi S, Mackay A, Robertson D. et al. GDNF-Ret signalling in ER-positive breast cancers is a key determinant of response and resistance to aromatase inhibitors. Cancer Res. 2013;73:3783–95.
Cardiff RD, Wellings SR. The comparative pathology of human and mouse mammary glands. J Mammary Gland Biol Neoplasia. 1999;4:105–22.
Iwamoto T, Takahashi M, Ito M, Hamaguchi M, Isobe K, Misawa N, et al. Oncogenicity of the ret transforming gene in MMTV/ret transgenic mice. Oncogene. 1990;5:535–42.
Parker JS, Mullins M, Cheang MC, Leung S, Voduc D, Vickery T, et al. Supervised risk predictor of breast cancer based on intrinsic subtypes. J Clin Oncol. 2009;27:1160–7.
Avalle L, Pensa S, Regis G, Novelli F, Poli V. Stat1 and Stat3 in tumorigenesis. A matter of balance. JAK-STAT. 2012;1:65–72.
Chan SR, Vermi W, Luo J, Lucini L, Rickert C, Fowler AM, et al. Stat1-deficient mice spontaneously develop estrogen receptor a-positive luminal mammary carcinomas. Brest Cancer Res. 2012;14:R16.
Rakha EA, El-Sayed ME, Green AR, Paish EC, Powe DG, Gee J, et al. Biologic and clinical characteristics of breast cancer with single hormone receptor positive phenotype. J Clin Oncol. 2007;25:4772–8.
Akeno-Stuart N, Croyle M, Knauf JA, Malaguarnera R, Vitagliano D, Santoro M, et al. The RET kinase inhibitor NVP-AST487 blocks growth and calcitonin gene expression through distinct mechanisms in medullary thyroid cancer cells. Cancer Res. 2007;67:6956–64.
Kohno T, Tsuta K, Tsuchihara K, Nakaodu T, Yoh K, Goto K. Ret fusion gene: Translation to personalized lung cancer therapy. Cancer Sci. 2013;104:1396–1400.
Kang J, Qian PX, Pandey V, Perry JK, Miller LD, Liu ET, et al. Artemin is estrogen regulated and mediates antiestrogen resistance in mammary carcinoma. Oncogene. 2010;31:402.
Baselga J, Campone M, Piccart M, Burris HA, Ruggo HS, Sahmoud T, et al. Everolimus in postmenopausal hormone-receptor-positive advanced breast cancer. N Engl J Med. 2012;366:520–9.
Acknowledgements
We thank Dr. Giorgio Caravatti (Novartis Institutes for Biomedical Research, Basel, Switzerland) and all members of Hynes and Gattelli labs for helpful discussions.
Funding
AG was supported by grant KG101234 from Susan G. Komen for the Cure® and grant GF N° 02 from Fundación para el Progreso de la Medicina Córdoba. NEH was supported by grants from Susan G. Komen for the Cure® SAC110041, from Krebsliga beider Basel 10-2030 and by the Novartis Research Foundation.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Gattelli, A., García Solá, M.E., Roloff, T.C. et al. Chronic expression of wild-type Ret receptor in the mammary gland induces luminal tumors that are sensitive to Ret inhibition. Oncogene 37, 4046–4054 (2018). https://doi.org/10.1038/s41388-018-0235-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41388-018-0235-y
This article is cited by
-
RET signaling in breast cancer therapeutic resistance and metastasis
Breast Cancer Research (2023)
-
Ret Receptor Has Distinct Alterations and Functions in Breast Cancer
Journal of Mammary Gland Biology and Neoplasia (2020)
-
RET rearrangements are actionable alterations in breast cancer
Nature Communications (2018)