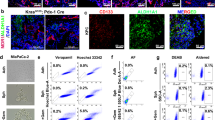
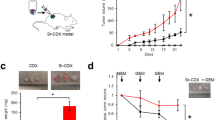
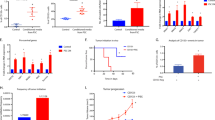
Abstract
Cancer research is increasingly dependent of patient-derived xenograft model (PDX). However, a major point of concern regarding the PDX model remains the replacement of the human stroma with murine counterpart. In the present work we aimed at clarifying the significance of the human-to-murine stromal replacement for the fidelity of colorectal cancer (CRC) and liver metastasis (CRC-LM) PDX model. We have conducted a comparative metabolic analysis between 6 patient tumors and corresponding PDX across 4 generations. Metabolic signatures of cancer cells and stroma were measured separately by MALDI-imaging, while metabolite changes in entire tumors were quantified using mass spectrometry approach. Measurement of glucose metabolism was also conducted in vivo using [18F]-fluorodeoxyglucose (FDG) and positron emission tomography (PET). In CRC/CRC-LM PDX model, human stroma was entirely replaced at the second generation. Despite this change, MALDI-imaging demonstrated that the metabolic profiles of both stromal and cancer cells remained stable for at least four generations in comparison to the original patient material. On the tumor level, profiles of 86 water-soluble metabolites as well as 93 lipid mediators underlined the functional stability of the PDX model. In vivo PET measurement of glucose uptake (reflecting tumor glucose metabolism) supported the ex vivo observations. Our data show for the first time that CRC/CRC-LM PDX model maintains the functional stability at the metabolic level despite the early replacement of the human stroma by murine cells. The findings demonstrate that human cancer cells actively educate murine stromal cells during PDX development to adopt the human-like phenotype.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Siolas D, Hannon GJ. Patient-derived tumor xenografts: transforming clinical samples into mouse models. Cancer Res. 2013;73:5315–9.
Tentler JJ, Tan AC, Weekes CD, Jimeno A, Leong S, Pitts TM, et al. Patient-derived tumour xenografts as models for oncology drug development. Nat Rev Clin Oncol. 2012;9:338–50.
Giovanella BC, Stehlin JS, Wall ME, Wani MC, Nicholas AW, Liu LF, et al. DNA topoisomerase I--targeted chemotherapy of human colon cancer in xenografts. Science. 1989;246:1046–8.
Houghton JA, Maroda SJ Jr., Phillips JO, Houghton PJ. Biochemical determinants of responsiveness to 5-fluorouracil and its derivatives in xenografts of human colorectal adenocarcinomas in mice. Cancer Res. 1981;41:144–9.
Kim MP, Evans DB, Wang H, Abbruzzese JL, Fleming JB, Gallick GE. Generation of orthotopic and heterotopic human pancreatic cancer xenografts in immunodeficient mice. Nat Protoc. 2009;4:1670–80.
Werner-Klein M, Proske J, Werno C, Schneider K, Hofmann HS, Rack B, et al. Immune humanization of immunodeficient mice using diagnostic bone marrow aspirates from carcinoma patients. PLoS ONE 2014;9:e97860.
Walsh NC, Kenney LL, Jangalwe S, Aryee KE, Greiner DL, Brehm MA, et al. Humanized mouse models of clinical disease. Annu Rev Pathol. 2017;12:187–215.
DeRose YS, Wang G, Lin YC, Bernard PS, Buys SS, Ebbert MT, et al. Tumor grafts derived from women with breast cancer authentically reflect tumor pathology, growth, metastasis and disease outcomes. Nat Med. 2011;17:1514–20.
Zhang X, Claerhout S, Prat A, Dobrolecki LE, Petrovic I, Lai Q, et al. A renewable tissue resource of phenotypically stable, biologically and ethnically diverse, patient-derived human breast cancer xenograft models. Cancer Res. 2013;73:4885–97.
Li S, Shen D, Shao J, Crowder R, Liu W, Prat A, et al. Endocrine-therapy-resistant ESR1 variants revealed by genomic characterization of breast-cancer-derived xenografts. Cell Rep. 2013;4:1116–30.
Cassidy JW, Caldas C, Bruna A. Maintaining tumor heterogeneity in patient-derived tumor xenografts. Cancer Res. 2015;75:2963–8.
Hylander BL, Punt N, Tang H, Hillman J, Vaughan M, Bshara W, et al. Origin of the vasculature supporting growth of primary patient tumor xenografts. J Transl Med. 2013;11:110.
Xu L, Pirollo KF, Tang WH, Rait A, Chang EH. Transferrin-liposome-mediated systemic p53 gene therapy in combination with radiation results in regression of human head and neck cancer xenografts. Hum Gene Ther. 1999;10:2941–52.
Sanz L, Cuesta AM, Salas C, Corbacho C, Bellas C, Alvarez-Vallina L. Differential transplantability of human endothelial cells in colorectal cancer and renal cell carcinoma primary xenografts. Lab Invest. 2009;89:91–7.
Chiavarina B, Turtoi A. Collaborative and Defensive Fibroblasts in Tumor Progression and Therapy Resistance. Curr Med Chem. 2017;24:2846–2859.
Kalluri R. The biology and function of fibroblasts in cancer. Nat Rev Cancer. 2016;16:582–98.
Lou S, Balluff B, Cleven AH, Bovee JV, McDonnell LA. An experimental guideline for the analysis of histologically heterogeneous tumors by MALDI-TOF mass spectrometry imaging. Biochimica et Biophysica Acta. 2017;1865:957–966.
McDonnell LA, Walch A, Stoeckli M, Corthals GL. MSiMass list: a public database of identifications for protein MALDI MS imaging. J Proteome Res. 2014;13:1138–42.
Lin D, Ettinger SL, Qu S, Xue H, Nabavi N, Choi SYC, et al. Metabolic heterogeneity signature of primary treatment-naive prostate cancer. Oncotarget. 2017;8:25928–41.
Amos-Landgraf JM, Heijmans J, Wielenga MC, Dunkin E, Krentz KJ, Clipson L, et al. Sex disparity in colonic adenomagenesis involves promotion by male hormones, not protection by female hormones. Proc Natl Acad Sci USA. 2014;111:16514–9.
Adams MC, Turkington TG, Wilson JM, Wong TZ. A systematic review of the factors affecting accuracy of SUV measurements. AJR Am J Roentgenol. 2010;195:310–20.
Sha W, Ye H, Iwamoto KS, Wong KP, Wilks MQ, Stout D, et al. Factors affecting tumor (18) F-FDG uptake in longitudinal mouse PET studies. EJNMMI Res. 2013;3:51.
Valtorta S, Moro M, Prisinzano G, Bertolini G, Tortoreto M, Raccagni I, et al. Metabolic evaluation of non-small cell lung cancer patient-derived xenograft models using 18F-FDG PET: a potential tool for early therapy response. J Nucl Med. 2017;58:42–7.
Kilkenny C, Browne WJ, Cuthill IC, Emerson M, Altman DG. Improving bioscience research reporting: the ARRIVE guidelines for reporting animal research. Osteoarthr Cartil. 2012;20:256–60.
Blomme A, Costanza B, de Tullio P, Thiry M, Van Simaeys G, Boutry S, et al. Myoferlin regulates cellular lipid metabolism and promotes metastases in triple-negative breast cancer. Oncogene. 2017;36:2116–2130.
Fueger BJ, Czernin J, Hildebrandt I, Tran C, Halpern BS, Stout D, et al. Impact of animal handling on the results of 18F-FDG PET studies in mice. J Nucl Med. 2006;47:999–1006.
Wahl RL, Jacene H, Kasamon Y, Lodge MA. From RECIST to PERCIST: evolving considerations for PET response criteria in solid tumors. J Nucl Med. 2009;50(Suppl 1):122S–50S.
Boellaard R, O’Doherty MJ, Weber WA, Mottaghy FM, Lonsdale MN, Stroobants SG, et al. FDG PET and PET/CT: EANM procedure guidelines for tumour PET imaging: version 1.0. Eur J Nucl Med Mol Imaging. 2010;37:181–200.
van Velden FH, Cheebsumon P, Yaqub M, Smit EF, Hoekstra OS, Lammertsma AA, et al. Evaluation of a cumulative SUV-volume histogram method for parameterizing heterogeneous intratumoural FDG uptake in non-small cell lung cancer PET studies. Eur J Nucl Med Mol Imaging. 2011;38:1636–47.
Acknowledgements
The authors acknowledge the experimental support of Dr. Chantal Humblet, Mrs. Alice Marquet (GIGA-histology platform, ULg), Mr. Vincent Hennequière, Mrs. Naima Maloujahmoum (Metastasis Research Laboratory), Mrs. Yezza Ben Salah (Tumor Microenvironment and Resistance to Treatment Lab, IRCM), Mr. Nicolas Passon (CMMI) and Mrs. Mari-Aline Laute (CMMI). The authors are thankful to Dr. Stéphanie Gofflot (Biobank, University Hospital Liege) for providing patient material as well as the Cyclotron Unit of Erasme Hospital (Brussels, Belgium) for providing [18F]-FDG. Prof. Robert N. Muller (CMMI, Director of international partnerships) is thanked for his scientific input and support. Mr. Mathieu Roch (CMMI) is thanked for tumor volume measurements on MR images.This work was supported with grants from the University of Liège, National Fund for Scientific Research (FNRS) and Gunma University (GIAR Research Program for Omics-Based Medical Science). The CMMI is supported by the European Regional Development Fund (ERDF), the Walloon Region, the Fondation ULB, the Fonds Erasme and the “Association Vinçotte Nuclear” (AVN). GD is supported by the ERDF and the Walloon Region. Akeila Bellahcène is a Research Director at the FNRS. AT is a senior research fellow of the French National Institute of Health and Medical Research (INSERM) and is supported by LabEx MabImprove Starting Grant. No funding bodies had any role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Vincent Castronovo and Andrei Turtoi contributed equally to this work.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Blomme, A., Van Simaeys, G., Doumont, G. et al. Murine stroma adopts a human-like metabolic phenotype in the PDX model of colorectal cancer and liver metastases. Oncogene 37, 1237–1250 (2018). https://doi.org/10.1038/s41388-017-0018-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41388-017-0018-x
This article is cited by
-
Personalizing non-small cell lung cancer treatment through patient-derived xenograft models: preclinical and clinical factors for consideration
Clinical and Translational Oncology (2024)
-
Patient-derived xenograft models in cancer therapy: technologies and applications
Signal Transduction and Targeted Therapy (2023)
-
Rethinking our approach to cancer metabolism to deliver patient benefit
British Journal of Cancer (2023)
-
An integrative approach for exploring the nature of fibroepithelial neoplasms
British Journal of Cancer (2023)
-
Patient-derived xenograft models in hepatopancreatobiliary cancer
Cancer Cell International (2022)