Abstract
Cocaine is thought to be more addictive when it reaches the brain rapidly. We predicted that variation in the speed of cocaine delivery influences the likelihood of addiction in part by determining the risk of relapse after abstinence. Under an intermittent-access schedule, rats pressed a lever for rapid (injected over 5 s) or slower (90 s) intravenous cocaine injections (0.5 mg/kg/injection). Control rats self-administered food pellets. A tone-light cue accompanied each self-administered reward. The 5s- and 90s-rats took a similar average amount of cocaine. One or 45 days after withdrawal from cocaine/forced abstinence, lever-pressing behaviour was extinguished during a 6-h session. Immediately thereafter, cue- or cocaine (10 mg/kg, i.p.)-induced reinstatement was assessed for 1 h. One or 45 days after withdrawal, only 5s-rats showed significant cocaine-induced reinstatement of reward-seeking behaviour. In both cocaine groups, cue-induced reinstatement behaviour was more pronounced after 45 days than after 1 day of withdrawal from cocaine, indicating incubation of conditioned drug craving. However, cue-induced reinstatement after extended abstinence was greatest in the 5s-rats. Brain-derived neurotrophic factor (BDNF) activity in the brain regulates reinstatement behaviour. Thus, 24 h after reinstatement tests, we measured BDNF protein concentrations in mesocorticolimbic regions. Only 5s-rats showed time-dependent increases in BDNF concentrations in the prelimbic cortex, nucleus accumbens core and ventral tegmental area after withdrawal from cocaine (day 45 > day 1). Thus, rapidly rising brain cocaine levels might facilitate addiction by evoking changes in the brain that intensify drug craving after abstinence, and these changes persist long after the last bout of cocaine use.
Similar content being viewed by others
Introduction
Many factors influence the risk of developing drug addiction. Individual and environmental factors have been studied extensively in this context [1, 2]. Pharmacokinetic variables—including how much, how often and how fast a drug reaches the brain—are also decisive in making the transition to addiction [3, 4]. For example, clinical observations are clear; when drugs of abuse reach the brain rapidly, addiction is more likely to develop. This is one reason why smoking or injecting cocaine intravenously is associated with a greater abuse liability and a greater propensity for drug addiction compared to intranasal use [5, 6].
Animal studies show that the rate of drug delivery determines the susceptibility to develop behavioural effects relevant to addiction. In much of this work, cocaine is used as a model drug. The findings show that injecting cocaine intravenously over 5 versus 90–100 s promotes psychomotor sensitization [7,8,9], escalation of drug intake [10, 11], and incentive motivation for cocaine [9, 12, 13]. These effects are likely mediated by differences in the temporal kinetics with which cocaine reaches its sites of action in the brain, rather than by differences in achieved dose. Indeed, varying infusion duration between 5 and 100 s produces differences in the rate of rise of cocaine concentrations in the brain, without producing large effects on peak concentrations [8, 9, 14].
The effects of the rate of drug delivery on psychomotor sensitization, drug intake and incentive motivation for cocaine have been studied, but effects on the risk of relapse after abstinence have not been examined systematically. Our goal here was to address this. Relapse to drug use during abstinence is a central problem in addiction [15]. In abstinent cocaine addicts, stress [16], the drug itself [17] or drug-associated contexts and cues [18] can trigger craving and relapse. Both cue-induced drug craving [19,20,21] and drug-seeking behaviour [22] intensify during abstinence. This is termed “incubation of drug craving”, and it promotes relapse. It is not known how the rate of drug delivery influences incubation of drug craving. One study reports that after 45 days of abstinence, only rats that had taken rapid cocaine injections in the past (delivered intravenously over 5 versus 45–90 s) showed cocaine-primed reinstatement of drug-seeking behaviour [11]. Cue-induced relapse or incubation of this effect, were not assessed. Another issue is that preclinical studies on the incubation of drug craving have used rats given continuous drug access during self-administration sessions (e.g., ‘Short-Access’ or ‘Long-Access’). Continuous cocaine access produces high and sustained brain drug concentrations [23]. But cocaine use in addicts is likely intermittent, both between and within bouts of use [4, 24]. This is modelled in a new intermittent-access self-administration procedure in rats [25, 26].
Given these considerations, key questions have yet to be answered by preclinical models. Does the speed of cocaine delivery influence incubation of conditioned drug craving after abstinence? How does intermittent-access cocaine self-administration influence the incubation effect? To answer these questions, we allowed two groups of rats to self-administer intravenous cocaine injections under intermittent-access. Each injection was given over 5 s in one group (5s-rats), and over 90 s in the other (90s-rats). A control group self-administered food pellets. Cue- and drug-induced relapse (the renewal of previously extinguished drug-seeking behaviour after abstinence) were then assessed after 1 or 45 days of withdrawal/forced abstinence. The actions of the neurotrophin, brain-derived neurotrophic factor (BDNF) in the mesocorticolimbic system mediate the persistence of cue-induced drug craving after extended withdrawal [27,28,29]. Thus, after the relapse tests given following 1 and 45 days of withdrawal from cocaine, we measured BDNF protein concentrations in the prelimbic cortex, the nucleus accumbens (Nac) core and the ventral tegmental area (VTA).
Materials and methods
For rats, apparatus, intravenous catheter implantation and acquisition of food self-administration, see Supplementary Material online. The Université de Montréal's Animal Care Committee approved all experimental procedures, and these complied with the guidelines of the Canadian Council on Animal Care.
Groups
Figure 1a shows the experimental events. In the initial daily 1-h self-administration training sessions, control rats self-administered food pellets and all cocaine rats self-administered 0.5 mg/kg/infusion cocaine, injected intravenously over 5 s. All rats self-administered under FR3, with an 85-s timeout. During the timeout, levers were retracted, the houselight was turned off, the light above the active lever was illuminated, and a 5-s, 1800-Hz tone was presented. The rats had at least 3 daily sessions, until they took cocaine (or food pellets) at regular intervals and pressed at least twice more on the active versus inactive lever, for two consecutive sessions. Then for 3 additional 1-h sessions, cocaine rats were assigned to a 5-s or a 90-s group. The food control rats continued to self-administer food, with an 85-s timeout. The 5-s group continued to self-administer 5-s cocaine injections, with an 85-s timeout. The 90-s group now received 90-s cocaine infusions, with no timeout, and the reward-associated cues described above were presented during the infusion. Thus, all rats had access to a cocaine injection (or food) every 90 s, and regardless of condition, each cue presentation lasted for 90 s.
Timeline of experimental events, estimated brain cocaine concentrations and cocaine self-administration behaviour. a Rats were implanted with intravenous catheters and trained to press a lever for 45-mg, banana-flavoured food pellets. A subset of the rats was then trained to self-administer intravenous cocaine (0.5 mg/kg/injection, delivered over 5 s). The cocaine rats were then divided into two groups. For three 1-h sessions, they were given access to cocaine under a fixed ratio 3 schedule of reinforcement. Each injection was delivered over 5 s to one group (5s-rats), and over 90 s (90s-rats) to the other group. The subset of cocaine-naïve rats continued to self-administered food pellets. Next, self-administration sessions were extended to 6 h, during which cocaine (or food) was available intermittently (intermittent-access or IntA; 6-min periods of reward access, separated by 26-min periods of no reward). Following 10 IntA sessions, rats were kept in their home cages for 1 or 45 days. At each time point, rats received a single extinction session (6 h), immediately followed by a 1-h cue-induced reinstatement test (all rats) or cocaine-induced reinstatement test (cocaine rats only). 24 h after the reinstatement tests, the rats were sacrificed and their brain were removed for BDNF protein quantification. b Cocaine intake patterns and (c) modelled brain cocaine concentrations in a representative 5s-rat (black) and a representative 90s-rat (grey). The 5s-rats were limited to a maximum of 3 injections/6-min cocaine period [indicated by the horizontal line in (e). c Under these conditions, the IntA procedure produces similar spikes and troughs in estimated brain cocaine concentrations in 5 s- and 90s-rats. In (d–f), WD1 and WD45 indicate rats that will be given reinstatement tests 1 or 45 days after withdrawal from drug self-administration, respectively. d During IntA sessions, both 5s-rats and 90s-rats groups pressed more on the active versus inactive lever, with no group differences in this behaviour. In (e), the left Y-axis shows the number of injections taken during each 6-min cocaine period. The right Y-axis shows the number of injections self-administered during each 6-h IntA session. Both e cocaine intake over the sessions and f cumulative drug intake were similar in 5s- and 90s-rats. All data are mean ± SEM. 5s-rats, n = 15 for WD1 and n = 22 for WD45; 90 s-rats, n = 14 for WD1 and n = 19 for WD45
Intermittent-access (IntA)
Following the pretraining phase above, all rats transitioned to 6-h IntA self-administration sessions. During each IntA session, cocaine (or food pellets for control rats) were available in twelve, 6-min periods, separated by 26-min no-cocaine/food periods where levers were retracted [9, 25]. These sessions were given every 2 days, for a total of 10 sessions. 5s-rats escalate their intake over intermittent-access sessions [26, 30, 31], while 90s-rats take 2–3 injections/6-min cocaine-available period [9]. Here, to avoid the confounding influence of differences in cocaine intake between 5s- and 90s-rats, we limited the 5s-rats to 3 cocaine injections per 6-min drug-available period, as adapted from [9]. The 90s-rats could take up to 4 injections/6-min drug-available period, as 6 min allow for four 90-s injections. The food control rats were also limited to 3 pellets/6-min period. These conditions ensured that all rats had access to a similar number of reinforcing events/6-min period. The food control rats remained cocaine-naïve and they served to determine the extent to which any differences in outcome might involve effects related to operant learning/performance. A 26-min no-reward period was initiated after each 6-min cocaine/food period, or as soon as the 5s-rats and food controls took their 3 allotted rewards.
Withdrawal, extinction and reinstatement
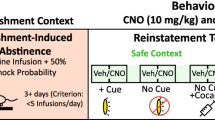
Following 10 intermittent-access sessions, rats stayed in their home cages for 1 or 45 days. On withdrawal day 1 (WD1) and withdrawal day 45 (WD45), subsets of rats from each group first received a single 6-h extinction session. Both levers were present continuously during the 6-h session and rats could press both levers, but no cocaine or food was available, and the tone-light cue was not presented. Active lever pressing during extinction can also reflect context-induced reward-seeking behaviour. However, we did not perform a formal test of context-induced drug seeking. This would require additional testing in a non-drug context as a control. Immediately after the extinction session on WD1 or WD45, rats received either a cue- or a cocaine-induced reinstatement test (1 h). For cue-induced reinstatement, the session started with a single non-contingent presentation of the tone-light cue. Thereafter, active lever presses were reinforced by the presentation of the tone-light cue, under FR3. For cocaine-induced reinstatement (5s- and 90s-rats only), rats received 10 mg/kg i.p. and pressing on either lever had no programmed consequences. The cocaine prime was given i.p. to remain consistent with a large literature on drug-primed reinstatement. However, we note that when a cocaine prime is given i.v., injecting the drug over 5, 50 or 100 s does not influence reinstatement behaviour [32].
Modelling brain cocaine concentrations
Brain cocaine concentrations (C in μM) during IntA were estimated using a two-compartment open model validated by Pan et al. [33], and recently applied to IntA self-administration studies [9, 23, 25, 30] (See Supplementary information).
Quantification of BDNF protein concentrations
Twenty-four hours after reinstatement tests on WD1 and WD45, rats were decapitated, and their brains were harvested. Bilateral tissue punches of the prelimbic cortex (PrL; + 3.7 mm from Bregma), Nac core (+ 1.7 mm) and VTA (−5.6 mm) were obtained from 1 mm coronal sections cut in a cryostat at −20 °C, according to the Rat Brain Atlas of Paxinos and Watson [34]. We quantified BDNF protein concentrations in the core rather than shell of the accumbens because BDNF protein concentrations increase in the core from cocaine WD45 onward, and are increased in the shell only after longer withdrawal periods (WD90) [35]. BDNF protein concentrations were measured using sandwich-style ELISA (BDNF Emax Immuno-Assay System kit; Promega Corporation, Madison, WI) according to the manufacturer’s instructions (See Supplementary information).
Data analysis
Group differences in behaviour during the IntA sessions and during the 6-h extinction sessions were analyzed using 3-way ANOVA (Time x Group/Withdrawal period x Lever type; Time and Lever type as within-subjects variables) and 2-way ANOVA (for cumulative intake after IntA and total lever presses during extinction, respectively; Group × Withdrawal period; both as between-subjects variables). 3-way ANOVAs were used to analyze lever-pressing behaviour during reinstatement sessions [Group/Withdrawal period × Lever type × Test type (Extinction versus Reinstatement); Lever and Test type as within-subjects variables]. Group differences in BDNF levels were analyzed with 2-way ANOVA (Group × Withdrawal period; as between-subjects variables). Significant interaction or main effects (p < 0.05) were followed by Newman–Keuls multiple comparisons tests. Data were analysed with Stat Soft. Inc. version 10.
Results
Cocaine self-administration behaviour is similar in 5s- and 90s-rats
Figures 1b, c show cocaine intake pattern and estimated brain drug concentrations during the 5th IntA session in representative 5s- and 90s-rats. During each session, cocaine was available during 6-min periods, separated by 26-min no-drug periods. Figure 1c shows that this would produce spikes and troughs in brain cocaine concentrations during the session (see also [9, 25]. Fig. 1c also shows that self-administering 5- or 90-s cocaine injections under IntA would produce a similar spiking pattern in brain cocaine concentrations, and that there would be no large effect of cocaine infusion rate on peak brain concentrations of the drug (see also refs [8, 9, 14]). Figures 1d, e show self-administration behaviour in 5s- and 90s-rats assigned to the WD1 or WD45 condition. During each session, the 5s-rats were limited to 3 injections/6-min drug-available period (indicated by the horizontal line in Fig. 1e). The 90s-rats could take up to 4 injections/6-min period. Over the IntA sessions, all groups pressed more on the active versus inactive lever (Fig. 1d, F1,66 = 415.52, p < 0.001), and there were no group differences in this behaviour (F3,66 = 2.06, p = 0.11). Both cocaine intake across the 10 sessions (Fig. 1e) and cumulative intake (Fig. 1f) were similar between groups. The control rats were also limited to 3 food pellets/6-min reward-available period and they generally took all 3 (Supplementary Figure S1).
Rapid cocaine injections promote incubation of extinction responding during abstinence
Figure 2 shows lever-pressing behaviour when rats were replaced in the previously reward-associated context (the self-administration chambers) on WD1 and WD45, under extinction conditions. All rats pressed more on the active versus inactive lever (Figs. 2a and c, F1,66 = 21.94; Figs. 2e, g, F1,9 = 94.75; All P’s < 0.001). Thus, both cocaine- and food-trained rats showed significant reward-seeking behaviour under extinction conditions. All rats also decreased active lever presses over the session, indicating extinction of reward-seeking behaviour (Fig. 2a, F5,330 = 117; Fig. 2e, F5,45 = 57.06; All P’s < 0.001). Figure 2a shows that the 5 s-rats tested on WD45 pressed more on the active lever than all other groups (Group × Withdrawal period interaction effect, F1,66 = 4.19; All P’s < 0.05). This is further highlighted in Fig. 2b, showing that total active lever presses under extinction were also highest in these rats (F1,66 = 4.19, All P’s < 0.05). Figures 2e, f show that in food control rats, pressing on the active lever did not change as a function of the duration of abstinence (All P’s > 0.05). However, these rats pressed less on the inactive lever on WD45 than on WD1 (Fig. 2h, t9 = 4.55, p = 0.001). In summary, the 5 s-rats showed greater extinction responding than 90s-rats, and only the 5s-rats showed incubation of this effect during abstinence.
Taking rapid (5 s), but not slower (90 s) intravenous cocaine injections promotes incubation of extinction responding during abstinence. After 1 or 45 days of forced abstinence/withdrawal (WD) from cocaine, all rats were placed in the cocaine-associated context and given a 6-h extinction session. Lever presses produced no cocaine and no discrete cocaine cues. a When replaced into the cocaine-taking context under extinction conditions, drug-seeking behaviour (pressing on the previously cocaine-associated lever) was greatest in the 5s-rats withdrawn for 45 days. b The total number of active lever presses during the extinction session was also greatest in this group. c, d During the extinction sessions, there were no group differences in pressing on the inactive lever. Rats that had previously self-administered food pellets were also replaced in the food-taking context and tested under extinction conditions, 1 and 45 days after the last self-administration session. e, f In food control rats, extinction responding did not vary with withdrawal time. g, h In food control rats, pressing on the inactive lever decreased from WD1 to WD45. All data are mean ± SEM. *5 s-rats on WD45 versus all other groups, p < 0.05; #versus WD1, p < 0.05. 5s-rats, n = 15 on WD1 and n = 22 on WD45; 90s-rats, n = 14 on WD1 and n = 19 on WD45; Food rats, n = 5 on WD1 and n = 6 on WD45
Rapid cocaine injections promote cue-induced reinstatement during abstinence
Cue-induced reinstatement of extinguished cocaine-seeking behaviour is indicated by pressing more on the active lever during cue-induced reinstatement tests than under extinction conditions. Figure 3 shows lever-pressing behaviour during cue-induced reinstatement tests (black bars), compared to the last hour of the extinction test (white bars). Figure 3a shows that active lever pressing in the last hour of the extinction session increased with withdrawal time (F1,39 = 15.32, p < 0.001). However, at each withdrawal time, the groups showed equivalent levels of responding in the last hour of the extinction session (All P’s > 0.05). On WD1 and WD45, only the 5 s-rats showed significant cue-induced reinstatement of cocaine-seeking behaviour [Group × Lever type × Test type (extinction or reinstatement) interaction effect, Figs. 3a, b, F3,39 = 3.72; Group × Test type Interaction effect, Fig. 3a, F3,39 = 4.21; All P’s < 0.05]. Fig. 3a also shows that on WD45, the 5 s-rats showed more cue-induced reinstatement of drug-seeking behaviour than either the 90 s-rats tested at the same time or the 5 s-rats tested on WD1 (All P’s < 0.01). As seen in Fig. 3a (right panel), the 90s-rats did not lever-press more during cue-induced reinstatement tests than during the last hour of extinction tests—at any withdrawal time (All P’s > 0.05). However, they pressed more on the active lever during the cue-induced reinstatement test on WD45 than on WD1 (p < 0.05). This can also be a form of incubation. Figures 3c, d show lever-pressing behaviour in food control rats during cue-induced reinstatement tests. Overall, these rats pressed more on the active versus inactive lever (Figs. 3c, d, F1,9 = 42.47, p < 0.001), but there were no effects of cue presentation or of withdrawal time (All P’s > 0.05). Thus, both 5s- and 90s-rats showed incubation of cue-induced reinstatement. However, on both WD1 and WD45, cue presentation increased lever-pressing behaviour above extinction levels only in the 5 s-rats. Moreover, on WD45, cue-induced reinstatement was more pronounced in 5s-rats than in 90s-rats. This suggests that a history of taking rapid cocaine injections enhances the reinstatement-triggering effects of drug cues.
Taking rapid (5 s), but not slower (90 s) intravenous cocaine injections promotes cue-induced reinstatement after forced abstinence/withdrawal (WD) from cocaine. Immediately after the 6-h extinction session given on WD1 or WD45, rats were given a 1-h cue-induced reinstatement test. During cue-induced reinstatement, pressing on the active lever did not produce cocaine, but it was reinforced by presentations of the tone-light drug cue. Responding during reinstatement tests (black bars) was compared to responding in the last hour of the extinction sessions (white bars). a On WD1 and WD45, only the 5s-rats showed cue-induced reinstatement of extinguished cocaine-seeking behaviour. This was indicated by greater pressing on the active lever during the cue-induced reinstatement test versus extinction. Cue-induced reinstatement intensified during abstinence in both the 5s- and 90s-rats (WD45 > WD1). b During cue-induced reinstatement tests, there were no group differences in pressing on the inactive lever. c, d In food control rats, there was no effect of food cue presentation on either active or inactive lever-pressing behaviour. All data are mean ± SEM. *versus extinction session in 5s-rats at the same withdrawal time, p < 0.05; &versus 5s-rats during cue-induced reinstatement on WD1, p < 0.05; #versus 90 s-rats during the reinstatement test on WD45, p < 0.05, $versus 90s-rats during the reinstatement test on WD1. 5s-rats, n = 8 on WD1 and n = 15 on WD45; 90 s-rats, n = 7 on WD1 and n = 13 on WD45; Food rats, n = 5 on WD1 and n = 6 on WD45
Rapid cocaine injections promote cocaine-induced reinstatement after abstinence
Cocaine-induced reinstatement of extinguished cocaine-seeking behaviour is indicated by pressing more on the active lever after being injected with the drug than under extinction conditions. Figure 4 shows lever-pressing behaviour during cocaine (10 mg/kg, i.p.)-induced reinstatement tests (black bars), compared to the last hour of the extinction tests (white bars). Figure 4a shows that active lever pressing in the last hour of the extinction session was similar between the groups and across withdrawal times (All P’s > 0.05). However, when injected with cocaine on WD1 or WD45, only 5 s-rats significantly reinstated drug-seeking behaviour (Figs. 4a, b, Group × Lever type × Test type (extinction or reinstatement) interaction effect, F3,23 = 2.89; Fig. 4a, Group × Test type interaction effect; F3,23 = 3.27; All P’s < 0.05). Figure 4a also shows that on WD45, the 5-s rats showed greater cocaine-induced reinstatement of drug-seeking behaviour than the 90-s rats (p < 0.001). On WD1, there was a tendency for the 90s-rats to show cocaine-induced reinstatement of drug-seeking behaviour (Fig. 4a, p = 0.11). No other comparisons were statistically significant. Thus, on WD1 and WD45, only 5s-rats showed significant cocaine-primed reinstatement of drug-seeking behaviour and there was no incubation of this effect.
Taking rapid (5 s), but not slower (90 s) intravenous cocaine injections promotes cocaine-primed reinstatement after forced abstinence/withdrawal (WD). Immediately after a 6-h extinction session given on WD1 or WD45, rats were given a 1-h cocaine (10 mg/kg, i.p.)-induced reinstatement test. During cocaine-induced reinstatement, lever pressing produced no cocaine and no cues. Responding during reinstatement tests (black bars) was compared to responding in the last hour of the extinction sessions (white bars). a After 1 or 45 days of forced abstinence/withdrawal from cocaine, only 5s-rats showed significant cocaine-induced reinstatement of extinguished drug-seeking behaviour. b During cocaine-induced reinstatement tests, there were no group differences in pressing on the inactive lever. All data are mean ± SEM. *versus extinction session in 5s-rats at the same withdrawal time, p < 0.05; #versus 90s-rats during the reinstatement test on WD45, p < 0.05. 5s-rats, n = 7 on each of WD1 and WD45; 90s-rats, n = 7 on WD1 and n = 6 on WD45
Rapid cocaine injections produce time-dependent increases in brain BDNF protein concentrations during abstinence
Reinstatement tests were given on WD1 or WD45. Twenty-four hours after each reinstatement test, we quantified BDNF protein concentrations in the prelimbic cortex, the NAc core and the VTA. Figure 5 shows BDNF protein concentrations presented as the percentage of the mean values of food control rats. Figures 5d–f show that in the cohort used for cue-induced reinstatement tests, there were no group differences in BNDF protein concentrations after WD1 (white bars). However, after WD45 (black bars), the 5s-rats had significantly higher BDNF protein concentrations compared to either 90s- or control rats, in all brain regions sampled (Fig. 5d, F2,27 = 4.24; Fig. 5e, F2,27 = 2.23; Fig. 5f, F2,27 = 9.87; All P’s < 0.05). In all brain regions sampled, the 5s-rats also had greater BDNF protein concentrations after WD45 than after WD1 (Figs. 5d–f, All P’s < 0.05). Figures 5g–i show that a different pattern of results emerged after the cocaine-induced reinstatement tests. There were no group differences in BDNF concentrations in the prelimbic cortex or Nac core (Figs. 5g, h, All P’s > 0.05). However, in the VTA (Fig. 5i), 5s- and 90s-rats had greater BDNF protein expression than control rats, and levels were greatest in the 5s-rats (F2,28 = 8.35; All P’s < 0.05). This did not depend on the withdrawal period (p > 0.05). The BDNF increase in the VTA of 5s- and 90s-rats is likely driven by re-exposure to cocaine 24 h prior to sacrifice, rather than by the self-administration history alone. Indeed, Fig. 5f shows that in the 90 s-rats used for cue-induced reinstatement (i.e., not re-exposed to cocaine), there were no significant BDNF changes in the VTA. In summary, following cocaine-induced reinstatement tests, both 5s- and 90s-rats had increased BDNF protein concentrations in the VTA relative to controls, and concentrations were highest in the 5s-rats. This was not influenced by withdrawal time. In contrast, in the rats tested for cue-induced reinstatement—and therefore not re-exposed to cocaine prior to sacrifice—only the 5s-rats had elevated BDNF protein concentrations in the prelimbic cortex, Nac core and VTA, and the time course of this increase paralleled that of incubation of cue-induced reinstatement in these rats (WD45 > WD1).
Forced abstinence/withdrawal (WD) from rapid (5 s), but not slower (90 s) intravenous cocaine injections evokes time-dependent increases in brain BDNF protein concentrations. 24 h after the reinstatement tests given on WD1 or WD45, brains were extracted and BDNF protein expression was quantified in a prelimbic cortex (PrL), b nucleus accumbens core (Nac) and c ventral tegmental area (VTA). Data are presented as the percentage of mean values of food control rats (± SEM). In the subset of rats tested for cue-induced reinstatement on WD45 (top three panels), 5s-rats had greater BDNF protein concentrations relative to both 90s-rats and food control rats in d the PrL, e Nac and f VTA. In the subset of rats used for cocaine-induced reinstatement on WD1 or WD45 (bottom three panels), 5s- and 90s-rats had unchanged BDNF protein concentrations in g the PrL and h Nac. i Both groups had increased BDNF protein expression in the VTA, and 5s-rats had the highest expression levels. All data are mean ± SEM. *versus 5s-rats tested on WD1, p < 0.05; #versus 90s- or food control rats tested on WD45, p < 0.05; $versus 90s-rats and food control rats, across withdrawal times, p < 0.05; &versus food control rats, across withdrawal times, p < 0.05. Cue-induced reinstatement rats: 5s-rats, n = 5 on WD1 and n = 6 on WD45; 90 s-rats, n = 5 on WD1 and n = 6 on WD45; Food rats, n = 5 on WD1 and n = 6 on WD45. Cocaine-induced reinstatement rats: 5s-rats, n = 6 on each of WD1 and WD45; 90s-rats, n = 6 on WD1 and n = 5 on WD45
Discussion
Addiction to cocaine is more likely and more severe in people who use rapid routes of drug administration [5, 6]. Here, we determined how variation in the speed of cocaine delivery influences the susceptibility to relapse after abstinence. First, we found that after extended abstinence (45 days versus 1 day), rats that had taken rapid cocaine injections in the past (5s-rats) show more vigorous drug-seeking behaviour under extinction conditions compared to rats that had taken slower injections (90s-rats), and only 5s-rats show incubation of extinction responding during abstinence. Second, only 5s-rats showed cocaine-induced reinstatement (see also [11]). This is in contrast to Crombag et al. [32], who used ‘Short-Access’ sessions (1–2 h) and found that taking rapid or slower cocaine or amphetamine injections did not influence subsequent drug-induced reinstatement. Third, while both 5s- and 90s-rats showed incubation of cue-induced reinstatement, cue presentation increased drug-seeking behaviour above extinction levels only in the 5 s-rats, and cue-induced reinstatement after extended abstinence (45 days) was greatest in the 5s-rats. This suggests that taking rapid cocaine injections increases the susceptibility to cue-induced relapse after abstinence. The effects of injection speed were seen in spite of equivalent exposure to cocaine, to the cocaine-taking context, and to the discrete cocaine-associated cues. These findings extend previous work showing that the speed at which cocaine reaches the brain predicts the development of behavioural symptoms relevant to addiction [7,8,9, 11,12,13].
The speed of cocaine delivery predicts the vulnerability to relapse
Cue-induced cocaine craving incubates after abstinence in both humans and rodents. This likely increases the vulnerability to relapse long after the last bout of drug use. Our findings show that withdrawal/forced abstinence from rapidly delivered cocaine changes the brain in ways that make rats more susceptible to relapse induced by the drug or by drug-conditioned cues, and this susceptibility intensifies during abstinence. The present study also assesses incubation of cue-induced reinstatement in rats given intermittent-access to cocaine. All studies to date showing incubation of conditioned drug craving have used continuous-access self-administration procedures (e.g., Long-Access). These procedures produce continuously high brain concentrations of drug, while intermittent-access produces a spiking pattern of drug concentrations in the brain (Fig. 2c, see also references [9, 23]). Cocaine ‘spikes’ might better model the pattern achieved by cocaine addicts during a bout of intoxication [24]. Intermittent-access is also uniquely effective in producing addiction-relevant neurobehavioural changes in animal models [23, 26, 30]. We did not have a continuous-access comparison group here and comparing across studies is done with caution. However, extinction responding in our rats was much more robust than seen in previous incubation studies using continuous-access cocaine self-administration. For instance, Grimm et al. [22], gave their continuous-access rats a single 6 to 8-h extinction session at different times following withdrawal from cocaine. On WD1, their rats pressed on average < 5 times on the previously active lever in the last hour of extinction, while our 5s- and 90s-rats pressed 17 (± 3 SEM) and 13 (± 4 SEM) times, respectively. On WD29, the Grimm et al. [22], rats pressed on average <10 times in the last hour of extinction, while our 5s- and 90s-rats, pressed 39 (± 4 SEM) and 29 times (± 5 SEM), respectively, on WD45. It is possible, then, that the intermittent-access protocol promotes extinction responding. Indeed, Kawa et al. [26], report that in early abstinence (~ 2–7 days), intermittent-access rats show similar levels of responding in the last hour of the extinction test as we observed here, and they also found greater cocaine- and cue-induced reinstatement than generally seen in long-access rats. Here we show that intermittent-access rats show incubation of cue-induced reinstatement of cocaine-seeking behaviour, and that taking rapid cocaine injections during intermittent-access sessions promotes cue-induced reinstatement after extended withdrawal.
Increasing the speed of cocaine delivery increases susceptibility to some, but not all effects of discrete drug cues. Drug-associated cues contribute to addiction in powerful ways [36]. They can trigger relapse during abstinence, as measured in the present study, and they can also act as conditioned rewards, where they support the learning of new instrumental behaviours [37]. The conditioned rewarding effects of cocaine cues are similar in rats that have taken rapid versus slower i.v. cocaine injections [38]. In contrast, we show here that rats withdrawn from rapid cocaine injections display more cue-induced reinstatement behaviour than rats that had taken slower injections. The cocaine self-administration procedures used here are different from those used in this previous study. It is also possible that different neural substrates mediate the conditioned rewarding versus relapse-triggering effects of drug cues. As this is examined further, the present findings suggest that people who use rapid routes of cocaine administration could be especially sensitive to the relapse-triggering effects of drug-associated cues.
The speed of cocaine delivery predicts changes in BDNF protein concentrations following abstinence
Rapid cocaine delivery more readily changes brain systems involved in incentive motivation [7, 9, 10, 39]. In agreement with this, we found that BDNF protein concentrations in the prelimbic cortex, NAc core and VTA increase after extended withdrawal from cocaine only in rats with a history of taking rapid drug injections (see also references [28, 35]. There were no time-dependent changes in BDNF concentrations in rats that had self-administered slower cocaine injections (90 s) or food in the past. This suggests two things. First, increased BDNF protein expression in these brain regions is an effect relevant to the process of addiction, rather than a consequence of either withdrawal from chronic cocaine use or operant responding for a reward. Second, BDNF-mediated plasticity could contribute to the persistence of the incentive motivational effects of cocaine cues after forced abstinence/withdrawal. It has been proposed that the time-dependent increase in BDNF-mediated activity might not cause incubation, but it could maintain high levels of craving after extended abstinence [40]. This effect could involve BDNF-induced increases in dendritic branching and spine density in neurons [41].
During abstinence, a single re-exposure to cocaine changed BDNF protein concentrations in the VTA. When sacrificed 24 h after an i.p. cocaine injection, both 5s- and 90s-rats had increased BDNF protein concentrations in the VTA compared to cocaine-naive rats, and this did not depend on abstinence length (Fig. 5i). We did not have a group of rats naive to behavioural testing before brain extraction. However, we speculate that the increase in BDNF in the VTA could be related to re-exposure to cocaine before sacrifice. Acute, experimenter-injected cocaine can change BDNF mRNA and protein expression in the brain [42, 43]. We have also shown previously that immediately after a final self-administration session, only rats that had taken rapid cocaine injections (5 versus 90 s) have altered levels of BDNF and TrkB mRNA in cortical areas and in the dorsal striatum (with no changes in the NAc) [10]. Here, consistent with prior work [28], cocaine self-administration did not change BDNF protein concentrations relative to food self-administration after 1 day of abstinence. The difference between our previous findings [10] and the current results could be due to the measurement of protein versus mRNA and also to the use of an intermittent-access self-administration procedure here.
Reinstatement behaviour following intermittent-access food self-administration
We also assessed incubation of cue-induced reinstatement of responding for a non-drug reward (i.e., food pellets) following intermittent-access self-administration sessions. Our rats did not show cue-induced reinstatement of food-seeking behaviour. However, when they were replaced in the food-associated context under extinction conditions, the rats pressed more on the previously food-associated lever than on the inactive lever. This effect did not vary as a function of the length of the abstinence period. Food-seeking behaviour induced by contextual cues can incubate over time [44]. Some studies also report incubation of food-seeking behaviour induced by discrete cues, along with differences in the time course of the effect [45,46,47,48], while other studies report no incubation [49, 50]. The studies reporting incubation show this over the first 21 days after withdrawal from food self-administration [44,45,46,47]. The studies reporting no incubation ([49, 50] and present study) tested cue-induced reinstatement of food-seeking ≥ 35 days following withdrawal. Cue-induced reinstatement of food-seeking behaviour can wane over time [28]. This being said, we did not observe cue-induced reinstatement of food-seeking behaviour, at any time after withdrawal. Our rats were food restricted. However, the literature suggests that both food-restricted [47, 49] and food-unrestricted rats [45, 46] can show cue-induced reinstatement of food-seeking. We also limited the number of self-administered pellets during each session. This could have prevented subsequent cue-induced reinstatement of food-seeking behaviour. Finally, all previous studies showing this reinstatement effect have used continuous-access food self-administration sessions prior to testing. Future studies can determine how continuous- versus intermittent-access food self-administration might change the long-term effects of food-associated cues on brain and behaviour.
Conclusions
The rapid delivery of cocaine increases the vulnerability to drug- and cue-induced reinstatement of drug-seeking behaviour after withdrawal from cocaine self-administration. This effect is accompanied by time-dependent increases in BDNF protein concentrations in mesocorticolimbic regions. Thus, beyond how much cocaine is taken, how fast it gets to the brain is decisive in predicting long-term outcome [3, 4]. We conclude that routes of cocaine administration that result in rapid brain uptake might promote addiction in part because they facilitate the neurobiological, psychological and behavioural changes that increase the risk of relapse, and these changes persist long after the last drug exposure.
Funding and disclosure
ANS is a consultant for H. Lundbeck A/S. ABG and FA declare no conflicts of interest. This research was supported by grants to ANS from the Canadian Foundation for Innovation (grant number 24326) and the Canadian Institutes of Health Research (grant number 200200). ANS is supported by a salary grant from the Fonds de Recherche du Québec – Santé (grant number 28988). FA is supported by a PhD fellowship from the Groupe de Recherche sur le Système Nerveux Central.
References
Badiani A, Robinson TE. Drug-induced neurobehavioral plasticity: the role of environmental context. Behav Pharmacol. 2004;15(5-6):327–39.
Le Moal M. Drug abuse: vulnerability and transition to addiction. Pharmacopsychiatry. 2009;42(1):S42–55.
Samaha AN, Robinson TE. Why does the rapid delivery of drugs to the brain promote addiction? Trends Pharmacol Sci. 2005;26(2):82–7.
Allain F, Minogianis EA, Roberts DC, Samaha AN. How fast and how often: the pharmacokinetics of drug use are decisive in addiction. Neurosci Biobehav Rev. 2015;56:166–79.
Gawin FH, Ellinwood EH Jr. Cocaine and other stimulants. Actions, abuse, and treatment. N Engl J Med. 1988;318(18):1173–82.
Hatsukami DK, Fischman MW. Crack cocaine and cocaine hydrochloride. Are the differences myth or reality? JAMA. 1996;276(19):1580–8.
Samaha AN, Mallet N, Ferguson SM, Gonon F, Robinson TE. The rate of cocaine administration alters gene regulation and behavioral plasticity: implications for addiction. J Neurosci. 2004;24(28):6362–6370.
Samaha AN, Li Y, Robinson TE. The rate of intravenous cocaine administration determines susceptibility to sensitization. J Neurosci. 2002;22(8):3244–3250.
Allain F, Roberts DCS, Lévesque D, Samaha AN. Intermittent intake of rapid cocaine injections promotes robust psychomotor sensitization, increased incentive motivation for the drug and mGlu2/3 receptor dysregulation. Neuropharmacology. 2017;117:227–237.
Bouayad-Gervais K, Minogianis EA, Lévesque D, Samaha AN. The self-administration of rapidly delivered cocaine promotes increased motivation to take the drug: contributions of prior levels of operant responding and cocaine intake. Psychopharmacol (Berl). 2014;231(21):4241–52.
Wakabayashi KT, Weiss MJ, Pickup KN, Robinson TE. Rats markedly escalate their intake and show a persistent susceptibility to reinstatement only when cocaine is injected rapidly. J Neurosci. 2010;30(34):11346–11355.
Liu Y, Roberts DC, Morgan D. Sensitization of the reinforcing effects of self-administered cocaine in rats: effects of dose and intravenous injection speed. Eur J Neurosci. 2005;22(1):195–200. p
Minogianis EA, Levesque D, Samaha AN. The speed of cocaine delivery determines the subsequent motivation to self-administer the drug. Neuropsychopharmacology. 2013;38(13):2644–56.
Minogianis, EA, Shams WM, Mabrouk OS, Wong JT, Brake WG, Kennedy RT, du Souich P, Samaha AN. Varying the rate of intravenous cocaine infusion influences the temporal dynamics of both drug and dopamine concentrations in the striatum. Eur J Neurosci, 2018. https://onlinelibrary.wiley.com/ https://doi.org/10.1111/ejn.13941.
O'Brien CP. A range of research-based pharmacotherapies for addiction. Science. 1997;278(5335):66–70.
Sinha R. How does stress increase risk of drug abuse and relapse? Psychopharmacol (Berl). 2001;158(4):343–59.
Jaffe JH, Cascella NG, Kumor KM, Sherer MA. Cocaine-induced cocaine craving. Psychopharmacol (Berl). 1989;97(1):59–64.
Gawin FH, Kleber HD. Abstinence symptomatology and psychiatric diagnosis in cocaine abusers. Clinical observations. Arch Gen Psychiatry. 1986;43(2):107–13.
Wang G, Shi J, Chen N, Xu L, Li J, Li P, Sun Y, Lu L. Effects of length of abstinence on decision-making and craving in methamphetamine abusers. PLoS One. 2013;8(7):e68791.
Li P, Wu P, Xin X, Fan YL, Wang GB, Wang F, Ma MY, Xue MM, Luo YX, Yang FD, Bao YP, Shi J, Sun HQ, Lu L. Incubation of alcohol craving during abstinence in patients with alcohol dependence. Addict Biol. 2015;20(3):513–22.
Parvaz MA, Moeller SJ, Goldstein RZ. Incubation of Cue-Induced Craving in Adults Addicted to Cocaine Measured by Electroencephalography. JAMA. Psychiatry. 2016;73(11):1127–1134.
Grimm JW, Hope BT, Wise RA., Shaham Y. Neuroadaptation. Incubation of cocaine craving after withdrawal. Nature. 2001;412(6843):141–2.
Zimmer BA, Oleson EB, Roberts DC. The motivation to self-administer is increased after a history of spiking brain levels of cocaine. Neuropsychopharmacology. 2012;37(8):1901–1910.
Beveridge TJR, Wray P, Brewer A, Shapiro B, Mahoney JJ, Newton TF, Haile CN, De La Garza II D. Analyzing Human Cocaine Use Patterns to Inform Animal Addiction model Development. (Published abstract for the College on Problems of Drug Dependence Annual Meeting, Palm Springs, CA). 2012.
Zimmer BA, Dobrin CV, Roberts DC. Brain-cocaine concentrations determine the dose self-administered by rats on a novel behaviorally dependent dosing schedule. Neuropsychopharmacology. 2011;36(13):2741–9.
Kawa AB, Bentzley BS, Robinson TE. Less is more: prolonged intermittent access cocaine self-administration produces incentive-sensitization and addiction-like behavior. Psychopharmacol (Berl). 2016;233(19-20):3587–602.
Li X, Wolf ME. Multiple faces of BDNF in cocaine addiction. Behav Brain Res. 2015;279:240–54.
Grimm JW, Lu L, Hayashi T, Hope BT, Su TP, Shaham Y. Time-dependent increases in brain-derived neurotrophic factor protein levels within the mesolimbic dopamine system after withdrawal from cocaine: implications for incubation of cocaine craving. J Neurosci. 2003;23(3):742–747.
Lu L, Grimm JW, Hope BT, Shaham Y. Incubation of cocaine craving after withdrawal: a review of preclinical data. Neuropharmacology. 2004;47(Suppl 1):214–26.
Allain F, Bouayad-Gervais K, Samaha AN. High and escalating levels of cocaine intake are dissociable from subsequent incentive motivation for the drug in rats. Psychopharmacol (Berl). 2018;235(1):317–328.
Pitchers KK, Wood TR, Skrzynski CJ, Robinson TE, Sarter M. The ability for cocaine and cocaine-associated cues to compete for attention. Behav Brain Res. 2017;320:302–315.
Crombag HS, Ferrario CR, Robinson TE. The rate of intravenous cocaine or amphetamine delivery does not influence drug-taking and drug-seeking behavior in rats. Pharmacol Biochem Behav. 2008;90(4):797–804.
Pan HT, Menacherry S, Justice JB Jr. Differences in the pharmacokinetics of cocaine in naive and cocaine-experienced rats. J Neurochem. 1991;56(4):1299–306.
Paxinos, G and C Watson, The Rat Brain in Stereotaxic Coordinates. second ed., 1986. Academic, New York, NY, USA.
Li X, DeJoseph MR, Urban JH, Bahi A, Dreyer JL, Meredith GE, Ford KA, Ferrario CR, Loweth JA, Wolf ME. Different roles of BDNF in nucleus accumbens core versus shell during the incubation of cue-induced cocaine craving and its long-term maintenance. J Neurosci. 2013;33(3):1130–1142.
O'Brien CP, Childress AR, McLellan AT, Ehrman R. Classical conditioning in drug-dependent humans. Ann N Y Acad Sci. 1992;654:400–15.
Mackintosh NJ. The psychology of animal learning. London: Academic; 1974.
Samaha AN, Minogianis EA, Nachar W. Cues paired with either rapid or slower self-administered cocaine injections acquire similar conditioned rewarding properties. PLoS One. 2011;6(10):e26481.
Porrino LJ. Functional consequences of acute cocaine treatment depend on route of administration. Psychopharmacol (Berl). 1993;112(2-3):343–51.
Loweth JA, Tseng KY, Wolf ME. Adaptations in AMPA receptor transmission in the nucleus accumbens contributing to incubation of cocaine craving. Neuropharmacology. 2014;76 Pt B:287–300.
Robinson TE, Kolb B. Alterations in the morphology of dendrites and dendritic spines in the nucleus accumbens and prefrontal cortex following repeated treatment with amphetamine or cocaine. Eur J Neurosci. 1999;11(5):1598–604.
Fumagalli F, Di Pasquale L, Caffino L, Racagni G, Riva MA. Repeated exposure to cocaine differently modulates BDNF mRNA and protein levels in rat striatum and prefrontal cortex. Eur J Neurosci. 2007;26(10):2756–63.
McGinty JF, Whitfield TW Jr., Berglind WJ. Brain-derived neurotrophic factor and cocaine addiction. Brain Res. 2010;1314:183–93.
Youtz REP. The change with time of a Thorndikian response in the rat. J Exp Psychol. 1938;23:128–140.
Shalev U, Morales M, Hope B, Yap J, Shaham Y. Time-dependent changes in extinction behavior and stress-induced reinstatement of drug seeking following withdrawal from heroin in rats. Psychopharmacol (Berl). 2001;156(1):98–107.
Grimm JW, Shaham Y, Hope BT. Effect of cocaine and sucrose withdrawal period on extinction behavior, cue-induced reinstatement, and protein levels of the dopamine transporter and tyrosine hydroxylase in limbic and cortical areas in rats. Behav Pharmacol. 2002;13(5-6):379–88.
Counotte DS, Schiefer C, Shaham Y, O'Donnell P. Time-dependent decreases in nucleus accumbens AMPA/NMDA ratio and incubation of sucrose craving in adolescent and adult rats. Psychopharmacol (Berl). 2014;231(8):1675–84.
Uejima JL, Bossert JM, Poles GC, Lu L. Systemic and central amygdala injections of the mGluR2/3 agonist LY379268 attenuate the expression of incubation of sucrose craving in rats. Behav Brain Res. 2007;181(2):292–6.
Nugent AL, Anderson EM, Larson EB, Self DW. Incubation of cue-induced reinstatement of cocaine, but not sucrose, seeking in C57BL/6J mice. Pharmacol Biochem Behav. 2017;159:12–17.
Halbout B, Bernardi RE, Hansson AC, Spanagel R. Incubation of cocaine seeking following brief cocaine experience in mice is enhanced by mGluR1 blockade. J Neurosci. 2014;34(5):1781–1790.
Acknowledgements
We thank Dr. Terry E. Robinson for revising an earlier version of the manuscript. We also thank Drs. Bruce T. Hope, Daniel Lévesque, Yavin Shaham and Teruo Hayashi for advice on BDNF protein quantification
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Gueye, A.B., Allain, F. & Samaha, AN. Intermittent intake of rapid cocaine injections promotes the risk of relapse and increases mesocorticolimbic BDNF levels during abstinence. Neuropsychopharmacol. 44, 1027–1035 (2019). https://doi.org/10.1038/s41386-018-0249-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41386-018-0249-8
This article is cited by
-
Brain-derived neurotrophic factor serum levels as a candidate biomarker for withdrawal in crack heroin dependence
Substance Abuse Treatment, Prevention, and Policy (2024)
-
Memory consolidation drives the enhancement of remote cocaine memory via prefrontal circuit
Molecular Psychiatry (2024)
-
Peripheral neurotrophin levels during controlled crack/cocaine abstinence: a systematic review and meta-analysis
Scientific Reports (2024)
-
Intermittent nicotine access is as effective as continuous access in promoting nicotine seeking and taking in rats
Psychopharmacology (2024)
-
Metabotropic glutamate group II receptor activation in the ventrolateral dorsal striatum suppresses incentive motivation for cocaine in rats
Psychopharmacology (2023)