Abstract
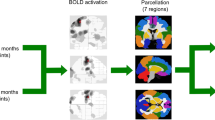
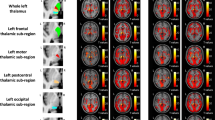
We aimed to investigate abnormal time-varying functional connectivity (FC) for thalamic sub-regions in multiple sclerosis (MS) and their clinical, cognitive and MRI correlates. Eighty-nine MS patients (49 relapsing-remitting [RR] MS; 40 progressive [P] MS) and 53 matched healthy controls underwent neurological, neuropsychological and resting state fMRI assessment. Time-varying connectivity (TVC) was quantified using sliding-window seed-voxel correlation analysis. Standard deviation of FC across windows was taken as measure of TVC, while mean connectivity across windows expressed static FC. MS patients showed reduced TVC vs controls between most of thalamic sub-regions and fronto-temporo-occipital regions. At the same time, they showed increased static FC between all thalamic sub-regions and structurally connected cortico-subcortical regions. TVC reduction was mainly driven by RRMS; while PMS exhibited a variable pattern of TVC abnormalities, characterized by reduced TVC between frontal/motor thalamic seeds and default-mode network areas and increased TVC vs controls/RRMS between posterior thalamic sub-regions and occipito-temporo-insular cortices, associated with severity of clinical disability. Compared with controls, both cognitively preserved and impaired patients showed reduced TVC between anterior thalamic sub-regions and frontal cortex. Cognitively impaired patients also showed increased TVC of the right postcentral thalamic sub-region with the cingulate cortex and postcentral gyrus vs both controls and cognitively preserved patients. Divergent patterns of TVC thalamic abnormalities were found between RRMS and PMS patients. TVC reduction in RRMS may represent the attempt of thalamic network to keep with stable connections. Conversely, increased TVC of posterior thalamic sub-regions characterized PMS and cognitively impaired MS, possibly reflecting maladaptive mechanisms.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Chard DT, Alahmadi AAS, Audoin B, Charalambous T, Enzinger C, Hulst HE et al. Mind the gap: from neurons to networks to outcomes in multiple sclerosis. Nat Rev Neurol. 2021;17:173–84.
Biswal BB, Mennes M, Zuo XN, Gohel S, Kelly C, Smith SM, et al. Toward discovery science of human brain function. Proc Natl Acad Sci USA. 2010;107:4734–9.
Chang C, Glover GH. Time-frequency dynamics of resting-state brain connectivity measured with fMRI. Neuroimage. 2010;50:81–98.
Sakoglu U, Pearlson GD, Kiehl KA, Wang YM, Michael AM, Calhoun VD. A method for evaluating dynamic functional network connectivity and task-modulation: application to schizophrenia. MAGMA. 2010;23:351–66.
Choe AS, Nebel MB, Barber AD, Cohen JR, Xu Y, Pekar JJ, et al. Comparing test-retest reliability of dynamic functional connectivity methods. Neuroimage. 2017;158:155–75.
Hindriks R, Adhikari MH, Murayama Y, Ganzetti M, Mantini D, Logothetis NK, et al. Can sliding-window correlations reveal dynamic functional connectivity in resting-state fMRI? Neuroimage. 2016;127:242–56.
Tomasi D, Shokri-Kojori E, Volkow ND. Temporal changes in local functional connectivity density reflect the temporal variability of the amplitude of low frequency fluctuations in gray matter. PLoS One. 2016;11:e0154407.
Zhang C, Baum SA, Adduru VR, Biswal BB, Michael AM. Test-retest reliability of dynamic functional connectivity in resting state fMRI. Neuroimage. 2018;183:907–18.
Chen J, Sun D, Shi Y, Jin W, Wang Y, Xi Q, et al. Alterations of static functional connectivity and dynamic functional connectivity in motor execution regions after stroke. Neurosci Lett. 2018;686:112–21.
Engels G, Vlaar A, McCoy B, Scherder E, Douw L. Dynamic functional connectivity and symptoms of Parkinson’s disease: a resting-state fMRI study. Front Aging Neurosci. 2018;10:388.
Falahpour M, Thompson WK, Abbott AE, Jahedi A, Mulvey ME, Datko M, et al. Underconnected, but not broken? Dynamic functional connectivity mri shows underconnectivity in autism is linked to increased intra-individual variability across time. Brain Connect. 2016;6:403–14.
Liu A, Lin SJ, Mi T, Chen X, Chan P, Wang ZJ, et al. Decreased subregional specificity of the putamen in Parkinson’s Disease revealed by dynamic connectivity-derived parcellation. Neuroimage Clin. 2018;20:1163–75.
Yue JL, Li P, Shi L, Lin X, Sun HQ, Lu L. Enhanced temporal variability of amygdala-frontal functional connectivity in patients with schizophrenia. Neuroimage Clin. 2018;18:527–32.
Nakajima M, Halassa MM. Thalamic control of functional cortical connectivity. Curr Opin Neurobiol. 2017;44:127–31.
Bisecco A, Rocca MA, Pagani E, Mancini L, Enzinger C, Gallo A, et al. Connectivity-based parcellation of the thalamus in multiple sclerosis and its implications for cognitive impairment: A multicenter study. Hum Brain Mapp. 2015;36:2809–25.
Minagar A, Barnett MH, Benedict RH, Pelletier D, Pirko I, Sahraian MA, et al. The thalamus and multiple sclerosis: modern views on pathologic, imaging, and clinical aspects. Neurology. 2013;80:210–9.
Schoonheim MM, Pinter D, Prouskas SE, Broeders TA, Pirpamer L, Khalil M et al. Disability in multiple sclerosis is related to thalamic connectivity and cortical network atrophy. Mult Scler 2021. https://doi.org/10.1177/13524585211008743.
Rocca MA, Barkhof F, De Luca J, Frisen J, Geurts JJG, Hulst HE, et al. The hippocampus in multiple sclerosis. Lancet Neurol. 2018;17:918–26.
Schoonheim MM, Geurts J, Wiebenga OT, De Munck JC, Polman CH, Stam CJ, et al. Changes in functional network centrality underlie cognitive dysfunction and physical disability in multiple sclerosis. Mult Scler. 2014;20:1058–65.
Tona F, Petsas N, Sbardella E, Prosperini L, Carmellini M, Pozzilli C, et al. Multiple sclerosis: altered thalamic resting-state functional connectivity and its effect on cognitive function. Radiology. 2014;271:814–21.
Hidalgo de la Cruz M, Valsasina P, Mesaros S, Meani A, Ivanovic J, Martinovic V et al. Clinical predictivity of thalamic sub-regional connectivity in clinically isolated syndrome: a 7-year study. Mol Psychiatry 2021;26:2163–74.
d’Ambrosio A, Hidalgo de la Cruz M, Valsasina P, Pagani E, Colombo B, Rodegher M, et al. Structural connectivity-defined thalamic subregions have different functional connectivity abnormalities in multiple sclerosis patients: Implications for clinical correlations. Hum Brain Mapp. 2017;38:6005–18.
Hidalgo de la Cruz M, d’Ambrosio A, Valsasina P, Pagani E, Colombo B, Rodegher M, et al. Abnormal functional connectivity of thalamic sub-regions contributes to fatigue in multiple sclerosis. Mult Scler. 2018;24:1183–95.
d’Ambrosio A, Valsasina P, Gallo A, De Stefano N, Pareto D, Barkhof F, et al. Reduced dynamics of functional connectivity and cognitive impairment in multiple sclerosis. Mult Scler. 2020;26:476–88.
Eijlers AJC, Wink AM, Meijer KA, Douw L, Geurts JJG, Schoonheim MM. Reduced network dynamics on functional MRI signals cognitive impairment in multiple sclerosis. Radiology. 2019;292:449–57.
Bommarito G, Tarun A, Farouj Y, Preti MG, Petracca M, Droby A et al. Functional network dynamics in progressive multiple sclerosis. medRxiv. 2020: https://www.medrxiv.org/content/10.1101/2020.11.26.20238923v1.
Oldfield RC. The assessment and analysis of handedness: the Edinburgh inventory. Neuropsychologia. 1971;9:97–113.
Rao SM, Leo GJ, Bernardin L, Unverzagt F. Cognitive dysfunction in multiple sclerosis. I. Frequency, patterns, and prediction. Neurology. 1991;41:685–91.
Amato MP, Portaccio E, Goretti B, Zipoli V, Ricchiuti L, De Caro MF, et al. The Rao’s Brief Repeatable Battery and Stroop Test: normative values with age, education and gender corrections in an Italian population. Mult Scler. 2006;12:787–93.
Allen EA, Damaraju E, Plis SM, Erhardt EB, Eichele T, Calhoun VD. Tracking whole-brain connectivity dynamics in the resting state. Cereb Cortex. 2014;24:663–76.
Biswal B, Yetkin FZ, Haughton VM, Hyde JS. Functional connectivity in the motor cortex of resting human brain using echo-planar MRI. Magn Reson Med. 1995;34:537–41.
Lowe MJ, Mock BJ, Sorenson JA. Functional connectivity in single and multislice echoplanar imaging using resting-state fluctuations. Neuroimage. 1998;7:119–32.
Schoonheim MM, Hulst HE, Brandt RB, Strik M, Wink AM, Uitdehaag BM, et al. Thalamus structure and function determine severity of cognitive impairment in multiple sclerosis. Neurology. 2015;84:776–83.
Kaiser RH, Whitfield-Gabrieli S, Dillon DG, Goer F, Beltzer M, Minkel J, et al. Dynamic resting-state functional connectivity in major depression. Neuropsychopharmacology. 2016;41:1822–30.
Buckner RL, Andrews-Hanna JR, Schacter DL. The brain’s default network: anatomy, function, and relevance to disease. Ann NY Acad Sci. 2008;1124:1–38.
van Geest Q, Douw L, van ‘t Klooster S, Leurs CE, Genova HM, Wylie GR, et al. Information processing speed in multiple sclerosis: relevance of default mode network dynamics. Neuroimage Clin. 2018;19:507–15.
Valsasina P, Hidalgo de la Cruz M, Filippi M, Rocca MA. Characterizing rapid fluctuations of resting state functional connectivity in demyelinating, neurodegenerative, and psychiatric conditions: from static to time-varying analysis. Front Neurosci. 2019;13:618.
Zhou K, Zhu L, Hou G, Chen X, Chen B, Yang C, et al. The contribution of thalamic nuclei in salience processing. Front Behav Neurosci. 2021;15:634618.
Hwang K, Bertolero MA, Liu WB, D’Esposito M. The human thalamus is an integrative hub for functional brain networks. J Neurosci. 2017;37:5594–607.
Cabral J, Vidaurre D, Marques P, Magalhaes R, Silva Moreira P, Miguel Soares J, et al. Cognitive performance in healthy older adults relates to spontaneous switching between states of functional connectivity during rest. Sci Rep. 2017;7:5135.
Kornblith S, Buschman TJ, Miller EK. Stimulus load and oscillatory activity in higher cortex. Cereb Cortex. 2016;26:3772–84.
Zhou F, Zhuang Y, Gong H, Zhan J, Grossman M, Wang Z. Resting state brain entropy alterations in relapsing remitting multiple sclerosis. PLoS One. 2016;11:e0146080.
Acknowledgements
AC is supported by a MAGNIMS/ECTRIMS research fellowship programme.
Author information
Authors and Affiliations
Contributions
MAR contributed to the conception and design of the study. AC, PV, MHdlC, LC, PP, OM, MF, MAR contributed to the acquisition and analysis of data. AC, PV, MHdlC, LC, PP, OM, MF, MAR contributed to drafting the text and preparing the figures. All the authors gave their approval to the current version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
AC has received research grants from the ECTRIMS-MAGNIMS and from Almirall, and served on advisory boards for: Merk, Novartis, Roche and Almirall. PV received speaker honoraria from Biogen Idec. MHdlC reports no disclosures. LC received speaker and consultant honoraria from ACCMED, Roche, BMS Celgene, and Sanofi. PP received speaker honoraria from Biogen, Novartis, Merck Serono, Bristol Myers Squibb and Genzyme. He is supported by a senior research fellowship FISM – Fondazione Italiana Sclerosi Multipla – cod. 2019/BS/009 and financed or co-financed with the ‘5 per mille’ public funding. OM reports no disclosures. MF is Editor-in-Chief of the Journal of Neurology, Associate Editor of Human Brain Mapping, Associate editor of Radiology, and Associate Editor of Neurological Sciences; received compensation for consulting services and/or speaking activities from Almiral, Alexion, Bayer, Biogen, Celgene, Eli Lilly, Genzyme, Merck-Serono, Novartis, Roche, Sanofi, Takeda, and Teva Pharmaceutical Industries; and receives research support from Biogen Idec, Merck-Serono, Novartis, Roche, Teva Pharmaceutical Industries, Italian Ministry of Health, Fondazione Italiana Sclerosi Multipla, and ARiSLA (Fondazione Italiana di Ricerca per la SLA). MAR received speaker honoraria from Bayer, Biogen, Bristol Myers Squibb, Celgene, Genzyme, Merck Serono, Novartis, Roche, and Teva, and receives research support from the MS Society of Canada and Fondazione Italiana Sclerosi Multipla.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Carotenuto, A., Valsasina, P., Hidalgo de la Cruz, M. et al. Divergent time-varying connectivity of thalamic sub-regions characterizes clinical phenotypes and cognitive status in multiple sclerosis. Mol Psychiatry 27, 1765–1773 (2022). https://doi.org/10.1038/s41380-021-01401-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41380-021-01401-w
This article is cited by
-
Potential application of hydrogel to the diagnosis and treatment of multiple sclerosis
Journal of Biological Engineering (2022)