Abstract
GABA-A receptors (GABAARs) are crucial for development and function of the brain. Altered GABAergic transmission is hypothesized to be involved in neurodevelopmental disorders. Recently, we identified Shisa7 as a GABAAR auxiliary subunit that modulates GABAAR trafficking and GABAergic transmission. However, the underlying molecular mechanisms remain elusive. Here we generated a knock-in (KI) mouse line that is phospho-deficient at a phosphorylation site in Shisa7 (S405) and combined with electrophysiology, imaging and behavioral assays to illustrate the role of this site in GABAergic transmission and plasticity as well as behaviors. We found that expression of phospho-deficient mutants diminished α2-GABAAR trafficking in heterologous cells. Additionally, α1/α2/α5-GABAAR surface expression and GABAergic inhibition were decreased in hippocampal neurons in KI mice. Moreover, chemically induced inhibitory long-term potentiation was abolished in KI mice. Lastly, KI mice exhibited hyperactivity, increased grooming and impaired sleep homeostasis. Collectively, our study reveals a phosphorylation site critical for Shisa7-dependent GABAARs trafficking which contributes to behavioral endophenotypes displayed in neurodevelopmental disorders.
Similar content being viewed by others
Introduction
GABA-A receptors (GABAARs) are ligand-gated ion channels which critically regulate fast inhibitory neurotransmission in the central nervous system (CNS). Diversity in GABAergic inhibition arises from the pentameric assembly of 19 subunits into distinct receptor subtypes, which are broadly classified as synaptic or extrasynaptic GABAARs depending on their localization and biophysical properties [1, 2]. Whereas synaptic GABAARs predominately contribute to phasic inhibition, extrasynaptic GABAARs have a more defined role in mediating tonic inhibition [3, 4]. Due to their ubiquity across the CNS, it is not surprising that impaired GABAAR-mediated signaling is associated with a broad range of neurological and psychiatric disorders [5,6,7,8]. Therefore, defining mechanisms that regulate GABAAR activity and determining how perturbation of these processes are involved in pathophysiological conditions continue to be of high importance.
Historically, probing ligand-gated receptor function has been constrained to the level of their pore-forming subunits. However, this dogma has recently been challenged by the identification of protein complexes that coexist with native receptors. Accessory proteins and auxiliary subunits have been identified to interact with several voltage-gated ion channels and ligand-gated ion channels such as voltage-gated calcium channels, nicotinic acetylcholine receptors, and AMPA receptors [9]. Adding to this growing list of receptor-interacting proteins, GABAAR-associated transmembrane proteins have become the most recent addition and, so far, consist of lipoma HMGIC fusion partner-like 4 (LH4), Cleft lip and palate transmembrane protein 1 (Clptm1), and Shisa7 [10, 11]. Shisa7 is a single-pass transmembrane protein that was identified in a proteomic screen and as part of native GABAAR complexes in previous work [12], although it has also been reported that Shisa7 could interact with AMPARs [13, 14]. Our initial study has identified that Shisa7 localizes at inhibitory synapses and directly interacts with several GABAAR subunits [15]. Coexpression of Shisa7 with specific GABAAR subtypes in heterologous cells resulted in enhanced GABA-evoked, whole-cell currents. Moreover, loss of Shisa7 impaired phasic GABAergic transmission in hippocampal CA1 pyramidal neurons. Interestingly, Shisa7 also accelerated IPSC decay kinetics and loss of Shisa7 blunted the pharmacological and behavioral effects of diazepam, a prototypical benzodiazepine. In addition, we found that Shisa7 regulated trafficking of extrasynaptic α5-GABAARs, but not δ-GABAARs [16]. Specifically, loss of Shisa7 resulted in diminished trafficking of α5-GABAARs and tonic currents. Interestingly, S405 on Shisa7 was identified as a critical phosphorylation site for protein kinase A (PKA) and important for extrasynaptic α5-GABAAR trafficking [16].
Recently, accumulating evidence has shown that disrupted coordination of excitatory and inhibitory inputs throughout the CNS are associated with neurodevelopmental disorders such as autism (ASD), attention-deficit hyperactivity disorder (ADHD), and obsessive-compulsive disorder (OCD) [17, 18]. Although a broad range of differential mechanisms link altered CNS function with neurodevelopmental disorders, there are shared and overlapping similarities in behavioral presentation such as language/communication deficiencies, and repetitive or altered vigilance with attention-based tasks [19]. Additionally, rodent models of ASD/ADHD/OCD show similar behavioral endophenotypes such as impulsivity, hyperactivity, deficits in learning and memory, and repetition of behaviors [20, 21]. Although polygenic inheritance likely drives the etiology and core behavioral traits observed in ASD/ADHD/OCD, studies implicate alterations in GABAAR expression. Particularly, changes in the 15q11–13 chromosomal region have been linked to ASD which contains the GABRB3, GABRA5, and GABRG3 genes that encode for β3, α5, and γ3 subunits, respectively [22]. Additionally, alterations in the GABRB3 gene are associated with ASD [23]. Moreover, it has been reported that β3 expression [24] and mRNA for the various GABAAR subunits are decreased in autistic individuals, albeit in discrete brain regions [25, 26]. Rodent knock-out (KO) studies also suggest that GABAAR loss could be involved in neurodevelopmental disorders. For example, KO of α5-GABAARs results in ASD-like behaviors in mice, indicating a critical role for tonic inhibition [27, 28]. In line with clinical observations, β3 KO mice also display some endophenotypes such as reduced sociability, hyperactivity, seizure susceptibility, and deficits in attention, as well as learning and memory [29,30,31]. Lastly, alterations in grooming behavior are seen in a wide variety of ASD models [32] which can be bi-directionally regulated using GABAAR pharmacology [33]. Taken together, these observations suggest that altered GABAAR function could be involved in neurodevelopmental disorders. Therefore, further investigation into how molecular regulation of GABAergic inhibition contributes to endophenotypes in neurodevelopmental disorders is warranted.
Materials and methods
Animals
Shisa7 S405A knock-in mouse line was generated by CRISPR-mediated homologous recombination. Additional information can be found in the Supplementary Material. All animal handling was performed in accordance with animal protocols approved by the Institutional Animal Care and Use Committee (IACUC) at NIH/NINDS. Mice were housed and bred in a conventional vivarium with ad libitum access to food and water under a 12-h light/12-h dark cycle (ZT0 - “light on” at 0600, ZT12 - “lights off” at 1800). Time-pregnant mice at E17.5–18.5 were used for dissociated hippocampal neuronal culture. Mice of both sexes at P16–21 were used to prepare acute hippocampal slices for electrophysiology experiments. Adult male mice (2–3 months old) were used for behavioral experiments.
Dissociated hippocampal neuronal culture
Mice hippocampal neurons were prepared from E17.5–18.5 mice embryos as previously described [16]. For details, see Supplementary Material.
Electrophysiology
Electrophysiology recordings were done as previously described [15, 16]. For details, see Supplementary Material.
Immunocytochemistry
Immunocytochemistry was done as previously described [16, 34]. For details, see Supplementary Material.
Co-immunoprecipitation and Western blot
Co-immunoprecipitation and western blot were done as previously described [15, 16]. For details, see Supplementary Material.
Behavioral experiments
All behavioral experiments were conducted during the light cycle (“lights on” ZT0-ZT12; 0600–1800) unless otherwise specified. Prior to all behavioral experiments, male mice were allowed 60–90 min to acclimate to the behavioral room following transport from the vivarium. Animals were tracked during behavioral tests using AnyMaze software from an overhead camera system. Behavioral equipment was cleaned between subjects with 75% ethanol and allowed to dry for ~5 min between each behavioral trial. For details of T-maze, Novel object recognition, Nestlet shredding test, Marble burying test, Forced swim test, see Supplementary Material.
Y-maze
The Y-maze apparatus was constructed of clear acrylic and had three arms (height: 12.5 cm; length: 39 cm; width: 9 cm) 120 degrees apart. Mice were placed at the end of one arm and were allowed to explore the maze freely for 5 min. Alternation behavior was defined as consecutive entry into all three arms without repetition and was expressed as percentage of the total arm entries [35]. At the end of the test period, mice were placed back in their home cages and the maze wiped and dried before the next animal was run.
Open field test
Mice were allowed to freely explore an open field arena (41 × 41 × 31 cm) for 30 min and their movements were tracked during the test period and analyzed by AnyMaze software. Locomotion was operationally defined as the total distance traveled. Average speed was calculated using the following formula: Average speed = total travel distance [m] / mobility time [s].
Grooming behavior
Mice were allowed to habituate to an empty standard mouse housing cage for 10 min. Behavior was then recorded for a total of 10 min and the total amount of time spent grooming was quantified in seconds.
Homecage locomotor activity
Mice were individually housed in a standard home cage under familiar vivarium conditions for a 2-day acclimation period. Activity was then recorded for 24 h using PAS System (San Diego Instruments). Locomotion was defined as the number of beam interruptions.
Sleep deprivation
Sleep deprivation were done as previously described [16]. For details, see Supplementary Material.
Piezoelectric sleep recording
Sleep-wake activity was recorded using a piezoelectric monitoring system (Signal Solutions) as described previously [16]. For details, see Supplementary Material.
Statistical analysis
For all biochemical, cell biological and electrophysiological recordings, at least three independent experiments were performed (independent cultures, transfections or different mice). Statistical analysis was performed in GraphPad Prism 9.0 software. Normality distribution was tested by the Shapiro–Wilk test before carrying out a subsequent statistical test. Direct comparisons between two groups were made using either one-tailed/two-tailed Student’s t test or Mann–Whitney U test. Multiple comparisons were performed using one-way ANOVA, Kruskal-Wallis test or two-way ANOVA with corrections for multiple comparisons.
Results
Expression of Shisa7 S405A mutant reduces surface expression of α2-GABAARs
Given that Shisa7 promoted trafficking of synaptic GABAARs [15], we investigated whether Shisa7 S405, a site previously found to be a PKA phosphorylation substrate and important for extrasynaptic α5-GABAAR trafficking [16], is also critical for synaptic GABAAR trafficking. To determine the role of S405 phosphorylation, we created phospho-null (S to A) and phosphomimetic (S to D) mutants of Shisa7. Using immunocytochemistry, we observed that heterologous cells expressing α2β3γ2 GABAARs had increased surface α2 expression when co-transfected with Shisa7 or S405D mutant, but not with S405A mutant (Fig. 1A), indicating that the regulation of α2-GABAAR surface expression by Shisa7 is dependent on S405 phosphorylation. Consistently, co-expression of Shisa7 S405A in heterologous cells transfected with α2β3γ2 decreased GABA-evoked whole-cell currents (Fig. 1B). Considering that Shisa7 binds to GABAARs to promote synaptic trafficking [15], we examined whether S405 phosphorylation affected this interaction. In HEK293T cells expressing α2β3γ2 GABAARs, we found that α2-GABAARs were co-immunoprecipitated with Flag-tagged Shisa7, S405A and S405D mutants (Fig. 1C) suggesting regulation of GABAAR trafficking is not due to any binding deficit. Collectively, these data extend our previous observations that Shisa7 regulates both synaptic and extrasynaptic GABAAR trafficking to the cell surface [15, 16].
A Surface α2 expression is increased following co-transfection of Shisa7, but not with S405A mutant in HEK293T cells expressing α2β3γ2 (n = 17–24 cells, Kruskal–Wallis test with Dunnett’s multiple comparison test; Ctrl versus Shisa7, p = 0.0046; Shisa7 versus Shisa7 S405A, p = 0.0002; Ctrl versus Shisa7 S405D, p = 0.0046). B S405A significantly reduces GABA-evoked whole-cell currents compared to Shisa7 in HEK293T cells expressing α2β3γ2 (n = 11–16 cells, Kruskal–Wallis test with Dunnett’s multiple comparison test; Ctrl versus Shisa7, p = 0.0011; Shisa7 versus Shisa7 S405A, p = 0.041). C Shisa7, Shisa7 S405A and S405D were co-immunoprcipitated with α2 in HEK293T cells expressing α2β3γ2. Error bars indicate S.E.M. **p < 0.01; ***p < 0.001; ****p < 0.0001.
Shisa7 S405 is required for surface expression of GABAARs and GABAergic transmission in hippocampal neurons
To further corroborate the involvement of Shisa7 S405 in GABAAR trafficking and GABAergic transmission, we generated a Shisa7 S405A (KI) mouse line that is phospho-deficient at S405 (Fig. 2A, B). These mice breed normally and exhibit normal body weight when compared to both WT and Shisa7 KO mice (Fig. 2C, D). To examine whether hippocampal expression of Shisa7 and/or inhibitory synaptic markers in KI mice were altered, we first conducted Western blots using hippocampal lysates which demonstrated that Shisa7 expression, as well as vGAT and gephyrin, were not significantly altered in KI mice (Fig. 2E, F). After confirming this, we performed immunocytochemical experiments in hippocampal neuronal cultures to investigate the impact of the S405A mutation on GABAAR surface expression. We found that α1-, α2-, and α5-GABAAR surface expression were significantly reduced in KI neurons at somatic and dendritic domains (Fig. 3A–C), indicating that S405 phosphorylation critically impacts surface expression via regulation of both synaptic and extrasynaptic GABAAR trafficking. We further confirmed this observation by utilizing electrophysiological recordings which showed decreased GABA-evoked whole-cell currents in dissociated hippocampal cultures prepared from KI mice (Fig. 3D).
A Schematic of the Shisa7 protein with the WT S405 site in the intracellular region. The serine encoded by UCC (green) in the WT was mutated to an alanine encoded by GCC (purple). B Mice carrying the S405A allele were genotyped by Sanger-sequencing of a 527 bp amplified fragment generated by PCR. As indicated in the dashed box: for WT, only one peak of nucleotide T was observed; for S405A heterozygous (HET), both peaks of nucleotide T and G were overlapped; for S405A homozygous (KI), only one peak of nucleotide G was observed. C Percentage of weaned pups of WT, HET, and KI genotype born from S405A-HET and S405A-HET breedings. D Shisa7 S405A KI mice display normal body weight in the first month after birth (n = 9 mice). E Representative western blot of Shisa7, vGAT and Gephyrin from WT, KI and KO hippocampal lysates. Arrow indicates the band corresponding to Shisa7. F Summary graphs showing there is no difference of Shisa7, vGAT or Gephyrin expression among WT, KI and KO mice (n = 3 independent experiments, one-way ANOVA).
A Surface α1 expression in cultured hippocampal neurons from KI mice is reduced (n = 13–17 cells, somatic: Mann–Whitney U test, p = 0.0076; dendritic: Mann–Whitney U test, p = 0.0048). B Surface α2 expression in cultured hippocampal neurons from KI mice is reduced (n = 20–31 cells, somatic: t test, p = 0.0184; dendritic: t test, p = 0.0001). C Surface α5 in cultured hippocampal neurons from KI mice is reduced (n = 20–32 cells, somatic: t test, p = 0.0154; dendritic: Mann–Whitney U test, p = 0.0004). D GABA-evoked whole-cell currents in cultured hippocampal neurons from KI mice are decreased (WT, n = 12 cells; KI, n = 19 cells, t test; p = 0.0049). Error bars indicate S.E.M. *p < 0.05; **p < 0.01; ***p < 0.001.
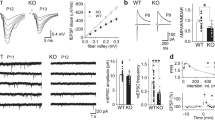
Next, we evaluated whether both GABAAR-mediated synaptic and tonic currents were altered in dissociated hippocampal cultures prepared from KI mice. Although no significant alterations in the amplitude of miniature inhibitory postsynaptic currents (mIPSCs) were observed, there was a significant decrease in mIPSC frequency, as well as diminished tonic currents in KI hippocampal neurons (Fig. 4A–D). We further investigated whether mIPSCs and tonic currents were affected in CA1 pyramidal neurons in acute hippocampal slices prepared from KI mice. Congruent with the evidence from dissociated hippocampal cultures, we observed a reduction in mIPSC frequency (Fig. 4E, F) and tonic currents in KI mice (Fig. 4I). However, there was no significant difference in mIPSC amplitude and decay time constant (Fig. 4G, H), nor were AMPAR-mediated mEPSCs affected (Fig. S1A–C). It was possible that baseline electrical noises might have masked those mIPSC events with low amplitudes, and thus a potential effect of Shisa7 S405A KI on mIPSC amplitude might not be revealed under the mIPSC recording configuration. To this end, we recorded spontaneous IPSCs (sIPSCs) in CA1 pyramidal neurons in acute hippocampal slices in the absence of TTX. The amplitude of sIPSCs was higher than that of mIPSCs (Fig. 4C and S1E). We found that both frequency and amplitude of sIPSCs were diminished in KI mice (Fig. S1D, E). Additionally, there were no changes of paired pulse ratio (PPR) of IPSCs (Fig. S1F, G), suggesting that the probability of presynaptic GABA release is not altered in hippocampal neurons in KI mice. Taken together, these data show that S405 phosphorylation is critical for GABAAR trafficking and GABAergic transmission in hippocampal neurons.
A Representative mIPSC traces from neuronal cultures. B–C mIPSC frequency is diminished in KI mice with no change in mIPSC amplitude (WT, n = 10 cells; KI, n = 13 cells, Mann–Whitney U test; p = 0.0024). D Tonic inhibition is reduced in neuronal cultures from KI mice (WT, n = 10 cells; KI, n = 10 cells, t test; p = 0.0233). E Representative mIPSC traces from CA1 neurons in acute hippocampal slices. F, G mIPSC frequency is decreased onto CA1 neurons in KI mice with no change in mIPSC amplitude (WT, n = 9 cells; KI, n = 8 cells, Mann–Whitney U test; p = 0.0099). H No significant changes in decay time constant in KI mice (WT, n = 9 cells; KI, n = 8 cells, Mann–Whitney U test). I Tonic inhibition is reduced in CA1 neurons from KI mice (WT, n = 9 cells; KI, n = 8 cells, t test; p = 0.024). J iLTP is impaired in CA1 neurons from Shisa7 S405A KI mice. Bath application of NMDA (3 min, 20 μM) increases mIPSC amplitude for up to 30 min compared to baseline in WT mice, but not KI mice. K Representative traces of mIPSCs from CA1 neurons in acute hippocampal slices at baseline and then 30 min following bath-application of NMDA. L Bath application of NMDA (3 min, 20 μM) increases mIPSC amplitude in WT mice only (WT, n = 8 cells; KI, n = 6 cells, two-way ANOVA, F 16, 204 = 2.324, p = 0.0036 with Sidak’s multiple comparison test; WT a versus WT b, p = 0.0005). Error bars indicate S.E.M. *p < 0.05; **p < 0.01; ***p < 0.001.
iLTP is impaired in KI mice
Regulation of GABAergic plasticity is dependent on the surface abundance of GABAARs and thus trafficking processes can affect the expression of iLTP [36, 37]. To this end, we adopted a strategy [38] (chem-iLTP) to assess whether iLTP was altered in hippocampal neurons from KI mice. Specifically, following transient application of NMDA (3 min, 20 μM) in the bath perfusate, we examined mIPSC amplitude in a time-dependent manner up to 30 min after NMDA exposure in CA1 pyramidal neurons from acute hippocampal slices. In agreement with previous studies [38,39,40], brief NMDA exposure to hippocampal neurons was sufficient to induce a persistent increase in mIPSC amplitude over a 30-min period in WT mice (Fig. 4J). Strikingly, mIPSC amplitude in CA1 pyramidal neurons in KI mice 30 min post-NMDA application did not significantly increase past baseline levels (Fig. 4K, L). These findings suggest that Shisa7 S405 phosphorylation is a requirement for activity-dependent trafficking of GABAARs and iLTP in CA1 pyramidal neurons.
KI mice display locomotor hyperactivity, increased grooming and impaired sleep homeostasis
Given that KI mice displayed impaired inhibitory transmission and iLTP, we examined the consequences of Shisa7 S405A mutation on animal behaviors. We first investigated whether KI mice had any deficits in learning and memory using three behavioral paradigms. In order to assess short-term spatial working memory, we utilized the Y-maze and T-maze. For Y-maze, mice were allowed to freely explore for 5 min and spontaneous alternation behavior was defined as consecutive entry into all three arms without repetition [35]. We observed that KI mice exhibited no impairment of spontaneous alternation behavior compared to WT mice (Fig. 5A). As with Y-maze, WT and KI mice also exhibited similar spontaneous alternation behavior in T-maze (Fig. 5B). Next, we assessed object recognition memory using the Novel Object Recognition (NOR) test. During the training session, no preference for one of the two objects was observed in both WT and KI mice (Fig. S2A). Following 1 h or 24 h delay, mice underwent a test session. In the test session, WT and KI mice performed similarly, spending more time with the novel object compared to the familiar object (Fig. S2A).
A KI mice have increased number of arm entries (WT, n = 10 mice; KI, n = 11 mice, t test; p = 0.0066) and total travel distance (WT, n = 10 mice; KI, n = 11 mice, t test; p = 0.0085), but no change in spontaneous alternation among the arms in the Y maze (WT, n = 10 mice; KI, n = 11 mice, t test). B KI mice have no change in spontaneous alternation in the T maze (n = 9 mice, t test). C In OFT, KI mice have increased travel distance (Travel distance in each of the 10-min test periods, two-way ANOVA, F 1, 16 = 5.962, p = 0.0266; Total travel distance: WT, n = 9; KI, n = 9, t test; p = 0.0265) and decreased immobility (WT, n = 9; KI, n = 9, t test; p = 0.0094), but no change in the average speed (WT, n = 9; KI, n = 9, Mann–Whitney U test) and the time spent in the center (WT, n = 9; KI, n = 9, t test) compared to WT mice. D KI mice display more activity (# of beam interruptions) in the homecage driven by enhanced activity during “lights off” (ZT12–24); WT, n = 8 mice; KI, n = 8 mice, two-way ANOVA, F 2, 28 = 5.329, p = 0.0109 with Sidak’s multiple comparison test; Dark: WT versus Dark: KI, p = 0.0191; Total: WT versus Total: KI, p = 0.0392). E KI mice have increased grooming behavior compared to WT (WT, n = 8; KI, n = 8, Mann–Whitney U test; p = 0.0103). F Sleep behavior is not different between WT and KI mice (WT, n = 9 mice; KI, n = 9 mice, two-way ANOVA with Sidak’s multiple comparison test). G KI mice display impaired sleep homeostasis. Left, sleep-wake pattern when the WT and KI mice were subjected to sleep deprivation (SD) for 6 h (ZT0–6). Right, Summary of the percentage of sleep during the two selected periods. (Baseline, n = 8 mice; SD, n = 8 mice, two-way ANOVA, F 1, 28 = 6.598, p = 0.0158 with Sidak’s multiple comparison test; ZT12–24: p = 0.04). Error bars indicate S.E.M. *p < 0.05; **p < 0.01.
From these assays, KI mice did not display any noticeable deficits in learning and memory. However, we observed that KI mice had increased arm entries and total distance traveled during the Y-maze test (Fig. 5A), suggesting that KI mice displayed locomotor hyperactivity. To confirm this, we measured general locomotor activity using the Open Field Test (OFT) and homecage activity recordings. During the 30-min OFT, KI mice exhibited larger travel distance and less immobility without changes in travel speed and the time spent in the center compared to WT mice (Fig. 5C). In the 24-h homecage activity recordings, KI mice also traveled greater distances as indicated by increased beam interruptions compared to WT mice; although, the increased movement seemed to be more prevalent during “lights off” (ZT12–24; Fig. 5D). Overall, these behavioral data indicate that KI mice have a hyperactive locomotor phenotype. Since hyperactivity has been observed in animal models of ASD/ADHD/OCD [20, 21], we proceeded to further characterize KI mice by assessing repetitive and compulsive behaviors using Nestlet Shredding Test (NST), grooming time, and Marble Burying Test (MBT) [36, 41]. Although KI mice performed similarly to WT mice in the NST and MBT (Fig. S2B, C), KI mice spent a greater amount of time grooming during a 10-min period (Fig. 5E). Given the co-morbidity of depression [42] with ASD/ADHD/OCD, we also assessed pro-depressive coping styles (learned helplessness) using the Forced Swim Test (FST). We observed that the FST yielded no difference between WT and KI mice with respect to immobility time (Fig. S2D).
Considering that sleep disturbances are also often comorbid with ASD/ADHD/OCD [43] and that our previous study indicated that Shisa7 KO mice have decreased sleep times [16], we monitored sleep behavior using a piezoelectric monitoring system [44, 45]. We first recorded baseline sleep in normal conditions, but did not observe a decrease in KI mice (Fig. 5F). Because sleep is a complex physiological process, we then utilized sleep deprivation to examine whether S405 affected recovery sleep. Mice were sleep-deprived for 6 h starting from the beginning of the light-on period, and recovery sleep was then measured during the subsequent 18 h. Both WT and KI mice showed compensatory sleep rebound after 6 h sleep deprivation (Fig. S3A, B). However, there was significantly less recovery sleep in KI mice during “lights off” (ZT12–24), but not for ZT6–12 (Fig. 5G), indicating impaired sleep homeostasis in KI mice.
In sum, hippocampal neurons from KI mice show impaired GABAAR trafficking and loss of NMDAR-dependent iLTP. Consistent with GABAergic signaling deficits, KI mice show endophenotypes of neurodevelopmental disorders, specifically locomotor hyperactivity, increased grooming behavior and impaired sleep homeostasis.
Discussion
GABAARs are critical for the regulation of neuronal excitability across the CNS and deficits in GABAAR-mediated signaling are associated with a myriad of psychiatric conditions [5,6,7]. Therefore, it remains essential to understand the mechanistic processes by which GABAARs are trafficked to the synaptic and extrasynaptic compartments. In this study, we have expanded upon our previous observations regarding the role of Shisa7 in GABAAR trafficking [15, 16] by providing specific insight into the role for S405 phosphorylation. We have demonstrated that S405 phosphorylation is not only instrumental for trafficking of extrasynaptic GABAARs [16], but is also important for trafficking of synaptic GABAARs. Importantly, we observed that GABAAR trafficking deficits in Shisa7 S405A KI mice not only resulted in diminished GABAergic transmission, but also blunted NMDAR-mediated iLTP. Lastly, we identified specific endophenotypes in Shisa7 KI mice which are associated with ASD/ADHD/OCD. Collectively, this study extends our knowledge regarding the role of Shisa7-dependent regulation of both synaptic and extrasynaptic GABAARs and reveals that the behavioral consequences of not allowing Shisa7 S405 to be phosphorylated are locomotor hyperactivity, longer grooming periods and impaired sleep homeostasis.
GABAARs control neuronal excitability due to their high chloride permeability which influxes and hyperpolarizes neurons upon activation. However, GABAergic signaling is diverse and is largely dependent on subunit composition and receptor localization [1, 2]. In addition, the strength of GABAergic inhibition is directly related to GABAAR abundance at the cell surface. In the case of GABAAR-associated transmembrane proteins, they all have been shown to play a critical role in inhibitory neurotransmission [10, 11]. In LH4 KO mice, recordings from hippocampal CA1 pyramidal neurons show decreased mIPSCs, indicating that LH4 is poised to directly affect the strength of synaptic inhibition [46, 47]. Given that Clptm1 restricts forward trafficking of both synaptic and extrasynaptic GABAARs, overexpression and knockdown of Clptm1 diminished and enhanced both mIPSCs and tonic currents, respectively [48]. We previously showed that KO of Shisa7 can diminish both phasic [15] and tonic inhibition [16] in hippocampal neurons. Since we identified S405 phosphorylation as a regulator of tonic inhibition [16], we explored whether phosphorylation of this residue also impacted phasic inhibition in hippocampal neurons. Using a newly generated KI mouse line, we observed a reduction in both phasic and tonic inhibition in hippocampal neurons. These decreases in GABAergic inhibition are attributed at least partly to diminished surface expression of α1-, α2- and α5-GABAARs. Additionally, these data recapitulate our previous findings that Shisa7 S405 is indeed critical for tonic currents [16] and provide further evidence that prohibiting S405 phosphorylation consequently results in diminished mIPSCs and sIPSCs. We did not, however, see any alteration in the amplitude and frequency of AMPAR-mediated mEPSCs. From a network perspective, this seems to suggest that only the inhibitory component of synaptic transmission is principally affected and there is no compensatory action at excitatory synapses onto CA1 pyramidal neurons. Therefore, in terms of the balance in the coordination of excitatory to inhibitory inputs, the diminished effect of inhibition potentially shifts network activity to favor excitation. Collectively, these data suggest that Shisa7 S405 phosphorylation has a comprehensive role in controlling GABAergic inhibition that encompasses both phasic and tonic inhibition.
Synaptic plasticity has been proposed to be the cellular basis of learning and memory that involves diverse processes that lead to either strengthening or weakening of synapses. Although the molecular mechanisms underlying the regulation of excitatory synaptic plasticity have been extensively studied [49, 50], much less is known about the regulation of inhibitory synaptic plasticity. Accumulating studies have revealed a critical role for NMDARs in the regulation of inhibitory synapse development and function [34, 51,52,53], and iLTP induction [38,39,40, 54]. It has been shown that NMDAR-mediated iLTP requires synaptic recruitment of gephyrin, CaMKII activation, and synaptic insertion of β2/3-containing GABAARs [38, 40, 54]. Recent studies have also identified a number of transmembrane accessory proteins for GABAARs [10, 11]. However, the role of these transmembrane proteins in iLTP remained unknown. We have now demonstrated a critical role of Shisa7 in the regulation of iLTP. Specifically, in Shisa7 S405A KI mice, iLTP in hippocampal CA1 pyramidal neurons was blunted, indicating that synaptic recruitment of GABAARs during iLTP depends on a Shisa7 S405 phosphorylation process. Since Shisa7 S405 is the substrate of PKA, our data also suggest a potential role of PKA in iLTP. Currently, how Shisa7 S405 dependent pathway functionally interacts with gephyrin and CaMKII dependent processes to regulate iLTP remains unknown. Additionally, it is worth noting that our iLTP experiment was measured through recording mIPSCs from CA1 pyramidal neurons. Therefore, it remains to be determined whether Shisa7 can regulate iLTP in a synapse-specific manner. Indeed, it has recently been reported that the expression of iLTP in hippocampal and cortical pyramidal neurons is input-dependent [54, 55]. It will be important to understand how Shisa7 regulates GABAergic plasticity in a synapse-specific context.
GABAARs are critical in the regulation of neural circuit function of the CNS [8, 56, 57]. Given our findings of impaired inhibitory transmission and iLTP in KI mice reported here, we performed a battery of behavioral tests to examine the consequences of Shisa7 S405A mutation on animal behaviors. We first investigated whether learning and memory behaviors were altered in KI mice. Interestingly, although KI mice did not display any deficits in both the Y-maze, T-maze and NOR tests, these mice exhibited increased locomotor activity in the Y-maze. We further confirmed this increased locomotor activity using the OFT and by examining homecage activity. Given that hyperactivity has been associated with ASD/OCD/ADHD [20, 21], we utilized behavioral paradigms to determine whether our KI mouse line had any additional endophenotypes associated with these neurodevelopmental disorders. Although we observed no difference in nestlet shredding or marble burying, two common assays for repetitive animal behaviors [41], we did observe a greater propensity to groom in KI mice compared to WT. This intriguing finding is in line with pharmacological experiments examining grooming behavior in that antagonism of GABAARs has been shown to precipitate greater grooming behavior [33]. Therefore, deficits in Shisa7-dependent trafficking of GABAARs could be impacting discrete brain regions whereby GABAARs influence grooming behaviors [33]. Additionally, clinical evidence has suggested that individuals afflicted with ASD/OCD/ADHD are more likely to develop mood disorders, such as depression [42]. Indeed, animal models of ASD/OCD/ADHD have also been shown to recapitulate depressive-like behaviors, such as increased immobility in the FST [58]. However, we did not see any differences in immobility time from KI mice compared to WT, demonstrating that these animals do not display any pro-depressive behaviors. Lastly, because sleep disturbances are often comorbid with many neurodevelopmental disorders and due to our previous observations that tonic inhibitory currents in hippocampal neurons differ based on sleep-wake states [16], we monitored sleep behavior using a piezoelectric monitoring system [44, 45]. Different from our previous study which showed decreased sleep in Shisa7 KO mice [16], we did not observe this effect in KI mice, although the impaired sleep homeostasis was observed. Given that sleep is a complex process, it is possible that Shisa7 is dynamically regulated based on different internal sleep states. One major difference between our two studies is that the former had no Shisa7 present throughout the development whereas the current study here has Shisa7, but with the S405 site rendered incapable of being phosphorylated. Although we note that Shisa7 S405 is important for trafficking of synaptic and extrasynaptic GABAARs, it is possible that S405-independent mechanisms are involved in Shisa7 regulation of sleep behavior in normal conditions. Currently, the mechanisms underlying the role of Shisa7 S405 in animal behavior remain unknown. It is possible that cell-type specific functions of Shisa7 S405 may contribute differentially and/or in a context-specific manner. Although it has been reported that Shisa7 is highly expressed in hippocampal pyramidal neurons, interneurons within the hippocampus also show Shisa7 expression, but the expression patterns differ based on the interneuron subtype [59, 60]. We also cannot discount the notion that these behavioral alterations are due to compensatory changes in circuits due to global loss of Shisa7 S405 phosphorylation throughout the development. The use of region- or cell-type- specific manipulation of Shisa7 S405 phosphorylation will be valuable in order to fully understand the contributions of Shisa7 S405 to behavior in a circuit-specific context.
In summary, we have expanded on our previous studies further elucidating the importance of the Shisa7-dependent trafficking of synaptic and extrasynaptic GABAARs in the regulation of GABAergic transmission. Phosphorylation of S405 is not only crucial for the ability of Shisa7 to traffic extrasynaptic α5-GABAARs, but also for synaptic α1/α2-GABAARs. Critically, we have discovered that NMDAR-dependent iLTP in hippocampal neurons requires Shisa7 S405 phosphorylation. Moreover, we find that our Shisa7 S405A KI mouse model produces enhanced grooming, locomotor hyperactivity, and deficits in sleep homeostasis, suggesting that impaired Shisa7 phosphorylation-dependent trafficking of GABAARs could be involved in neurodevelopmental disorders.
References
Sieghart W, Savic MM. International union of basic and clinical pharmacology. CVI: GABAA Receptor Subtype- and Function-selective Ligands: Key Issues in Translation to Humans. Pharm Rev. 2018;70:836–78.
Olsen RW, Sieghart W. GABA A receptors: subtypes provide diversity of function and pharmacology. Neuropharmacology. 2009;56:141–8.
Farrant M, Nusser Z. Variations on an inhibitory theme: phasic and tonic activation of GABA(A) receptors. Nat Rev Neurosci. 2005;6:215–29.
Jacob TC, Moss SJ, Jurd R. GABA(A) receptor trafficking and its role in the dynamic modulation of neuronal inhibition. Nat Rev Neurosci. 2008;9:331–43.
Luscher B, Shen Q, Sahir N. The GABAergic deficit hypothesis of major depressive disorder. Mol Psychiatry. 2011;16:383–406.
Mohler H. GABAA receptors in central nervous system disease: anxiety, epilepsy, and insomnia. J Recept Signal Transduct Res. 2006;26:731–40.
Treiman DM. GABAergic mechanisms in epilepsy. Epilepsia. 2001;42:8–12.
Hines RM, Davies PA, Moss SJ, Maguire J. Functional regulation of GABAA receptors in nervous system pathologies. Curr Opin Neurobiol. 2012;22:552–8.
Maher MP, Matta JA, Gu S, Seierstad M, Bredt DS. Getting a Handle on Neuropharmacology by Targeting Receptor-Associated Proteins. Neuron. 2017;96:989–1001.
Han W, Shepard RD, Lu W. Regulation of GABAARs by Transmembrane Accessory Proteins. Trends Neurosci. 2021;44:152–65.
Castellano D, Shepard RD, Lu W. Looking for Novelty in an “Old” Receptor: Recent Advances Toward Our Understanding of GABAARs and Their Implications in Receptor Pharmacology. Front Neurosci. 2020;14:616298.
Nakamura Y, Morrow DH, Modgil A, Huyghe D, Deeb TZ, Lumb MJ, et al. Proteomic Characterization of Inhibitory Synapses Using a Novel pHluorin-tagged gamma-Aminobutyric Acid Receptor, Type A (GABAA), alpha2 Subunit Knock-in Mouse. J Biol Chem. 2016;291:12394–407.
Farrow P, Khodosevich K, Sapir Y, Schulmann A, Aslam M, Stern-Bach Y, et al. Auxiliary subunits of the CKAMP family differentially modulate AMPA receptor properties. eLife 2015;4:e09693. https://doi.org/10.7554/eLife.09693.
Schmitz LJM, Klaassen RV, Ruiperez-Alonso M, Zamri AE, Stroeder J, Priyanka R-R. et al. The AMPA receptor-associated protein Shisa7 regulates hippocampal synaptic function and contextual memory. eLife. 2017;6:e24192, https://doi.org/10.7554/eLife.24192.
Han W, Li J, Pelkey KA, Pandey S, Chen X, Wang YX, et al. Shisa7 is a GABAA receptor auxiliary subunit controlling benzodiazepine actions. Science. 2019;366:246–50.
Wu K, Han W, Tian Q, Li Y, Lu W. Activity- and sleep-dependent regulation of tonic inhibition by Shisa7. Cell Rep. 2021;34:108899.
Gatto CL, Broadie K Genetic controls balancing excitatory and inhibitory synaptogenesis in neurodevelopmental disorder models. Front Synaptic Neurosci. 2010;2:4.
Lee E, Lee J, Kim E. Excitation/Inhibition imbalance in animal models of autism spectrum disorders. Biol Psychiatry. 2017;81:838–47.
Coghlan S, Horder J, Inkster B, Mendez MA, Murphy DG, Nutt DJ. GABA system dysfunction in autism and related disorders: from synapse to symptoms. Neurosci Biobehav Rev. 2012;36:2044–55.
Kazdoba TM, Leach PT, Yang M, Silverman JL, Solomon M, Crawley JN. Translational mouse models of autism: advancing toward pharmacological therapeutics. Curr Top Behav Neurosci. 2016;28:1–52.
Ahmari SE. Using mice to model Obsessive Compulsive Disorder: From genes to circuits. Neuroscience. 2016;321:121–37.
Tager-Flusberg H, Joseph R, Folstein S. Current directions in research on autism. Ment Retard Dev Disabil Res Rev. 2001;7:21–9.
Buxbaum JD, Silverman JM, Smith CJ, Greenberg DA, Kilifarski M, Reichert J, et al. Association between a GABRB3 polymorphism and autism. Mol Psychiatry. 2002;7:311–6.
Samaco RC, Hogart A, LaSalle JM. Epigenetic overlap in autism-spectrum neurodevelopmental disorders: MECP2 deficiency causes reduced expression of UBE3A and GABRB3. Hum Mol Genet. 2005;14:483–92.
Fatemi SH, Reutiman TJ, Folsom TD, Thuras PD. GABA(A) receptor downregulation in brains of subjects with autism. J Autism Developmental Disord. 2009;39:223–30.
Fatemi SH, Reutiman TJ, Folsom TD, Rustan OG, Rooney RJ, Thuras PD. Downregulation of GABAA receptor protein subunits alpha6, beta2, delta, epsilon, gamma2, theta, and rho2 in superior frontal cortex of subjects with autism. J Autism Developmental Disord. 2014;44:1833–45.
Mesbah-Oskui L, Penna A, Orser BA, Horner RL. Reduced expression of alpha5GABAA receptors elicits autism-like alterations in EEG patterns and sleep-wake behavior. Neurotoxicol Teratol. 2017;61:115–22.
Zurek AA, Kemp SW, Aga Z, Walker S, Milenkovic M, Ramsey AJ, et al. alpha5GABAA receptor deficiency causes autism-like behaviors. Ann Clin Transl Neurol. 2016;3:392–8.
Culiat CT, Stubbs LJ, Montgomery CS, Russell LB, Rinchik EM. Phenotypic consequences of deletion of the gamma 3, alpha 5, or beta 3 subunit of the type A gamma-aminobutyric acid receptor in mice. Proc Natl Acad Sci USA. 1994;91:2815–8.
DeLorey TM, Sahbaie P, Hashemi E, Homanics GE, Clark JD. Gabrb3 gene deficient mice exhibit impaired social and exploratory behaviors, deficits in non-selective attention and hypoplasia of cerebellar vermal lobules: a potential model of autism spectrum disorder. Behavioural Brain Res. 2008;187:207–20.
Homanics GE, DeLorey TM, Firestone LL, Quinlan JJ, Handforth A, Harrison NL, et al. Mice devoid of gamma-aminobutyrate type A receptor beta3 subunit have epilepsy, cleft palate, and hypersensitive behavior. Proc Natl Acad Sci USA. 1997;94:4143–8.
Gandhi T, Lee CC. Neural mechanisms underlying repetitive behaviors in rodent models of autism spectrum disorders. Front Cell Neurosci. 2020;14:592710.
Kalueff AV, Stewart AM, Song C, Berridge KC, Graybiel AM, Fentress JC. Neurobiology of rodent self-grooming and its value for translational neuroscience. Nat Rev Neurosci. 2016;17:45–59.
Wu K, Castellano D, Tian Q, Lu W. Distinct regulation of tonic GABAergic inhibition by NMDA receptor subtypes. Cell Rep. 2021;37:109960.
Hughes RN. The value of spontaneous alternation behavior (SAB) as a test of retention in pharmacological investigations of memory. Neurosci Biobehav Rev. 2004;28:497–505.
Luscher B, Fuchs T, Kilpatrick CL. GABAA receptor trafficking-mediated plasticity of inhibitory synapses. Neuron. 2011;70:385–409.
Castillo PE, Chiu CQ, Carroll RC. Long-term plasticity at inhibitory synapses. Curr Opin Neurobiol. 2011;21:328–38.
Marsden KC, Beattie JB, Friedenthal J, Carroll RC. NMDA receptor activation potentiates inhibitory transmission through GABA receptor-associated protein-dependent exocytosis of GABA(A) receptors. J Neurosci: Off J Soc Neurosci. 2007;27:14326–37.
Wiera G, Lebida K, Lech AM, Brzdak P, Van Hove I, De Groef L, et al. Long-term plasticity of inhibitory synapses in the hippocampus and spatial learning depends on matrix metalloproteinase 3. Cellular and molecular life sciences: CMLS. 2020.
Petrini EM, Ravasenga T, Hausrat TJ, Iurilli G, Olcese U, Racine V, et al. Synaptic recruitment of gephyrin regulates surface GABAA receptor dynamics for the expression of inhibitory LTP. Nat Commun. 2014;5:3921.
Angoa-Perez M, Kane MJ, Briggs DI, Francescutti DM, Kuhn DM. Marble burying and nestlet shredding as tests of repetitive, compulsive-like behaviors in mice. J Vis Exp. 2013;82:50978.
Magnuson KM, Constantino JN. Characterization of depression in children with autism spectrum disorders. J Dev Behav Pediatr. 2011;32:332–40.
Gisbert Gustemps L, Lugo Marin J, Setien Ramos I, Ibanez Jimenez P, Romero Santo-Tomas O, Jurado Luque MJ, et al. Sleep disturbances in autism spectrum disorder without intellectual impairment: relationship with executive function and psychiatric symptoms. Sleep Med. 2021;83:106–14.
Mang GM, Nicod J, Emmenegger Y, Donohue KD, O’Hara BF, Franken P. Evaluation of a piezoelectric system as an alternative to electroencephalogram/ electromyogram recordings in mouse sleep studies. Sleep. 2014;37:1383–92.
Yaghouby F, Donohue KD, O’Hara BF, Sunderam S. Noninvasive dissection of mouse sleep using a piezoelectric motion sensor. J Neurosci Methods. 2016;259:90–100.
Davenport EC, Pendolino V, Kontou G, McGee TP, Sheehan DF, Lopez-Domenech G, et al. An essential role for the tetraspanin LHFPL4 in the cell-type-specific targeting and clustering of synaptic GABAA receptors. Cell Rep. 2017;21:70–83.
Yamasaki T, Hoyos-Ramirez E, Martenson JS, Morimoto-Tomita M, Tomita S. GARLH family proteins stabilize GABAA receptors at synapses. Neuron. 2017;93:1138–52 e6.
Ge Y, Kang Y, Cassidy RM, Moon KM, Lewis R, Wong ROL, et al. Clptm1 limits forward trafficking of GABAA receptors to scale inhibitory synaptic strength. Neuron. 2018;97:596–610 e8.
Diering GH, Huganir RL. The AMPA receptor code of synaptic plasticity. Neuron. 2018;100:314–29.
Huganir RL, Nicoll RA. AMPARs and synaptic plasticity: the last 25 years. Neuron. 2013;80:704–17.
Gu X, Zhou L, Lu W. An NMDA receptor-dependent mechanism underlies inhibitory synapse development. Cell Rep. 2016;14:471–8.
Horn ME, Nicoll RA. Somatostatin and parvalbumin inhibitory synapses onto hippocampal pyramidal neurons are regulated by distinct mechanisms. Proc Natl Acad Sci USA. 2018;115:589–94.
Gu X, Lu W. Genetic deletion of NMDA receptors suppresses GABAergic synaptic transmission in two distinct types of central neurons. Neurosci Lett. 2018;668:147–53.
Chiu CQ, Martenson JS, Yamazaki M, Natsume R, Sakimura K, Tomita S, et al. Input-Specific NMDAR-Dependent Potentiation of Dendritic GABAergic Inhibition. Neuron. 2018;97:368–77 e3.
Udakis M, Pedrosa V, Chamberlain SEL, Clopath C, Mellor JR. Interneuron-specific plasticity at parvalbumin and somatostatin inhibitory synapses onto CA1 pyramidal neurons shapes hippocampal output. Nat Commun. 2020;11:4395.
Braat S, Kooy RF. The GABAA receptor as a therapeutic target for neurodevelopmental disorders. Neuron. 2015;86:1119–30.
Brickley SG, Mody I. Extrasynaptic GABA(A) receptors: their function in the CNS and implications for disease. Neuron. 2012;73:23–34.
Commons KG, Cholanians AB, Babb JA, Ehlinger DG. The Rodent forced swim test measures stress-coping strategy, not depression-like behavior. ACS Chem Neurosci. 2017;8:955–60.
Paul A, Crow M, Raudales R, He M, Gillis J, Huang ZJ. Transcriptional architecture of synaptic communication delineates GABAergic neuron identity. Cell. 2017;171:522–39 e20.
Zeisel A, Munoz-Manchado AB, Codeluppi S, Lonnerberg P, La Manno G, Jureus A, et al. Brain structure. Cell types in the mouse cortex and hippocampus revealed by single-cell RNA-seq. Science. 2015;347:1138–42.
Acknowledgements
We are grateful to all members from the Lu laboratory for critical comments on the manuscript. We thank Daniel Abebe at NIH/NICHD for assistance with behavioral tests.
Funding
This work was supported by the NIH/NINDS Intramural Research Program (to WL), the NIH/NEI Intramural Research Program (to LD), Postdoctoral Fellowship from the NIH Center on Compulsive Behaviors (to RDS) and NINDS Diversity Training Fellowship (to DC).
Author information
Authors and Affiliations
Contributions
KW, RDS, and WL designed the project, and WL supervised the project. KW performed imaging and electrophysiological experiments. KW and WH performed biochemical experiments. KW and RDS performed behavioral assays. DC recorded GABA-evoked currents in HEK293T cells and neurons. QT performed neuronal cultures. LD generated Shisa7 S405A mutant mice. KW, RDS, and WL wrote the manuscript, and all authors read and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Wu, K., Shepard, R.D., Castellano, D. et al. Shisa7 phosphorylation regulates GABAergic transmission and neurodevelopmental behaviors. Neuropsychopharmacol. 47, 2160–2170 (2022). https://doi.org/10.1038/s41386-022-01334-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41386-022-01334-0