Abstract
Eosinophils are granulocytes, typically implicated as end-stage effector cells in type-II immune responses. They are capable of producing a wide array of pre-formed molecules which render them with vast potential to influence a wide variety of processes. Nonetheless, eosinophil research has traditionally focused on their role in anti-helminthic responses and pathophysiological processes in type-II immune disorders, such as allergy and asthma, where eosinophilia is a hallmark phenotype. However, a number of key studies over the past decade have placed this restricted view of eosinophil function into question, presenting additional evidence for eosinophils as critical regulators of various homeostatic processes including immune maintenance, organ development, and tissue regeneration.
Similar content being viewed by others
Eosinophils at steady-state: development, recruitment, and maintenance
Eosinophils are multi-functional granulocytes, capable of producing a wide array of molecules that are stored pre-formed in various granules and lipid bodies throughout their cytoplasm.1,2,3 Their capacity to release a large repertoire of cytokines, chemokines, lipid mediators, and granule constituents (e.g., eosinophil cationic protein, eosinophil peroxidase, major basic protein) renders these cells with vast potential to influence a wide range of processes.
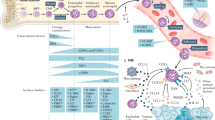
Eosinophils develop in the bone marrow from GATA-binding factor 1 (GATA1) positive common granulocyte-monocyte precursor cells, which also give rise to basophils and mast cells. Differentiation into eosinophils is distinguished by the requirement for high GATA1 expression and is largely dependent on various cytokines including the eosinophil-specific factor, IL-5 (Fig. 1). These cells exit the bone marrow niche into the circulation as mature cells with a relatively short half-life (estimated to be between 3 and 18 h) where they represent <5% of total circulating leukocytes under homeostatic conditions (Fig. 1). Eosinophil chemotaxis is primarily dependent on surface expression of C-C chemokine receptor 3 (CCR3), a receptor for eotaxins, which regulates their migration and extravasation into all tissues (Fig. 1). Eotaxins include three family members, CCL11/eotaxin-1, CCL24/eotaxin-2, and CCL26/eotaxin3, each exhibiting distinct, albeit overlapping, patterns of tissue expression. The primary sources of eotaxins within tissues are stromal in origin, such as fibroblasts, smooth muscle cells and endothelial cells, but they can also be produced by epithelial cells.4,5,6,7
Eosinophil development occurs in the bone marrow, arising from GATA-1+ granulocyte/macrophage progenitors (GMPs). Development into eosinophil-lineage-committed progenitors to (EoP) requires transcription factors c-EBPs and high expression of GATA1. During this transition between GMPs to EoPs, IL-33 can expand the pool of EoPs, upregulating expression of IL-5Ra. Development of EoPs into granule-mature eosinophils is facilitated by cytokines ILC2-derived IL-5 plus IL-3 and GM-CSF. Under homeostatic conditions, eosinophils exit the bone marrow into peripheral blood as mature cells identified as Siglec-F+ CCR3+ F4/80+ CD62L+ which have a relatively short half-life. Eosinophils are recruited into peripheral tissues in a manner dependent on local CCL11/eotaxin production. Tissue-resident eosinophils are primarily found in mucosal sites (i.e., GI-tract*, lung, mammary gland), but can also be found in the adipose and thymus where they are noted to contribute to various homeostatic and tissue-protective functions. The tissue microenvironment has significant influence on eosinophil characteristics demonstrated by discrepancies in surface marker expression and turnover rates.
While eosinophil recruitment and accumulation in tissues has largely been studied in the context of active type-II immune responses (i.e., helminth infection, allergic asthma), eosinophils are enriched within the thymus, adipose, and various mucosal tissues, including the intestine, uterus, lung, mammary gland, under homeostatic conditions (Table 1). In the gastrointestinal (GI)-tract (stomach, small intestine, large intestine) and thymus, homeostatic eosinophil recruitment occurs during fetal development.8,9 This is in contrast to the lung, where resident eosinophils are only recruited post-natally.10,11 Intestinal eosinophils are recruited independent of the microbiota, with high eosinophil numbers sustained throughout life, while resident eosinophil recruitment in the lung is speculated to be influenced by the microbiota given its post-natal emergence.10,12 The uterus and mammary gland, whose development and remodeling is highly sensitive to sex-hormones (e.g., estrogen), have varying levels of resident eosinophils throughout life in a manner speculated to be driven (in part) by estrogen-induced eotaxin-1 production at these sites.13,14 Increased eosinophil recruitment to homeostatic adipose tissue requires IL-5, eotaxins and a population of CCR2-expressing cells, however the precise mechanisms by which they are recruited to healthy versus obese adipose is not known.15
In terms of eosinophil distribution, the intestine harbors the largest number of tissue-resident eosinophils in the body per total area (5–25% of total leukocytes) whereas eosinophils in the adipose tissue and the lung constitute only ~4% of stromal/vascular fraction and ~1% of total leukocytes respectively.10,16 In the uterus, there is an estimated 1 × 105 cells within the uterine horn but this number varies depending on the estrus cycle, with the highest eosinophil recruitment at peak.13,17 Similarly, eosinophil numbers in the thymus and mammary gland also fluctuate over time. While eosinophils are recruited to the cortico-medullary region (CMR) of the thymus during fetal development, peaking around 2 weeks after birth, their numbers decrease shortly thereafter and only rise again, in both the CMR and medullary region, around 16 weeks corresponding to the time of thymic involution.9 Eosinophils are recruited in the mammary gland around ~2.5–3 weeks of age in mice and presumably sustained only throughout the duration of mammary duct development (~8 weeks). However, they are recruited back during pregnancy when mammary development is restarted, and their presence is sustained until the weaning of pups.
Tissue-resident eosinophils are noted to demonstrate increased survival upon migration into peripheral tissues, where they exhibit differential turnover rates (Table 1) and surface marker expression (Fig. 1) depending on the tissue in question, indicating the importance of the tissue microenvironment/tissue-trophic cues in fine-tuning eosinophil function. In mucosal sites, the eosinophils have prolonged survival in the intestine and uterus in contrast to the lung where their half-life is relatively shorter (Table 1). Differences in eosinophil turnover rates have been attributed to distinct expression of common γ-chain receptor, which increases eosinophil survival (Table 1). This receptor is expressed in intestinal and uterine eosinophils but is absent from lung resident eosinophils. The influence of the tissue microenvironment on eosinophil characteristics demonstrates the capacity of these cells to respond to and integrate tissue-trophic cues. Given the diversity of molecules eosinophils store (and can synthesize de novo) and their prolonged survival in certain tissues, it would be interesting to see how and whether tissue-trophic cues in various compartments that harbor homeostatic eosinophils fine-tune the repertoire of the molecules released upon activation (e.g., degranulation) according to the demands of a given tissue. This could help explain the diversity of functions eosinophils may have in the tissues in which they reside (discussed in later sections).
In this review, we highlight recent literature aimed at investigating the functional significance of eosinophil residency within healthy tissues with a specific focus on mucosal tissues. We also discuss the novel questions these studies raise as well as their implications on our understanding of the factors contributing to mucosal homeostasis.
Factors that regulate mucosal eosinophils in health and disease
IL-5 is a canonical, eosinophil-specific factor, which plays a central role in various aspects of eosinophil activation and survival in different tissues.18,19 IL-5 is known to prime eosinophils, lowering their threshold for degranulation in response to CCL11. Genetic deletion and antibody blockade of either CCL11 or IL-5 results in a significant decrease in eosinophils in all peripheral sites1 (Fig. 1). It is likely that both CCL11 and IL-5 contribute to eosinophil recruitment into tissues under normal conditions, but that they can compensate for each other when one factor is absent. For the lung IL-5 mainly acts systemically to induce eosinophilia, whilst IL-4/13-induced eotaxin promotes the transmigration of eosinophils across the vascular bed into the pulmonary tissue.20 Of note, blockade of both factors does not result in total ablation of eosinophil tissue recruitment, suggesting that other factors may also influence eosinophil recruitment.21 Similarly to IL-5, other type-II immune cytokines such as IL-4 and IL-13, which can be sourced from Th2 cells, mast cells and basophils, can also promote eosinophil activation.22,23,24 Amongst the innate cells able to regulate eosinophil development and survival, type 2 innate lymphoid cells (ILC2s) are perhaps the most important (Fig. 1), as these cells are a rich source of type II cytokines, particularly IL-5, and represent the major tissue source of such cytokines under homeostatic conditions.25,26,27
Apart from cytokines, various receptors expressed by eosinophils on their surface can also regulate their activation and survival.28,29,30 Siglec-F, which is expressed on the surface of eosinophils, mediates eosinophil apoptosis upon cross-linkage or ligand binding to other sialylated or sulphated glycans. Surface expression of Siglec-F correlates with eosinophil activity and is noted to be an important negative feedback mechanism limiting eosinophilia under inflammatory conditions. Eosinophils can also regulate their own survival and activation through release of autocrine signals. For example, upon contact with the extracellular matrix (ECM) protein fibronectin via VLA-4 (α4β1 integrin, CD49d/CD29)-mediated adhesion, eosinophil survival is prolonged through autocrine release of GM-CSF and IL-3.31,32 Autocrine production of IL-5 can also be induced in response to stimulation by antibody (IgA, IgG) immune complexes.33,34 IL-5, GM-CSF, and IL-3 are known eosinopoetins and can function synergistically to sustain eosinophil activation and survival in peripheral tissues, and support eosinophil development in the bone marrow2,35 (Fig. 1).
The epithelial-derived alarmin IL-33 is also an important regulator of eosinophil function36,37,38 and is produced both during inflammation and in homeostasis.36,39 In line with this, the IL-33 receptor, ST2, is expressed on the surface of eosinophils both during homeostasis (Fig. 1) and under inflammatory conditions. Eosinophilia is a major hallmark phenotype of in vivo IL-33 administration, which regulates eosinophil activation and expansion both directly, via ST2-signaling on eosinophils, and indirectly via ILC2s.26,40,41,42 For example, transfer of ST2-deficient eosinophils into recipient mice subjected to allergen challenge resulted in decreased eosinophil numbers in the lung, despite normal migratory capacity.43 This suggests that in vivo, tissue-derived IL-33 can act directly on eosinophils to sustain their survival. IL-33 also functions as a potent modulator of eosinophil function, driving changes in the transcriptional profile and/or promoting degranulation.23,44 For example, Bouffi et al.,23 show that IL-33 stimulation of eosinophils results in the alteration of over 500 genes resulting in the upregulation of various factors such as IL-6, IL-13, CCL17, and TGF-β. In addition, IL-33 stimulation of eosinophils results in autocrine production of IL-4, which synergizes with ongoing IL-33 signaling to regulate the eosinophil transcriptome through an NFκB-dependent mechanism. However, while the IL-4 autocrine-loop seemed to amplify IL-33 mediated activation of eosinophils, it does not account for all noted alterations, as upregulation of genes such as IL-6 and IL-13 seem to be independent of this process.23 Eosinophil-mediated disorders such as asthma, eosinophilic esophagitis (EoE) and inflammatory bowel disease (IBD), are known to have significant input from IL-33 signaling.39,45,46 However, it is unclear whether dysregulation of IL-33 (and consequently—responsive cells like eosinophils) is involved in the initiation of the inflammatory response or is rather involved in its propagation.
IL-33 can also affect eosinophil function through indirect mechanisms (Fig. 1). IL-33 is a potent activator of tissue ILC2s promoting their expansion and cytokine production. ILC2s are lineage-negative, GATA3+ cells that are a major source of IL-13, IL-5, and amphiregulin in the early phase of type II inflammatory responses such as helminth infection, asthma and allergy.25 ILC2 effector function in inflammation is achieved primarily through regulation of eosinophils (via IL-5) and goblet cell hyperplasia and mucus secretion (via IL-13). However, ILC2 are also a source of these Type II cytokines in certain tissues, like adipose, lung and intestine, under homeostatic condition.16,26,47 Lastly, IL-33 can induce CCL11 production in fibroblasts, which potentiates eosinophil recruitment.48,49
Another epithelial-derived cytokine capable of regulating eosinophil function is IL-25, which signals through IL17RB.25,50,51 Similar to IL-33, IL-25 can also induce expansion of ILC2s and stimulate their production of IL-5 and 13, highlighting a degree of functional redundancy. However, unlike the IL-33 receptor ST2, it is still unclear whether or not eosinophils truly express the IL17RB receptor under homeostatic conditions. Eosinophil upregulation of surface IL17RB has been observed in a few studies, but always in the context of inflammatory responses such as allergic asthma.52
Contributions of tissue-resident eosinophils to tissue homeostasis
Despite the long-observed phenomenon of eosinophil presence in healthy tissues at steady-state conditions, the dogmatic view of eosinophil function is that they evolved exclusively as part of an early innate immune response against parasitic helminths and additionally function in driving pathophysiological responses in type-II disorders such as asthma and EoE where eosinophilia is a common hallmark phenotype.53,54 The development of eosinophil-deficient mouse models (outlined in Table 2) has enabled researchers to better understand eosinophil function, particularly in inflammatory settings. As the focus of this review/commentary is on homeostatic eosinophil function we refer readers to excellent reviews for more in-depth information on the role of eosinophils in the context of eosinophil-associated disorders1,2,55,56 or anti-parasitic immunity.57,58 These models have also opened new doors and avenues to explore the functional significance of eosinophil residency in tissues under homeostasis. Given that during homeostasis the vast majority of eosinophils in the body reside in peripheral tissues, particularly small and large intestine, adipose tissue, thymus, mammary gland and uterus, it is of interest to understand the functions of eosinophils in these diverse niches and to identify the factors that regulate their recruitment and activation state. Below we discuss relevant studies that have begun to address these fundamental questions, with a focus on mucosal tissues.
Non-mucosal tissues
Although the exact function of thymic eosinophils remains unknown, their post-natal recruitment is associated with the negative selection of MHC class I restricted T cells in the thymus9 and mice lacking eosinophils exhibit impaired clearance of apoptotic cells.59 By contrast, a wealth of literature exists detailing a protective role for eosinophils within adipose tissue. The presence of eosinophils within white adipose tissue (WAT) provides protection against obesity, promotes thermogenic energy expenditure and metabolic homeostasis in mice and this is, at least in part, due to their production of IL-4, which acts to promote the alternative activation of macrophages (see reviews by60,61).
Mammary gland development
Puberty hormones and growth factors trigger mammary gland development, which is characterized by the elongation and branching of the prenatal rudimentary ductal tree. Eosinophils are recruited to the postnatal mammary gland when the formation and outgrowth of terminal end buds (TEBs) occurs.14 Macrophages and eosinophils localize around the TEBs and represent the major leukocyte populations present. Their contribution to ductal outgrowth was demonstrated using sub-lethal irradiation and mice deficient in the gene encoding CSF1, a major macrophage growth factor.14 In the same study the authors used CCL11 deficient mice to show that this chemokine was the main factor responsible for eosinophil localization around the ETBs and noted reduced mammary gland branch formation. The authors conclude that macrophages regulate the elongation of the ductal tree whilst eosinophils contribute to ductal branching, perhaps by providing inhibitory signals at branching points.62 Intriguingly, eosinophils are again recruited to the mammary gland during pregnancy when further ductal growth occurs63 and nursing of pups on IL-5 deficient dams was observed to result in a decreased survival of weanlings.64
Endometrial maintenance
Eosinophils are also recruited to the healthy uterus during the degradation cycle of oestrus to metoestrus. Most of the eosinophils present are located in the endometrium adjacent to the muscular layer.13 Eosinophil recruitment and degranulation coincide with tissue-degradation and subsequent uterine repair and remodeling processes.65,66 Rodent eosinophils are also recruited to the cervix during gestation, increasing significantly during labor, when tissue degradative and tissue regenerative processes occur.67,68 However, despite this observation, eosinophil-deficient mice exhibit normal pregnancy and parturition, suggesting that the role of eosinophils in this process is redundant, or that adaptation takes place to compensate for its absence.13 Nonetheless, a recent study by Vicetti Miguel et al.,17 suggests that eosinophils play a critical role in endometrial maintenance during infection with Chlamydia trachomatis. Genital C. trachomatis infection promoted type II immune responses, with IL-4 signaling required for clearing infection and preventing endometrial damage.17 The use of IL-4-IRES-eGPF (4get) mice revealed eosinophils as the main source of IL-4 present during, suggesting a central role for these cells in promoting endometrial repair.17
Lung development and immunity
Eosinophils are not present in the lung of mice at birth but increase (together with ILC2s, basophils and mast cells) to reach maximal levels between postnatal day 7 and 14.69 Their recruitment follows IL-33 release in response to the ‘first breath’ and is closely associated with the postnatal lung ‘alveolarization period’ that is characterized by epithelial cell differentiation, angiogenesis and ECM remodeling.11 IL-33 plays a role in in promoting type II immunity and functions to maintain alveolar macrophages with a M2 phenotype that reduce pro-inflammatory responses, although they do not impact on postnatal alveolarization.11,69 However early IL-33 production also results in delayed anti-bacterial effector responses11 and promotes sensitization towards allergens.69 Activated eosinophils have been reported to provide protection against lethal infection with rodent pneumovirus,70 raising the possibility that these cells may be present in the neonatal lung to provide an extra level so innate defence against respiratory viruses. In contrast to the mouse neonatal lung, the healthy adult lung contains very few resident eosinophils,10,69,71 yet intriguingly these cells may serve a regulatory function even at low numbers. Mesnil et al.,10 demonstrated that the adult asthmatic lung contains two populations of eosinophils, with ‘homeostatic’ or resident eosinophils characterized as Siglec-FhiCD62L+CD101− cells, and ‘inflammatory’ or recruited eosinophils as Siglec-FhiCD62L−CD101+ cells. Furthermore, resident eosinophils were important for limiting Th2-responses occurring in response to house-dust mite challenge.10
Immune homeostasis and barrier integrity in the intestine
Under homeostatic conditions the intestine is home to the largest pool of eosinophils in the body.71,72 As mentioned earlier their presence is microbiota-independent, but requires both CCL11 production8 and the presence of ILC2s26 (Fig. 2). Their distribution is site specific, with the proximal small intestine harboring the highest frequency (~10% of total leukocytes) in mice. Despite the high numbers of intestinal eosinophils under homeostasis, their purpose in this tissue remains largely debated. Yet, out of all mucosal sites, tissue-resident eosinophils in the intestine are arguably the most studied under homeostatic conditions. Importantly, recent studies are revealing eosinophils as critical regulators of immune homeostasis within the intestine (Fig. 2).
The small intestine is home to a large population of eosinophils under homeostatic conditions. Here they communicate to immune cells such as IgA+ plasma cells, innate lymphoid cells (ILCs) and macrophages via the release of cytokines including IL-4, IL-13, IL-5, GM-CSF, IL-1b, BAFF, TGFb, and IL-6. They are recruited via ILC2-derived IL-5 and the chemokine CCL11, and their function and/or survival may be modulated by other secreted factors including the alarmin IL-33, and tissue specific cues such as acidic pH. Eosinophils may also promote epithelial cell renewal through the release of amphiregulin—and regulate tissue remodeling by activating fibroblasts, by virtue of their ability to release metalloproteinases (MMPs) and the stromal cell growth factors, fibroblast growth factor (FGF), nerve growth factor (NGF) and vascular endothelial growth factor (VEGF).
A key function for intestinal eosinophils during homeostasis may be the regulation of mucosal IgA (Fig. 2). IgA+ B cells and IgA-secreting plasma cells are very abundant in the small intestine resulting in production of vast quantities of IgA that are transferred into the intestinal lumen. Much of the IgA produced in the small intestine has specificity against intestinal microbes and homeostatic anti-microbial secretory IgA has been suggested to mediate multiple functions including immune exclusion, inhibition of bacterial motility, neutralization, modification of microbial gene expression, enhancement of antigen uptake, and/or providing a protective niche for certain microbes (see review73). Given the potential importance of IgA in mucosal homeostasis, understanding the factors that regulate IgA plasma cell survival and turn-over is important—and two studies using mice deficient in eosinophils reported decreased serum, intestinal and fecal IgA levels and reduced numbers of intestinal IgA+ B cells and IgA-secreting plasma cells.74,75 One also reported dysregulated mucus production and an altered microbiota.74 Eosinophils might mediate their effect on IgA producing cells through a couple different mechanisms. Chu et al.,74 implicated eosinophil production of B cell-activating factor belonging to the TNF family (BAFF), IL-6, TGF-β, and matrix metalloproteinase 9 (MMP9), whereas Jung et al.,75 identified an alternative pathway involving eosinophil production of IL-1β and lymphotoxins. Intestinal eosinophils can express many toll-like receptors (TLRs),74 and thus might sense microbial signals, which could modulate their cytokine production and ultimately regulate anti-microbial IgA production. In contrast to these reports, a later study by Forman et al.,76 found only modest reductions in IgA+ B cell numbers for the small intestine, and no difference for the large intestine. Eosinophils have also been implicated in regulation of intestinal T cells. Notably, separate reports detailed eosinophil-deficient mice as harboring fewer Foxp3+ Treg,74 and more Th17 cells,77 in the small intestine under steady-state conditions. Although the exact mechanism(s) as to how eosinophils impact on T cell responses remain unknown, fewer Tregs in eosinophil-deficient mice correlate with decreased TGFβ activating factors, MMP3 and MMP9,74 and the absence of GM-CSF-induced IL-1Rα expression by eosinophils may contribute to reduced Th17 cell numbers.77
While these studies demonstrate that eosinophils exert tissue-protective functions in the intestine, there are discrepancies regarding their impact on IgA. It is also important to note that reported studies did not always compare the phenotype of eosinophil-deficient mice to co-housed or littermate wild-type control, and microbial differences present in separately housed cohorts of mice may lead to altered immune function independently of genotype-driven changes.78 Nevertheless, it is clear is that eosinophils are able to secrete a multitude of factors and may impact on intestinal homeostasis through the regulation of immune cells, fibroblasts and/or epithelial cells (Fig. 2). At the current time the processes outlined in Fig. 2 remain hypothetical and further studies in which environmental factors, such as the diet and microbiota, are carefully controlled will be necessary to elucidate the exact homeostatic function of eosinophils within the healthy intestine.
LIAR hypothesis revisited: perspectives on the functional significance of mucosal eosinophils
In 2010, Lee53 and colleagues published a provocative hypothesis challenging the dogmatic view of eosinophils as solely evolving for the purpose of functioning as innate immune end-stage effector cells in type II responses. The LIAR hypothesis, which states that eosinophils are intrinsically homeostatic cells that regulate Local Immunity And/or Remodeling/Repair, proposes an alternate view which aims to unify the seemingly paradoxical phenomenon of eosinophil accumulation in healthy tissues and their role in immune defense. Furthermore, it proposes that the mechanisms by which eosinophils propagate pathophysiological responses may be the very same mechanisms utilized in the maintenance of tissue homeostasis in healthy tissues where they reside. In this model, the key determinant of eosinophil effector functions, such as the magnitude and extent of activities, is largely dependent on the context in a given tissue microenvironment. Indeed, tissue-resident eosinophils exhibit properties distinct from mature cells newly released from the bone marrow. These findings suggest that eosinophils integrate micro-environmental cues that may shape their function and/or context-specific roles within a given tissue. However, the precise factors and mechanisms that underlie these altered states and the implications for tissue-resident eosinophil function remain unclear.
As new data expand our understanding of the functional significance of these cells in health and disease, we are constantly reminded of the complex nature and multi-functional capability of these cells—and the ideas proposed in the LIAR hypothesis become ever more relevant.
Addressing the role of the microenvironment in shaping eosinophil function
The pressing need for identification of micro-environmental factors regulating eosinophil function is most evident when considering the varied and sometimes contradictory findings in studies on tissue-resident eosinophils in the intestine. The discrepancies apparent between these studies likely highlight the influence of the microenvironment (particularly the microbiota) on eosinophil function. Prenatal mice have detectable eosinophils in their intestines and similar levels of eosinophils are found in the adult intestines of germ-free and colonized mice, indicating that the microbiota is not required for eosinophil recruitment to this tissue (Table 1 and ref. 8). However, this does not rule out a potential role for the microbiome in modulating eosinophil survival or effector function. This theoretically could occur directly via microbial-associated molecular patterns (MAMPS) or indirectly through microbial release of metabolites. The signals could also impact eosinophils directly or indirectly through microbiota regulation of other immune cells or stromal cells (i.e., epithelial cells, fibroblasts or sensory nerves), which could then produce factors that modulate eosinophil activity. In addition, feeding may influence eosinophil residency in the intestine as the number of circulating eosinophils has long been known to increase in response to ingested food,79 and intestinal ILC2s have been shown to enhance their secretion of IL-5 and IL-13 in response to the release of vasoactive intestinal peptide (VIP) following food intake26 (Fig. 2). Both ILC2s and eosinophils also respond to IL-33 which is abundantly expressed in the healthy intestine (Fig. 2), although a contribution of this cytokine to the recruitment or function of resident intestinal eosinophils has not been determined. Intriguingly, eosinophils were shown to respond to acidic conditions by virtue of their expression of GPR65,80 implicating a possible role for this receptor in their intestinal recruitment (Fig. 2). Although we specifically discuss the intestine here, the same rules could apply to other mucosal sites such as the lung and the uterus that have their own unique composition of cellular networks and microbial communities.
It remains to be clarified whether eosinophil plasticity or heterogeneity underlies the apparent ability of these cells to contribute to mucosal tissue homeostasis during the steady state, versus their proposed pathological role in disease states such as asthma, EoE1, and colitis.81 Tissue eosinophils may adopt different phenotypes depending on the microenvironment in which they find themselves (plasticity). Alternatively, tissue-resident eosinophils and eosinophils newly recruited from the bloodstream in response to inflammation could exhibit different phenotypes (heterogeneity), as recently described for the lung.10 Future studies comparing eosinophil phenotype and function in healthy versus diseased mucosal tissues are needed.
A role for mucosal eosinophils in homeostatic tissue remodeling?
The form and function of tissues is highly dependent on the ECM, a mesh-like network of molecules, including proteins (collagens, elastins), proteoglycans (glycosaminoglycans, heparan sulfate), and hyaluronic acid. All cells, including eosinophils, form close interactions with the ECM, which occur predominately through cell surface receptors (integrins) and biophysical stimuli (mechanical stiffness) (reviewed in refs. 82,83,84). The ECM not only determines tissue structure but also regulates many cellular processes through determining the bio-availability of cytokines, chemokines and growth factors,85,86,87 and by directly influencing the migration and activation of cells83,88 hence influencing immune responses. Disruption of the homeostatic structure of the ECM results in a process of wound healing that involves four overlapping phases: the hemostatic phase, inflammatory phase, proliferation stage and remodeling stage.89 Restoration of the ECM to is original state re-establishes homeostasis, whilst excessive deposition of connective tissue or inadequate remodeling can result in fibrosis.
Eosinophils are associated with the reinstatement of tissue homeostasis in some tissues, playing critical roles in driving regenerative responses in models of liver90 and muscle injury91 in an IL-4 dependent manner. In keeping with the ability of eosinophils to alter muscle development, these cells are reported to promote helminth survival following primary infection with Trichinella spiralis.92,93 In this case eosinophils promote the growth of larvae within skeletal muscle,92 likely by enhancing nutrient metabolism within the infected cell and creating a nutrient-rich niche for the larvae.93 This involves IL-4 production by eosinophils, which also functions to suppress pro-inflammatory responses in the surrounding tissue.93 Of note, however, a secondary challenge of mice with the same helminth lead to the more classically observed ability of eosinophils to provide protective immunity,94 a function that is typically associated with eosinophil activation in response to antibody binding to helminth antigens leading to adherence of the cells to the worm and the release of toxic granule proteins (reviewed in ref. 95). Lastly, tumors are typically thought to represent a form of tissue injury and eosinophils have been noted to drive anti-tumoral responses.96,97 Mechanistically, the beneficial role of eosinophils under these contexts requires crosstalk and coordination with complex cellular networks encompassing other immune cells and stromal cells in a manner that blurs the lines between canonical type I and type II immune responses.
It is interesting to note that although, tissues that harbor resident eosinophils are heterogenous with regards to cellular complexity and physiology/function, they are often sites that experience homeostatic remodeling (e.g., uterus, mammary gland), mechanical stress (e.g., intestine, lung) and/or cellular turnover (e.g., thymus, intestine, uterus), as was predicated by the LIAR hypothesis. As mentioned above eosinophils also accumulate in certain sites (liver, muscle) in the context of injury, further highlighting their affinity to sites that experience significant cellular turnover and/or remodeling. These observations—and the commonalities between them—warrant further investigation as they could provide much needed insight into the functional significance of eosinophil tissue accumulation during homeostasis. Once within the tissue undergoing remodeling, eosinophils could integrate signals from the microenvironment and tailor their function accordingly. Whether eosinophils would then promote homeostatic or pathophysiological remodeling would thus likely depend on (1) other cell types present, (2) external stimuli (microbiota, diet, pathogens, extent of the tissue damage), (3) physical environment (e.g., ECM, pH, mechanical stimuli) and (4) balance between inflammatory vs. anti-inflammatory signals (Fig. 2).
As mentioned earlier eosinophils are equipped with ability to interact with the ECM via expression of specific integrins (CD11c, CD11b, α5 integrins) and their presence during pathological inflammation (EoE, transplantation, allergy) is often associated with fibrosis1 Under conditions of allergic inflammation airway eosinophils exhibit a hyper adhesive phenotype towards a provisional ‘immature’ matrix that is deposited following tissue injury98 and their presence promotes collagen deposition and increased airway smooth muscle.99 Eosinophils may regulate the deposition and/or remodeling of the ECM through numerous mechanisms including the direct activation of fibroblasts to secrete matrix components,100 the remodeling of these components through the release of MMP9101 and the regulation of heparanase activity102 (Fig. 2). Eosinophils also modulate the secretion of various stromal cell growth factors including fibroblast growth factor (FGF), nerve growth factor (NGF) and vascular endothelial growth factor (VEGF)103 (Fig. 2). Lastly, eosinophils may directly impact renewal of the epithelial barrier as human eosinophils can release amphiregulin104 (Fig. 2), which is intimately involved in promoting the proliferation of epithelial cells in the intestine,105 lung106 and mammary gland.107 In contrast to this finding, mice expressing both the IL-5 and eotaxin2 transgene’s harbor increased activated eosinophils in the lung and exhibit reduced barrier function.108 Thus whether eosinophils promote or reduce the integrity of epithelial layers remains unclear. Last but not least eosinophils share many functions with mast cells and these two cells often interact with each other109 indicating that they may contribute in a paired manner to tissue function.
The modulation of stromal cell function (fibroblasts, neurons, epithelial cells) by eosinophils present in healthy mucosal tissues is likely (Fig. 2) but remains to be addressed experimentally (with the exception of the mammary gland where they are closely associated with ductal elongation). Nor has the possible contribution of eosinophils to homeostatic tissue remodeling been investigated. The latter point represents a particularly important point for mucosal tissues like the intestine that are constantly subject to environmental and physical stressors necessitating timely regeneration. Dysregulation of this homeostatic remodeling, or a failure to regulate the mechanical properties of the ECM during reparative responses, would be expected to impact on a variety of process cellular and tissue functions at mucosal sites.
Concluding remarks
The evidence cited in this review supports the view that in addition to their commonly cited role in mediating tissue damage in various disorders, eosinophils participate in ongoing processes through immune regulation and tissue remodeling, with important implications for the local tissue microenvironment.
Although our appreciation of the entire spectrum of eosinophil functions has improved markedly over the last decade, we still have much to learn before the full significance of eosinophil residency within healthy mucosal tissues will be appreciated. With new tools and a broader appreciation of the possible roles of these enigmatic cells, the field is poised to move quickly and will likely deliver intriguing new insights into the functioning of homeostatic mucosal tissues. We suggest that only by deciphering the contribution of eosinophils to the complex, multi-faceted, systems that regulate homeostasis will we truly understand the etiology of the complex diseases and disorders with which eosinophils are historically associated and develop novel treatments that are targeted and efficient. This will likely require investigators utilizing models of eosinophil deficiency to look beyond cell–cell interactions to incorporate biochemical and physiological measurements of tissue function. It will also involve determining the possible existence of distinct eosinophil subsets and the impact of distinct tissue microenvironments.
References
Rosenberg, H. F., Dyer, K. D. & Foster, P. S. Eosinophils: changing perspectives in health and disease. Nat. Rev. Immunol. 13, 9–22 (2013).
Rothenberg, M. E. & Hogan, S. P. The eosinophil. Annu. Rev. Immunol. 24, 147–174 (2006).
Weller, P. F. & Spencer, L. A. Functions of tissue-resident eosinophils. Nat. Rev. Immunol. 17, 746–760 (2017).
Ahrens, R. et al. Intestinal macrophage/epithelial cell-derived CCL11/eotaxin-1 mediates eosinophil recruitment and function in pediatric ulcerative colitis. J. Immunol. 181, 7390–7399 (2008).
Gounni, A. S. et al. IL-9-mediated induction of eotaxin1/CCL11 in human airway smooth muscle cells. J. Immunol. 173, 2771–2779 (2004).
Matthews, A. N. et al. Eotaxin is required for the baseline level of tissue eosinophils. Proc. Natl Acad. Sci. USA 95, 6273–6278 (1998).
Salcedo, R. et al. Eotaxin (CCL11) induces in vivo angiogenic responses by human CCR3+ endothelial cells. J. Immunol. 166, 7571–7578 (2001).
Mishra, A., Hogan, S. P., Lee, J. J., Foster, P. S. & Rothenberg, M. E. Fundamental signals that regulate eosinophil homing to the gastrointestinal tract. J. Clin. Invest. 103, 1719–1727 (1999).
Throsby, M., Herbelin, A., Pleau, J. M. & Dardenne, M. CD11c+ eosinophils in the murine thymus: developmental regulation and recruitment upon MHC class I-restricted thymocyte deletion. J. Immunol. 165, 1965–1975 (2000).
Mesnil, C. et al. Lung-resident eosinophils represent a distinct regulatory eosinophil subset. J. Clin. Invest. 126, 3279–3295 (2016).
Saluzzo, S. et al. First-breath-induced type 2 pathways shape the lung immune environment. Cell Rep. 18, 1893–1905 (2017).
Marichal, T., Mesnil, C. & Bureau, F. Homeostatic eosinophils: characteristics and functions. Front. Med. 4, 101 (2017).
Gouon-Evans, V. & Pollard, J. W. Eotaxin is required for eosinophil homing into the stroma of the pubertal and cycling uterus. Endocrinology 142, 4515–4521 (2001).
Gouon-Evans, V., Rothenberg, M. E. & Pollard, J. W. Postnatal mammary gland development requires macrophages and eosinophils. Development 127, 2269–2282 (2000).
Goldberg, E. L. & Dixit, V. D. Editorial: “Crowning” eosinophils in adipose tissue: does location matter? J. Leukoc. Biol. 98, 451–452 (2015).
Wu, D. et al. Eosinophils sustain adipose alternatively activated macrophages associated with glucose homeostasis. Science 332, 243–247 (2011).
Vicetti Miguel, R. D. et al. IL-4-secreting eosinophils promote endometrial stromal cell proliferation and prevent Chlamydia-induced upper genital tract damage. Proc. Natl Acad. Sci. USA 114, E6892–E6901 (2017).
Greenfeder, S., Umland, S. P., Cuss, F. M., Chapman, R. W. & Egan, R. W. Th2 cytokines and asthma. The role of interleukin-5 in allergic eosinophilic disease. Respir. Res. 2, 71–79 (2001).
Takatsu, K. & Nakajima, H. IL-5 and eosinophilia. Curr. Opin. Immunol. 20, 288–294 (2008).
Foster, P. S. et al. Elemental signals regulating eosinophil accumulation in the lung. Immunol. Rev. 179, 173–181 (2001).
Yang, M. et al. Eotaxin-2 and IL-5 cooperate in the lung to regulate IL-13 production and airway eosinophilia and hyperreactivity. J. Allergy Clin. Immunol. 112, 935–943 (2003).
Bagnasco, D., Ferrando, M., Varricchi, G., Passalacqua, G. & Canonica, G. W. A critical evaluation of anti-IL-13 and anti-IL-4 strategies in severe asthma. Int. Arch. Allergy Immunol. 170, 122–131 (2016).
Bouffi, C. et al. IL-33 markedly activates murine eosinophils by an NF-kappaB-dependent mechanism differentially dependent upon an IL-4-driven autoinflammatory loop. J. Immunol. 191, 4317–4325 (2013).
Pope, S. M. et al. IL-13 induces eosinophil recruitment into the lung by an IL-5- and eotaxin-dependent mechanism. J. Allergy Clin. Immunol. 108, 594–601 (2001).
Neill, D. R. et al. Nuocytes represent a new innate effector leukocyte that mediates type-2 immunity. Nature 464, 1367–1370 (2010).
Nussbaum, J. C. et al. Type 2 innate lymphoid cells control eosinophil homeostasis. Nature 502, 245–248 (2013).
Schneider, C. et al. A metabolite-triggered tuft cell-ILC2 circuit drives small intestinal remodeling. Cell 174, 271–284 e214 (2018).
Mao, H. et al. Mechanisms of Siglec-F-induced eosinophil apoptosis: a role for caspases but not for SHP-1, Src kinases, NADPH oxidase or reactive oxygen. PLoS ONE 8, e68143 (2013).
Zhang, M. et al. Defining the in vivo function of Siglec-F, a CD33-related Siglec expressed on mouse eosinophils. Blood 109, 4280–4287 (2007).
Zimmermann, N. et al. Siglec-F antibody administration to mice selectively reduces blood and tissue eosinophils. Allergy 63, 1156–1163 (2008).
Anwar, A. R., Walsh, G. M., Cromwell, O., Kay, A. B. & Wardlaw, A. J. Adhesion to fibronectin primes eosinophils via alpha 4 beta 1 (VLA-4). Immunology 82, 222–228 (1994).
Munoz, N. M. et al. Eosinophil VLA-4 binding to fibronectin augments bronchial narrowing through 5-lipoxygenase activation. Am. J. Physiol. 270, L587–L594 (1996).
Dubucquoi, S. et al. Interleukin 5 synthesis by eosinophils: association with granules and immunoglobulin-dependent secretion. J. Exp. Med. 179, 703–708 (1994).
Muraki, M., Gleich, G. J. & Kita, H. Antigen-specific IgG and IgA, but not IgE, activate the effector functions of eosinophils in the presence of antigen. Int Arch. Allergy Immunol. 154, 119–127 (2011).
Willebrand, R. & Voehringer, D. IL-33-induced cytokine secretion and survival of mouse eosinophils is promoted by autocrine GM-CSF. PLoS ONE 11, e0163751 (2016).
Cayrol, C. & Girard, J. P. IL-33: an alarmin cytokine with crucial roles in innate immunity, inflammation and allergy. Curr. Opin. Immunol. 31, 31–37 (2014).
Cherry, W. B., Yoon, J., Bartemes, K. R., Iijima, K. & Kita, H. A novel IL-1 family cytokine, IL-33, potently activates human eosinophils. J. Allergy Clin. Immunol. 121, 1484–1490 (2008).
Johnston, L. K. & Bryce, P. J. Understanding interleukin 33 and its roles in eosinophil development. Front. Med. 4, 51 (2017).
Hodzic, Z., Schill, E. M., Bolock, A. M. & Good, M. IL-33 and the intestine: The good, the bad, and the inflammatory. Cytokine 100, 1–10 (2017).
Rankin, A. L. et al. IL-33 induces IL-13-dependent cutaneous fibrosis. J. Immunol. 184, 1526–1535 (2010).
Schmitz, J. et al. IL-33, an interleukin-1-like cytokine that signals via the IL-1 receptor-related protein ST2 and induces T helper type 2-associated cytokines. Immunity 23, 479–490 (2005).
Stolarski, B., Kurowska-Stolarska, M., Kewin, P., Xu, D. & Liew, F. Y. IL-33 exacerbates eosinophil-mediated airway inflammation. J. Immunol. 185, 3472–3480 (2010).
Wen, T., Besse, J. A., Mingler, M. K., Fulkerson, P. C. & Rothenberg, M. E. Eosinophil adoptive transfer system to directly evaluate pulmonary eosinophil trafficking in vivo. Proc. Natl Acad. Sci. USA 110, 6067–6072 (2013).
Willetts, L. et al. Vesicle-associated membrane protein 7-mediated eosinophil degranulation promotes allergic airway inflammation in mice. Commun. Biol. 1, 83 (2018).
Liew, F. Y., Girard, J. P. & Turnquist, H. R. Interleukin-33 in health and disease. Nat. Rev. Immunol. 16, 676–689 (2016).
Pastorelli, L., De Salvo, C., Vecchi, M. & Pizarro, T. T. The role of IL-33 in gut mucosal inflammation. Mediators Inflamm. 2013, 608187 (2013).
Monticelli, L. A. et al. Innate lymphoid cells promote lung-tissue homeostasis after infection with influenza virus. Nat. Immunol. 12, 1045–1054 (2011).
Kurokawa, M. et al. Expression and effects of IL-33 and ST2 in allergic bronchial asthma: IL-33 induces eotaxin production in lung fibroblasts. Int Arch. Allergy Immunol. 155, 12–20 (2011).
Masterson, J. C. et al. Eosinophils and IL-33 perpetuate chronic inflammation and fibrosis in a pediatric population with stricturing Crohn’s Ileitis. Inflamm. Bowel Dis. 21, 2429–2440 (2015).
Fort, M. M. et al. IL-25 induces IL-4, IL-5, and IL-13 and Th2-associated pathologies in vivo. Immunity 15, 985–995 (2001).
Salimi, M. et al. A role for IL-25 and IL-33-driven type-2 innate lymphoid cells in atopic dermatitis. J. Exp. Med 210, 2939–2950 (2013).
Tang, W. et al. IL-25 and IL-25 receptor expression on eosinophils from subjects with allergic asthma. Int Arch. Allergy Immunol. 163, 5–10 (2014).
Lee, J. J., Jacobsen, E. A., McGarry, M. P., Schleimer, R. P. & Lee, N. A. Eosinophils in health and disease: the LIAR hypothesis. Clin. Exp. Allergy 40, 563–575 (2010).
Wills-Karp, M. & Karp, C. L. Biomedicine. Eosinophils in asthma: remodeling a tangled tale. Science 305, 1726–1729 (2004).
O’Sullivan, J. A. & Bochner, B. S. Eosinophils and eosinophil-associated diseases: an update. J. Allergy Clin. Immunol. 141, 505–517 (2018).
Spencer, L. A. & Weller, P. F. Eosinophils and Th2 immunity: contemporary insights. Immunol. Cell Biol. 88, 250–256 (2010).
Behm, C. A. & Ovington, K. S. The role of eosinophils in parasitic helminth infections: insights from genetically modified mice. Parasitol. Today 16, 202–209 (2000).
Huang, L. & Appleton, J. A. Eosinophils in Helminth Infection: Defenders and Dupes. Trends Parasitol. 32, 798–807 (2016).
Kim, H. J., Alonzo, E. S., Dorothee, G., Pollard, J. W. & Sant’Angelo, D. B. Selective depletion of eosinophils or neutrophils in mice impacts the efficiency of apoptotic cell clearance in the thymus. PLoS ONE 5, e11439 (2010).
Bolus, W. R. & Hasty, A. H. Contributions of innate type 2 inflammation to adipose function. J. Lipid Res. 60, 1698–1709 (2019).
Zhang, Y. et al. Eosinophils reduce chronic inflammation in adipose tissue by secreting Th2 cytokines and promoting M2 macrophages polarization. Int J. Endocrinol. 2015, 565760 (2015).
Gouon-Evans, V., Lin, E. Y. & Pollard, J. W. Requirement of macrophages and eosinophils and their cytokines/chemokines for mammary gland development. Breast Cancer Res. 4, 155–164 (2002).
Aupperlee, M. D. et al. Epidermal growth factor receptor (EGFR) signaling is a key mediator of hormone-induced leukocyte infiltration in the pubertal female mammary gland. Endocrinology 155, 2301–2313 (2014).
Colbert, D. C., McGarry, M. P., O’Neill, K., Lee, N. A. & Lee, J. J. Decreased size and survival of weanling mice in litters of IL-5-/-mice are a consequence of the IL-5 deficiency in nursing dams. Contemp. Top. Lab Anim. Sci. 44, 53–55 (2005).
Perez, M. C., Furth, E. E., Matzumura, P. D. & Lyttle, C. R. Role of eosinophils in uterine responses to estrogen. Biol. Reprod. 54, 249–254 (1996).
Tchernitchin, A. N. & Galand, P. Oestrogen levels in the blood, not in the uterus, determine uterine eosinophilia and oedema. J. Endocrinol. 99, 123–130 (1983).
Knudsen, U. B., Uldbjerg, N., Rechberger, T. & Fredens, K. Eosinophils in human cervical ripening. Eur. J. Obstet. Gynecol. Reprod. Biol. 72, 165–168 (1997).
Timmons, B. C., Fairhurst, A. M. & Mahendroo, M. S. Temporal changes in myeloid cells in the cervix during pregnancy and parturition. J. Immunol. 182, 2700–2707 (2009).
de Kleer, I. M. et al. Perinatal Activation of the Interleukin-33 Pathway Promotes Type 2 Immunity in the Developing Lung. Immunity 45, 1285–1298 (2016).
Percopo, C. M. et al. Activated mouse eosinophils protect against lethal respiratory virus infection. Blood 123, 743–752 (2014).
Chojnacki, A. et al. Intravital imaging allows real-time characterization of tissue resident eosinophils. Commun. Biol. 2, 181 (2019).
Kato, M. et al. Eosinophil infiltration and degranulation in normal human tissue. Anat. Rec. 252, 418–425 (1998).
Bunker, J. J. & Bendelac, A. IgA responses to microbiota. Immunity 49, 211–224 (2018).
Chu, V. T. et al. Eosinophils promote generation and maintenance of immunoglobulin-A-expressing plasma cells and contribute to gut immune homeostasis. Immunity 40, 582–593 (2014).
Jung, Y. et al. IL-1beta in eosinophil-mediated small intestinal homeostasis and IgA production. Mucosal Immunol. 8, 930–942 (2015).
Forman, R. et al. Eosinophils may play regionally disparate roles in influencing IgA(+) plasma cell numbers during large and small intestinal inflammation. BMC Immunol. 17, 12 (2016).
Sugawara, R. et al. Small intestinal eosinophils regulate Th17 cells by producing IL-1 receptor antagonist. J. Exp. Med. 213, 555–567 (2016).
Mamantopoulos, M., Ronchi, F., McCoy, K. D. & Wullaert, A. Inflammasomes make the case for littermate-controlled experimental design in studying host-microbiota interactions. Gut Microbes 9, 374–381 (2018).
Lungu, A. & Manitescu, F. [The evolution of the daily rhythm of the circulating eosinophils during inanition and resumption of feeding]. Stud. Cercet Endocrinol. 13, 31–38 (1962).
Kottyan, L. C. et al. Eosinophil viability is increased by acidic pH in a cAMP- and GPR65-dependent manner. Blood 114, 2774–2782 (2009).
Griseri, T. et al. Granulocyte macrophage colony-stimulating factor-activated eosinophils promote interleukin-23 driven chronic colitis. Immunity 43, 187–199 (2015).
Bonnans, C., Chou, J. & Werb, Z. Remodelling the extracellular matrix in development and disease. Nat. Rev. Mol. Cell Biol. 15, 786–801 (2014).
Humphrey, J. D., Dufresne, E. R. & Schwartz, M. A. Mechanotransduction and extracellular matrix homeostasis. Nat. Rev. Mol. Cell Biol. 15, 802–812 (2014).
Hynes, R. O. & Naba, A. Overview of the matrisome–an inventory of extracellular matrix constituents and functions. Cold Spring Harb. Perspect. Biol. 4, a004903 (2012).
Alcaraz, L. B. et al. Tenascin-X promotes epithelial-to-mesenchymal transition by activating latent TGF-beta. J. Cell Biol. 205, 409–428 (2014).
Robertson, I. B. & Rifkin, D. B. Regulation of the Bioavailability of TGF-beta and TGF-beta-related proteins. Cold Spring Harb. Perspect. Biol. 8, https://doi.org/10.1101/cshperspect.a021907 (2016).
Wohl, A. P., Troilo, H., Collins, R. F., Baldock, C. & Sengle, G. Extracellular regulation of bone morphogenetic protein activity by the microfibril component fibrillin-1. J. Biol. Chem. 291, 12732–12746 (2016).
Schwartz, M. A. Integrins and extracellular matrix in mechanotransduction. Cold Spring Harb. Perspect. Biol. 2, a005066 (2010).
Chester, D. & Brown, A. C. The role of biophysical properties of provisional matrix proteins in wound repair. Matrix Biol. 60–61, 124–140 (2017).
Goh, Y. P. et al. Eosinophils secrete IL-4 to facilitate liver regeneration. Proc. Natl Acad. Sci. USA 110, 9914–9919 (2013).
Heredia, J. E. et al. Type 2 innate signals stimulate fibro/adipogenic progenitors to facilitate muscle regeneration. Cell 153, 376–388 (2013).
Fabre, V. et al. Eosinophil deficiency compromises parasite survival in chronic nematode infection. J. Immunol. 182, 1577–1583 (2009).
Huang, L. et al. Eosinophils and IL-4 support nematode growth coincident with an innate response to tissue injury. PLoS Pathog. 11, e1005347 (2015).
Huang, L. et al. Eosinophils mediate protective immunity against secondary nematode infection. J. Immunol. 194, 283–290 (2015).
Klion, A. D. & Nutman, T. B. The role of eosinophils in host defense against helminth parasites. J. Allergy Clin. Immunol. 113, 30–37 (2004).
Carretero, R. et al. Eosinophils orchestrate cancer rejection by normalizing tumor vessels and enhancing infiltration of CD8(+) T cells. Nat. Immunol. 16, 609–617 (2015).
Hollande, C. et al. Inhibition of the dipeptidyl peptidase DPP4 (CD26) reveals IL-33-dependent eosinophil-mediated control of tumor growth. Nat. Immunol. 20, 257–264 (2019).
Barthel, S. R., Jarjour, N. N., Mosher, D. F. & Johansson, M. W. Dissection of the hyperadhesive phenotype of airway eosinophils in asthma. Am. J. Respir. Cell Mol. Biol. 35, 378–386 (2006).
Humbles, A. A. et al. A critical role for eosinophils in allergic airways remodeling. Science 305, 1776–1779 (2004).
Flood-Page, P. et al. Anti-IL-5 treatment reduces deposition of ECM proteins in the bronchial subepithelial basement membrane of mild atopic asthmatics. J. Clin. Invest. 112, 1029–1036 (2003).
Okada, S., Kita, H., George, T. J., Gleich, G. J. & Leiferman, K. M. Migration of eosinophils through basement membrane components in vitro: role of matrix metalloproteinase-9. Am. J. Respir. Cell Mol. Biol. 17, 519–528 (1997).
Temkin, V. et al. Eosinophil major basic protein: first identified natural heparanase-inhibiting protein. J. Allergy Clin. Immunol. 113, 703–709 (2004).
Kariyawasam, H. H. & Robinson, D. S. The role of eosinophils in airway tissue remodelling in asthma. Curr. Opin. Immunol. 19, 681–686 (2007).
Matsumoto, K., Fukuda, S., Nakamura, Y. & Saito, H. Amphiregulin production by human eosinophils. Int Arch. Allergy Immunol. 149, 39–44 (2009).
Monticelli, L. A. et al. IL-33 promotes an innate immune pathway of intestinal tissue protection dependent on amphiregulin-EGFR interactions. Proc. Natl Acad. Sci. USA 112, 10762–10767 (2015).
Schuger, L., Johnson, G. R., Gilbride, K., Plowman, G. D. & Mandel, R. Amphiregulin in lung branching morphogenesis: interaction with heparan sulfate proteoglycan modulates cell proliferation. Development 122, 1759–1767 (1996).
Ciarloni, L., Mallepell, S. & Brisken, C. Amphiregulin is an essential mediator of estrogen receptor alpha function in mammary gland development. Proc. Natl Acad. Sci. USA 104, 5455–5460 (2007).
Ochkur, S. I. et al. Coexpression of IL-5 and eotaxin-2 in mice creates an eosinophil-dependent model of respiratory inflammation with characteristics of severe asthma. J. Immunol. 178, 7879–7889 (2007).
Robida, P. A., Puzzovio, P. G., Pahima, H., Levi-Schaffer, F. & Bochner, B. S. Human eosinophils and mast cells: Birds of a feather flock together. Immunol. Rev. 282, 151–167 (2018).
Carlens, J. et al. Common gamma-chain-dependent signals confer selective survival of eosinophils in the murine small intestine. J. Immunol. 183, 5600–5607 (2009).
Robertson, S. A., Mau, V. J., Young, I. G. & Matthaei, K. I. Uterine eosinophils and reproductive performance in interleukin 5-deficient mice. J. Reprod. Fertil. 120, 423–432 (2000).
Ross, R. & Klebanoff, S. J. The eosinophilic leukocyte. Fine structure studies of changes in the uterus during the estrous cycle. J. Exp. Med. 124, 653–660 (1966).
Bolus, W. R., Gutierrez, D. A., Kennedy, A. J., Anderson-Baucum, E. K. & Hasty, A. H. CCR2 deficiency leads to increased eosinophils, alternative macrophage activation, and type 2 cytokine expression in adipose tissue. J. Leukoc. Biol. 98, 467–477 (2015).
Molofsky, A. B. et al. Innate lymphoid type 2 cells sustain visceral adipose tissue eosinophils and alternatively activated macrophages. J. Exp. Med. 210, 535–549 (2013).
Yu, C. et al. Targeted deletion of a high-affinity GATA-binding site in the GATA-1 promoter leads to selective loss of the eosinophil lineage in vivo. J. Exp. Med. 195, 1387–1395 (2002).
Lee, J. J. et al. Defining a link with asthma in mice congenitally deficient in eosinophils. Science 305, 1773–1776 (2004).
Jacobsen, E. A. et al. Eosinophil activities modulate the immune/inflammatory character of allergic respiratory responses in mice. Allergy 69, 315–327 (2014).
Ochkur, S. I. et al. Frontline Science: eosinophil-deficient MBP-1 and EPX double-knockout mice link pulmonary remodeling and airway dysfunction with type 2 inflammation. J. Leukoc. Biol. 102, 589–599 (2017).
Kopf, M. et al. IL-5-deficient mice have a developmental defect in CD5+ B-1 cells and lack eosinophilia but have normal antibody and cytotoxic T cell responses. Immunity 4, 15–24 (1996).
Lee, N. A. et al. Expression of IL-5 in thymocytes/T cells leads to the development of a massive eosinophilia, extramedullary eosinophilopoiesis, and unique histopathologies. J. Immunol. 158, 1332–1344 (1997).
Doyle, A. D. et al. Homologous recombination into the eosinophil peroxidase locus generates a strain of mice expressing Cre recombinase exclusively in eosinophils. J. Leukoc. Biol. 94, 17–24 (2013).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Shah, K., Ignacio, A., McCoy, K.D. et al. The emerging roles of eosinophils in mucosal homeostasis. Mucosal Immunol 13, 574–583 (2020). https://doi.org/10.1038/s41385-020-0281-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41385-020-0281-y
This article is cited by
-
Cebp1 and Cebpβ transcriptional axis controls eosinophilopoiesis in zebrafish
Nature Communications (2024)
-
Stress-free single-cell transcriptomic profiling and functional genomics of murine eosinophils
Nature Protocols (2024)
-
The elusive Luminal B breast cancer and the mysterious chemokines
Journal of Cancer Research and Clinical Oncology (2023)
-
Bidirectional crosstalk between eosinophils and esophageal epithelial cells regulates inflammatory and remodeling processes
Mucosal Immunology (2021)
-
Mendelian randomization analysis of the association between human blood cell traits and uterine polyps
Scientific Reports (2021)