Abstract
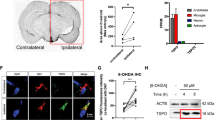
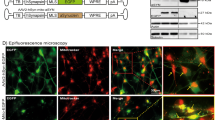
Respiratory chain complex I deficiency elicits mitochondrial dysfunction and reactive oxidative species (ROS), which plays a crucial role in Parkinson’s disease (PD) pathogenesis. However, it remains unclear whether the impairment in other complexes in the mitochondrial oxidative phosphorylation chain is also sufficient to trigger PD onset. Here we show that inhibition of Complex II or III in the electron transport chain (ETC) induces the motor disorder and PD pathologies in neuronal Thy1-C/EBPβ transgenic mice. Through a cell-based screening of mitochondrial respiratory chain inhibitors, we identified TTFA (complex II inhibitor) and Atovaquone (complex III inhibitor), which robustly block the oxidative phosphorylation functions, strongly escalate ROS, and activate C/EBPβ/AEP pathway that triggers dopaminergic neuronal cell death. Oral administration of these inhibitors to Thy1-C/EBPβ mice elicits constipation and motor defects, associated with Lewy body-like inclusions. Deletion of SDHD (Succinate dehydrogenase) gene from the complex II in the Substantia Nigra of Thy1-C/EBPβ mice triggers ROS and PD pathologies, resulting in motor disorders. Hence, our findings demonstrate that mitochondrial ETC inactivation triggers PD pathogenesis via activating C/EBPβ/AEP pathway.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
All data and materials used are available in the main text or as supplementary materials. The authors will also provide data and materials upon request. Source data are provided with this paper.
References
Spillantini MG, Schmidt ML, Lee VM, Trojanowski JQ, Jakes R, Goedert M. Alpha-synuclein in Lewy bodies. Nature. 1997;388:839–40.
Faustini G, Bono F, Valerio A, Pizzi M, Spano P, Bellucci A. Mitochondria and alpha-synuclein: friends or foes in the pathogenesis of parkinson’s disease? Genes (Basel) 2017;8:377–85.
Gao F, Yang J, Wang D, Li C, Fu Y, Wang H, et al. Mitophagy in Parkinson’s disease: pathogenic and therapeutic implications. Front Neurol. 2017;8:527.
Pozo Devoto VM, Falzone TL. Mitochondrial dynamics in Parkinson’s disease: a role for alpha-synuclein? Dis Model Mech. 2017;10:1075–87.
Grunewald A, Kumar KR, Sue CM. New insights into the complex role of mitochondria in Parkinson’s disease. Prog Neurobiol. 2019;177:73–93.
Keeney PM, Xie J, Capaldi RA, Bennett JP Jr. Parkinson’s disease brain mitochondrial complex I has oxidatively damaged subunits and is functionally impaired and misassembled. J Neurosci. 2006;26:5256–64.
Mizuno Y, Ohta S, Tanaka M, Takamiya S, Suzuki K, Sato T, et al. Deficiencies in complex I subunits of the respiratory chain in Parkinson’s disease. Biochem Biophys Res Commun. 1989;163:1450–5.
Blesa J, Przedborski S. Parkinson’s disease: animal models and dopaminergic cell vulnerability. Front Neuroanat. 2014;8:155.
Storch A, Ludolph AC, Schwarz J. Dopamine transporter: involvement in selective dopaminergic neurotoxicity and degeneration. J Neural Transm (Vienna). 2004;111:1267–86.
Desai VG, Feuers RJ, Hart RW, Ali SF. MPP(+)-induced neurotoxicity in mouse is age-dependent: evidenced by the selective inhibition of complexes of electron transport. Brain Res. 1996;715:1–8.
Nicklas WJ, Vyas I, Heikkila RE. Inhibition of NADH-linked oxidation in brain mitochondria by 1-methyl-4-phenyl-pyridine, a metabolite of the neurotoxin, 1-methyl-4-phenyl-1,2,5,6-tetrahydropyridine. Life Sci. 1985;36:2503–8.
Jackson-Lewis V, Blesa J, Przedborski S. Animal models of Parkinson’s disease. Parkinsonism Relat Disord. 2012;18:S183–185.
Sterneck E, Johnson PF. CCAAT/enhancer binding protein beta is a neuronal transcriptional regulator activated by nerve growth factor receptor signaling. J Neurochem. 1998;70:2424–33.
Nadeau S, Hein P, Fernandes KJ, Peterson AC, Miller FD. A transcriptional role for C/EBP beta in the neuronal response to axonal injury. Mol Cell Neurosci. 2005;29:525–35.
Alberini CM, Ghirardi M, Metz R, Kandel ER. C/EBP is an immediate-early gene required for the consolidation of long-term facilitation in Aplysia. Cell. 1994;76:1099–114.
Taubenfeld SM, Milekic MH, Monti B, Alberini CM. The consolidation of new but not reactivated memory requires hippocampal C/EBPbeta. Nat Neurosci. 2001;4:813–8.
Poli V. The role of C/EBP isoforms in the control of inflammatory and native immunity functions. J Biol Chem. 1998;273:29279–82.
Wu Z, Xia Y, Wang Z, Su Kang S, Lei K, Liu X, et al. C/EBPbeta/delta-secretase signaling mediates Parkinson’s disease pathogenesis via regulating transcription and proteolytic cleavage of alpha-synuclein and MAOB. Mol Psychiatry. 2021;26:568–85.
Zhang Z, Kang SS, Liu X, Ahn EH, Zhang Z, He L, et al. Asparagine endopeptidase cleaves alpha-synuclein and mediates pathologic activities in Parkinson’s disease. Nat Struct Mol Biol. 2017;24:632–42.
Kang SS, Ahn EH, Zhang Z, Liu X, Manfredsson FP, Sandoval IM, et al. alpha-Synuclein stimulation of monoamine oxidase-B and legumain protease mediates the pathology of Parkinson’s disease. EMBO J. 2018;37:1–19.
Wang ZH, Gong K, Liu X, Zhang Z, Sun X, Wei ZZ, et al. C/EBPbeta regulates delta-secretase expression and mediates pathogenesis in mouse models of Alzheimer’s disease. Nat Commun. 2018;9:1784.
Lei K, Gu X, Alvarado AG, Du Y, Luo S, Ahn EH, et al. Discovery of a dual inhibitor of NQO1 and GSTP1 for treating glioblastoma. J Hematol Oncol. 2020;13:1–21.
Edgington LE, Verdoes M, Ortega A, Withana NP, Lee J, Syed S, et al. Functional imaging of legumain in cancer using a new quenched activity-based probe. J Am Chem Soc. 2013;135:174–82.
Cortes-Canteli M, Pignatelli M, Santos A, Perez-Castillo A. CCAAT/enhancer-binding protein beta plays a regulatory role in differentiation and apoptosis of neuroblastoma cells. J Biol Chem. 2002;277:5460–7.
Cortes-Canteli M, Aguilar-Morante D, Sanz-Sancristobal M, Megias D, Santos A, Perez-Castillo A. Role of C/EBPbeta transcription factor in adult hippocampal neurogenesis. PLoS One. 2011;6:e24842.
Goto K, Kato G, Kawahara I, Luo Y, Obata K, Misawa H, et al. In vivo imaging of enteric neurogenesis in the deep tissue of mouse small intestine. PLoS One. 2013;8:e54814.
Araujo FG, Lin T, Remington JS. The activity of atovaquone (566C80) in murine toxoplasmosis is markedly augmented when used in combination with pyrimethamine or sulfadiazine. J Infect Dis. 1993;167:494–7.
Xiao WH, Bennett GJ. Effects of mitochondrial poisons on the neuropathic pain produced by the chemotherapeutic agents, paclitaxel and oxaliplatin. Pain. 2012;153:704–9.
Rosen ED. The transcriptional basis of adipocyte development. Prostaglandins Leukot Ess Fat Acids. 2005;73:31–34.
Millward CA, Heaney JD, Sinasac DS, Chu EC, Bederman IR, Gilge DA, et al. Mice with a deletion in the gene for CCAAT/enhancer-binding protein beta are protected against diet-induced obesity. Diabetes. 2007;56:161–7.
Ahn EH, Kang SS, Liu X, Chen G, Zhang Z, Chandrasekharan B, et al. Initiation of Parkinson’s disease from gut to brain by delta-secretase. Cell Res. 2020;30:70–87.
Musgrove RE, Helwig M, Bae EJ, Aboutalebi H, Lee SJ, Ulusoy A, et al. Oxidative stress in vagal neurons promotes parkinsonian pathology and intercellular alpha-synuclein transfer. J Clin Invest. 2019;129:3738–53.
Lei K, Xia Y, Wang X-C, Ahn EH, Jin L, Ye K. C/EBPβ mediates NQO1 and GSTP1 anti-oxidative reductases expression in glioblastoma, promoting brain tumor proliferation. Redox Biol. 2020;34:101578.
Lei K, Kang SS, Ahn EH, Chen C, Liao J, Liu X, et al. C/EBPβ/AEP signaling regulates the oxidative stress in malignant cancers, stimulating the metastasis. Mol Cancer Ther. 2021;9:1–13.
Sun F, Huo X, Zhai Y, Wang A, Xu J, Su D, et al. Crystal structure of mitochondrial respiratory membrane protein complex II. Cell. 2005;121:1043–57.
Moreno-Sanchez R, Hernandez-Esquivel L, Rivero-Segura NA, Marin-Hernandez A, Neuzil J, Ralph SJ, et al. Reactive oxygen species are generated by the respiratory complex II-evidence for lack of contribution of the reverse electron flow in complex I. FEBS J. 2013;280:927–38.
Gutteridge WE. 566C80, an antimalarial hydroxynaphthoquinone with broad spectrum: experimental activity against opportunistic parasitic infections of AIDS patients. J Protozool. 1991;38:141S–143S.
Sun IL, Sun EE, Crane FL, Morre DJ, Lindgren A, Low H. Requirement for coenzyme Q in plasma membrane electron transport. Proc Natl Acad Sci USA. 1992;89:11126–30.
Srivastava IK, Rottenberg H, Vaidya AB. Atovaquone, a broad spectrum antiparasitic drug, collapses mitochondrial membrane potential in a malarial parasite. J Biol Chem. 1997;272:3961–6.
Takabe H, Warnken ZN, Zhang Y, Davis DA, Smyth HDC, Kuhn JG, et al. A repurposed drug for brain cancer: enhanced atovaquone amorphous solid dispersion by combining a spontaneously emulsifying component with a polymer carrier. Pharmaceutics. 2018;10:1–20.
Symersky J, Osowski D, Walters DE, Mueller DM. Oligomycin frames a common drug-binding site in the ATP synthase. Proc Natl Acad Sci USA. 2012;109:13961–5.
Han SY, Jeong YJ, Choi Y, Hwang SK, Bae YS, Chang YC. Mitochondrial dysfunction induces the invasive phenotype, and cell migration and invasion, through the induction of AKT and AMPK pathways in lung cancer cells. Int J Mol Med. 2018;42:1644–52.
Tettamanti G, Malagoli D, Marchesini E, Congiu T, de Eguileor M, Ottaviani E. Oligomycin A induces autophagy in the IPLB-LdFB insect cell line. Cell Tissue Res. 2006;326:179–86.
Vaamonde-Garcia C, Loureiro J, Valcarcel-Ares MN, Riveiro-Naveira RR, Ramil-Gomez O, Hermida-Carballo L, et al. The mitochondrial inhibitor oligomycin induces an inflammatory response in the rat knee joint. BMC Musculoskelet Disord. 2017;18:254.
Acknowledgements
This work was supported by grants from NIH grant (RF1, AG051538) to K.Y. We thank ADRC at Emory University for human PD patients and healthy control samples. This study was supported in part by the Rodent Behavioral Core (RBC), which is subsidized by the Emory University School of Medicine and is one of the Emory Integrated Core Facilities. Additional support was provided by the Viral Vector Core of the Emory Neuroscience NINDS Core Facilities (P30NS055077). Further support was provided by the Georgia Clinical & Translational Science Alliance of the National Institutes of Health under Award Number UL1TR002378, Hallym University Research Fund, 2021(HRF-202103-009), National Key R&D Program of China (2018YFC1314700), National Natural Science Foundation of China (81974196, 81873779) and Program of Shanghai Academic Research Leader (20XD1403400). Dr. Kecheng Lei was supported by grants from China Postdoctoral Science Foundation (2018M632168).
Author contributors
KY conceived the project, designed the experiments, analyzed the data and wrote the manuscript. EHA and KL designed and performed most of the experiments. SSK, XL prepared SH-SY5Y cells and assessed animal experiments. ZHW assisted with data analysis. WH and YTW provided important reagents and scientific discussion. LEM and LJ critically read the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Ahn, E.H., Lei, K., Kang, S.S. et al. Mitochondrial dysfunction triggers the pathogenesis of Parkinson’s disease in neuronal C/EBPβ transgenic mice. Mol Psychiatry 26, 7838–7850 (2021). https://doi.org/10.1038/s41380-021-01284-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41380-021-01284-x
This article is cited by
-
Tumor microenvironment in a minipig model of spinal cord glioma
Journal of Translational Medicine (2023)
-
Helicobacter hepaticus augmentation triggers Dopaminergic degeneration and motor disorders in mice with Parkinson’s disease
Molecular Psychiatry (2023)
-
The Protective Effect of Lithium Against Rotenone may be Evolutionarily Conserved: Evidence from Eisenia fetida, a Primitive Animal with a Ganglionic Brain
Neurochemical Research (2023)
-
Mitophagy and reactive oxygen species interplay in Parkinson’s disease
npj Parkinson's Disease (2022)
-
Mitochondrial Sirtuins in Parkinson’s Disease
Neurochemical Research (2022)