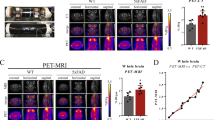
Abstract
18F-FIBT, 2-(p-Methylaminophenyl)-7-(2-[18F]fluoroethoxy)imidazo-[2,1-b]benzothiazole, is a new selective PET tracer under clinical investigation to specifically image β-amyloid depositions (Aβ) in humans in-vivo that binds to Aβ with excellent affinity (Kd 0.7 ± 0.2) and high selectivity over tau and α-synuclein aggregates (Ki > 1000 nM). We aimed to characterize 18F-FIBT in a series of patients with different clinical-pathophysiological phenotypes and to compare its binding characteristics to the reference compound PiB. Six patients (mild late-onset and moderate early-onset AD dementia, mild cognitive impairment due to AD, intermediate likelihood, mild behavioral variant of frontotemporal dementia, subjective memory impairment without evidence of neurodegeneration, and mild dementia due to Posterior Cortical Atrophy) underwent PET imaging with 18F-FIBT on PET/MR. With the guidance of MRI, PET images were corrected for partial volume effect, time-activity curves (TACs) of regions of interest (ROIs) were extracted, and non-displaceable binding potentials (BPnd), standardized uptake value ratios (SUVR), and distribution volume ratio (DVR) were compared. Specific binding was detected in the cases with evidence of the AD pathophysiological process visualized in images of BPnd, DVR and SUVR, consistently with patterns of different tracers in previous studies. SUVR showed the highest correlation with clinical severity. The previous preclinical characterization and the results of this case series suggest the clinical usefulness of FIBT as a selective and highly affine next-generation 18F-labeled tracer for amyloid-imaging with excellent pharmacokinetics in the diagnosis of neurodegenerative diseases. The results compare well to the gold standard PiB and hence support further investigation in larger human samples.
Similar content being viewed by others
Introduction
Amyloid-β (Aβ) peptides are aggregated to amyloid oligomers and plaques and involved in the pathogenesis of Alzheimer’s disease (AD). Aβ plaques represent one of the key hallmarks of AD and therefore, have been depicted as a major target for non-invasive imaging such as PET to aid diagnosis of AD pathophysiology [1, 2]. Aβ-PET has been increasingly used in clinical trials since the first PiB human scan [3] and is more and more used to aid differential diagnosis [4, 5]. Subsequently, several 18F-labeled Aβ tracers were developed which expand the Aβ-PET applications, among those are 18F-florbetapir (Amyvid®), 18F-florbetaben (NeuraCeq®), 18F-flutemetamol (Vizamyl®), all FDA and EMA approved, and 18F-NAV4694.
18F-FIBT (2-(p-Methylaminophenyl)-7-(2-[18F]fluoroethoxy)imidazo[2,1-b]benzothiazole) is another recent PET tracer to measure selectively cerebral amyloid deposition in AD. It has already been characterized in previous studies: its preclinical evaluations [6], a head-to-head comparison to other PET tracers in APP/PS1 mice [7], and the first-in-human PET results [8] indicated excellent imaging properties of FIBT. This includes, for instance, good pharmacokinetics (high initial brain uptake, fast clearance of the brain), excellent binding affinity (Kd 0.7 ± 0.2 towards Aβ in postmortem AD brain) and so far the highest selectivity (>1000 folds selectivity towards Aβ vs. tau and α-synuclein aggregates) as directly compared to the established amyloid tracers. Hence, it is preclinical and characterization results encouraged us to verify the scope of FIBT PET as selective Aβ tracer and usefulness of the tracer in diagnosis of dementing disorders.
In this, we additionally aimed to define the best evaluation method weighting optimal signal-to-noise ratio, feasibility, and scan duration against each other. Dynamic PET acquisition is usually applied to explore proper evaluation methods [9, 10]. The standardized uptake value ratio (SUVR), and distribution volume ratio (DVR) or binding potential (BPnd) are typical indices for evaluation of Aβ PET [11,12,13]. DVR can be estimated using graphical analysis (Logan plot) based on derived arterial input function [11, 14]. However, the extraction of arterial input function requires invasive procedures and is subject to artefacts such as partial volume effects or spill over. In contrast, reference tissue models avoid these difficulties and provide a method to estimate DVR or BPnd [15,16,17,18]. Typical full reference tissue model is derived from two-tissue-compartment model and estimates four parameters during the fitting [18]. DVR estimated using reference tissue model is reported to have highest differentiation and to be superior to SUVR for the differentiation of AD and normal controls with 18F-Florbetaben [19]. However, the full reference tissue model is usually restricted to the calculation of regional TAC curves. For pixel-wise analysis, it suffers from noise due to nonlinear fitting. Simplified reference tissue model (SRTM) assumes that the target and reference tissue can be modeled with one-tissue compartment model and reduces the number of parameters for fitting and is numerically more stable for pixel-wise estimation [15]. It is the most popular reference tissue model [20] and has been recommended for the analysis of 11C-PiB imaging [12]. Reference Logan plot replaces the input curve of Logan graphical analysis with a reference tissue curve [21] and it was demonstrated to be superior in the evaluation of 18F-FDDNP with a priori constant population efflux rate [22]. Multilinear reference tissue model (MRTM) solves the Logan graphical model with multilinear regression and has been reported to be able to differentiate AD and normal controls on 18F-Florbetaben [19]. Although pharmacokinetic modeling has advantages, the reported data have shown that SUVR has better differentiation than DVR for 18F-NAV4694 [23].
The quantification of PET imaging is influenced by partial volume effect [24]. High nonspecific uptake in white matter has been reported in PiB imaging and the severe spillover from white matter hampers the quantification of Aβ deposition [25]. A number of techniques have been developed to correct for partial volume effect [26]. Point-spread functions (PSF) are usually characterized to model the partial volume effect for the recovery of spatial resolution [27]. MR-guided partial volume correction (PVC) methods [28] utilize the anatomical information from high-resolution MR imaging and are widely applied in neurological applications.
We aimed to characterize 18F-FIBT in a series of patients with different clinical-pathophysiological phenotypes to preliminary evaluate the potential of the tracer to image disease severity and for differential diagnosis. In addition, we compared different evaluation methods of tracer kinetics and SUVRs of 18F-FIBT and compered them to a cohort of comparable subjects with 11C-PiB.
Materials and methods
Radiosynthesis
All chemicals, solvents, and materials were purchased and directly used without further purification. 18F-FIBT synthesis followed already published semi-automated procedures [7, 8]. In brief, 18F-FIBT was synthesized and purified by an automated method using HotBoxtwo module (Scintomics, Fuerstenfeldbruck, Germany) by alkylation of 2-(4-(methylamino)phenyl)benzo[d]imidazo[2,1-b]thiazol-7-ol with 18F-fluoroethyl tosylate in dimethylformamide as solvent and sodium hydride as a base. After purification on C18 reversed-phase HPLC-specific activity was always higher than 18 GBq/μmol (0.49 Ci/μmol). 18F-FIBT was directly used with chemical and radioactive purity >98% for all subjects.
Patients
The human subjects were recruited from the outpatient memory clinic at the Centre for Cognitive Disorders of the Department of Psychiatry and Psychotherapy at Technische Universität München (TUM). They had been referred for diagnostic evaluation of cognitive impairment and underwent a standardized diagnostic protocol. Examinations were part of their routine check-up in the course of the evaluation of the patient’s suspected neurodegenerative disorders. Psychometric workup was based on the Consortium to Establish a Registry for AD neuropsychological assessment battery (CERAD-NAB) [29], which includes the Mini-Mental-State Examination (MMSE) [30]. The severity of cognitive impairment was rated on the Clinical Dementia Rating scale (CDR), the sub-scores were used to calculate the CDR sum of boxes (CDR SOB) [31]. All patients provided written informed consent regarding scientific evaluation of their data. The patients′ characteristics are shown in Table 1.
Patient 1 is a 75-year-old man with gradual onset of symptoms 6 years ago and progressive decline since then. He initially presented with memory loss, impaired language, and executive functions. He was diagnosed with AD 5 years ago. From 5 years until 3 months before FIBT imaging he participated in an active immunization trial (randomized to verum during the whole time period) against cerebral amyloid. During this time, only mild progression of symptoms was noted. At the time of 18F-FIBT, he suffered from mild dementia. The initial amyloid scan (4 years before) with 11C-PiB showed substantial tracer uptake, CSF Aβ1–42 and tTau as well as FDG PET (all 4 years before) all have been AD positive.
Patient 2 is a 58-year-old man with gradual onset of symptoms 10 years ago and progressive decline since then. He initially presented with memory loss. He was diagnosed with AD 7 years ago. At the time of 18F-FIBT, he suffered from moderate dementia. Previous AD biomarkers, namely CSF Aβ42 and tTau as well as amyloid PET with PiB and FDG PET (6 years before), were all AD positive.
Patient 3 is a 69-year-old man with gradual onset of symptoms 3 years ago and progressive decline since then. He initially presented with memory loss. At the time of 18F-FIBT, he performed his activities of daily living independently, thus he was diagnosed with mild cognitive impairment. FDG PET (2 months before) was typical for AD.
Participant 4 is a 70-year-old man, who presented with subjective memory loss since 5 years. However, he performed on all neuropsychological tests within normal rage and was not impaired in his activities of daily living. CSF Aβ42 and tTau (5 months before) were both within normal limits. In addition, FDG PET (12 days later) did not reveal any metabolic deficit. Thus, his final diagnosis was subjective memory impairment.
In the 62-year-old male (patient 5), a change of personality was noted 3 years ago: he became disorganized, apathetic, and short-tempered. He was diagnosed with behavioral variant frontotemporal dementia (bvFTD) 2 years before. CSF Aβ42 and tTau (2 years before) were both within normal limits. FDG PET (2 years before) revealed a hypometabolic pattern typical for bvFTD, namely frontobasal, and to a lesser extent temporopolar.
Patient 6, a 71-year-old female, presented with spatial orientation deficits and impairments of visuoconstruction and visual perception. Symptoms gradually started 6 years ago and progressively declined since then. 3 years ago she was diagnosed with Posterior Cortical Atrophy (PCA). At the time of 18F-FIBT, she suffered from mild dementia. The biomarkers indicating amyloid pathology, namely 11C-PiB PET and CSF Aβ42 (3 years before) were clearly positive; however, CSF tTau was (still) within normal limits. FDG PET (3 years before) showed a pattern compatible to PCA, with extensive parieto-occipital hypometabolism, including the visual cortex on both sides, and to a lesser extent temporal and the posterior cingulate cortex (left < right).
PiB patients for comparison
For the comparison of different calculation methods, the patients imaged with dynamic 18F-FIBT were compared to four patients scanned with dynamic 11C-PiB. Demographics of these patients are provided in Supplementary Table 1. For the comparison of SUVRs, each case imaged with 18F-FIBT was compared to a group of matched patients (five patients/group) imaged with 11C-PiB. Clinical characteristics of these matched groups are shown in Supplementary table 2.
FIBT PET/MR
18F-FIBT imaging was performed in accordance with German Drug Act (Arzneimittelgesetz) and German Radiation Safety Act (Strahlenschutzverordnung), § 13 Abs. 2b AMG for individual application applied to specific patients under the professional responsibility of the treating physician. Retrospective analyses and publication of data was approved by the local ethics committee.
Scanning procedure
Human 18F-FIBT PET data were acquired on a fully integrated Siemens Biograph mMR (Siemens Medical Solutions, Knoxville, USA) PET/MR scanning system [32] as described previously [33]. Fully dynamic PET/MR data were in 3D list mode over 90 min beginning with tracer injection. Before, a localizer MRI scan was performed to define correct head positioning. A coronal Dixon T1 MRI sequence for later attenuation correction was run together with PET to ensure optimal temporal and regional correspondence between the two modalities. Simultaneous 18F-FIBT PET/MR was initiated with a bolus injection of 200–300 MBq. Parallel to PET, two major structural MRI sequences were taken (T1-weighted MPRAGE and T2-weighted 3D FLAIR) for anatomical correspondence. PET emission data were corrected for random, dead time, scatter, and attenuation. Resulting sinograms were reconstructed with 3D ordered-subsets expectation maximations (3D OSEM) [34] into two single static frames (40–60 min and 70–90 min) and one dynamic image series with 37 frames (4 × 15 s, 8 × 30 s, 9 × 60 s, 2 × 180 s, 14 × 300 s).
Image preprocessing
All the preprocessing of the data was performed using PMOD software (Version 3.2, PMOD Technologies Ltd. Zurich, Switzerland). All dynamic 18F-FIBT images were corrected for motion by rigid co-registration of each frame with the baseline MRI images. The co-registration between MRI and PET images was inspected before the analysis. Segmentation of gray matter (GM), white matter (WM) and cerebrospinal fluid (CSF) was applied on MPRage images. The Mueller-Gartner method [34] was employed for partial volume correction based on the segmentations applied on the PET data. The co-registered MPRage images were then spatially normalized to the Automated Anatomical Labeling (AAL) atlas [35]. Then, the derived transformation matrices were applied to the dynamic 18F-FIBT images for spatial normalization as well as segmentations of GM, WM, and CSF. Normalized dynamic PET images were parcellated into anatomical regions (frontal, temporal, parietal and occipital lobe as well as cerebellum) as volume-of interests (VOIs). Mean time-activity curves (TACs) of each anatomical region were calculated excluding WM. For reasons of simplicity all VOIs of the same right and left anatomical regions were united to yield simple global values.
The statistical analysis was performed with Matlab Version 8.5 R2015a (Mathworks Inc., Sherborn, MA, US).
FIBT image analyses
For the assessment of the potential of pharmacokinetic modeling, parametric images are preferred for assessment. Thus, we only consider those methods that are robust for parametric image estimation. Three different calculation methods for binding potentials (BPnd), including SRTM, reference Logan and MRTM2 using cerebellum as reference tissue with and without PVC of the 6 subjects scanned with dynamic 18F-FIBT and of the 4 subjects scanned with dynamic 11C-PiB PET were calculated and compared. The relative tracer transport rate R1 using SRTM using the cerebellum as reference tissue was calculated to characterize the tracer transport from blood to brain and was compared to dynamic 11C-PiB scans of four patients with dementia.
Thereafter, BPnd using reference Logan model were compared to DVR and SUVR of the six subjects scanned with dynamic 18F-FIBT and of the four subjects scanned with dynamic 11C-PiB PET.
SUVR images were calculated using subcortical white matter in the centrum semiovale as reference tissue [36,37,38,39,40]. Distribution volume (DV) was calculated using Logan plot. ROIs of internal carotid was placed (5 mm diameter) to generate image-derived arterial input function using a recovery coefficient of 0.7 to correct the partial volume effect of the image-derived input function [41]. Cerebellum was employed as reference tissue to calculate DVR.
To estimate the best time window for image acquisition correlations between SUVR and DVR at different time windows and between different image windows and clinical severity were compared.
Evaluation of FIBT analysis
The signal-to-noise ratios (SNR) were calculated as the ratio of mean value to standard deviation on a selected physiologically homogenous area [42], in this case, the subcortical white matter in the centrum semiovale, to characterize the image quality for visual inspection.
Despite the well-known interval between the development of Aβ pathology and the onset of clinical severity Aβ PET tracer uptake may be associated with the AD severity to a certain degree [43] and an appropriate analysis method should lead to better severity differentiation. We employed this to evaluate the performance of the investigated FIBT analysis methods in the differentiation of AD pathology. The estimated parameters were correlated with CDR SOB using Spearman’s rank correlation.
Results
Time-activity curves
The mean time–activity curves (TACs) of each anatomical region of a patient with moderate AD dementia and a normal control are shown in Fig. 1.
Examples of time-activity curves (TACs) of the investigated cortical regions for FIBT and PiB with and without partial volume correction (PVC): (a) FIBT imaging of a patient with moderate probable AD dementia; (b) FIBT imaging of a patient with subjective memory impairment without evidence of neurodegeneration (“normal control”); (c) PiB imaging of a patient with moderate probable AD dementia for comparison
Comparison of tracer transport rates of FIBT and PiB
The comparison of the relative tracer transport rate R1 using SRTM on data with and without partial volume correction (PVC) of the 6 subjects scanned with dynamic18F-FIBT PET to the 4 subjects with dynamic 11C-PIB PET resulted in comparable transport rates. A decrease of R1 was observed on FIBT scans for parietal and temporal regions as CDR-SOB score increases. Example slices are provided in Supplementary Figure 1. The average values of five investigated anatomical regions were plotted in Supplementary Figure 2.
Comparison of different calculation methods of binding potentials (BPnd) of FIBT and PiB
Example slices of the BPnd (SRTM, rLogan and MRTM2) on the data of the 6 subjects with FIBT and 4 subjects with PiB without and with partial volume correction (PVC); are provided in Supplementary Figure 3. Comparisons between the different calculation methods of BPnd (SRTM, rLogan and MRTM2), DVR and SUVR with and without PVC are provided in Supplementary Figure 4 for dynamic 18F-FIBT PET and in Supplementary Figure 5 for dynamic 11C-PiB PET, respectively.
On the basis of the results, BPnd using reference Logan model has the best SNR among the three reference tissue models for FIBT. In addition, SNRs for FIBT were higher than for PiB (compare Fig. 4 for direct comparison of SNRs).
Comparison of different analysis methods of FIBT and PIB
Sample images of the different analysis methods (BPnd-rLogan, DVR, SUVR) with PVC are shown for the 6 subjects with FIBT and are compared to 4 subjects with PiB in Fig. 2. Moreover, the whole series of these images with and without PVC are provided as Video 1.
An example slice of different analysis methods on the data with partial volume correction (PVC) of the 6 subjects for FIBT and 4 subjects for PiB for comparison. The first row shows the corresponding MRI images. The second row is binding potentials (BPnd) estimated using reference Logan model with cerebellum as reference tissue. The third row is DVR estimated using image-derived arterial input function with cerebellum as reference tissue
The results of different analysis methods (BPnd-rLogan; DVR, SUVRs) on the data of the FIBT imaging and PiB imaging with PVC are compared in Fig. 3.
A comparison of the results of different analysis methods on the data of the FIBT imaging and PiB imaging with partial volume correction (PVC): (a) binding potential (BPnd) of 6 FIBT subjects using reference Logan model; (b) binding potential (BPnd) of 4 PiB subjects using reference Logan model; (c) distribution volume ratio (DVR) of 6 FIBT subjects using Logan plot and the image-derived arterial input function (recovery coefficient of 0.7 to correct the partial volume effect); (d) distribution volume ratio (DVR) of 4 PiB subjects using Logan plot and the image-derived arterial input function (recovery coefficient of 0.7 to correct the partial volume effect); (e) SUVR values of 6 subjects of FIBT imaging; (f) SUVR values of 6 groups of matched subjects (5 patients/group) to (e) with PiB imaging. The cerebellum was chosen as a reference region for the calculation of BPnd and DVR. The subcortical white matter was chosen as reference tissue for SUVR
DVR using image-derived input function and SUVR had considerably higher SNRs than reference tissue model. SNRs were substantially higher in FIBT than in PiB imaging (compare Fig. 4 for direct comparison of SNRs), which can be explained due to higher noise in C-11 imaging.
Estimating the optimal imaging window for FIBT
Correlations between FIBT SUVR and DVR at different time points with and without PVC correction are shown in Supplementary Figure 6. The correlation with DVR was highest for the time window of 40–60 min. However, the window of 70–90 min showed the best washout of nonspecific uptake as depicted by the shape of TACs in our previous study [8]. Hence, the SUVRs for both time-windows, 40–60 min and 70–90 min, were compared in Supplementary Figure 7 retrieving higher SUVRs for the 40–60 min time window. The comparison between the associations of the SUVRs of both time windows with clinical severity (Supplementary Figure 8) also resulted in closer correlations of the 40–60 min time window.
FIBT case series
In the following FIBT uptake in the cohort is described based on visual reading. Corresponding global SUV ratios are provided in Table 2, corresponding regional uptake of the different analyses methods in Fig. 3, respectively.
Patient 1: In this, patient with a mild stage of dementia after participation in an active anti-amyloid immunization study only mildly elevated amyloid deposition was detected as reflected in mildly elevated FIBT uptake.
Patient 2: Strong specific uptake consistent with extensive amyloid deposition that would be expected at a moderate stage of dementia due to underlying AD pathology was detected.
Patient 3: In this, patient with MCI due to underlying AD mild amyloid deposition was expected. Consistently, mild elevated specific cerebral FIBT uptake was detected.
Patient 4: In this, participant without evidence of any neurodegeneration no specific cerebral uptake of FIBT was detected.
Patient 5: In this, patient with bvFTD no amyloid deposition is expected. Consistently with non-AD pathology no specific cerebral tracer uptake was observed.
Patient 6: Strong specific uptake consistent with amyloid deposition was detected that would be compatible with underlying AD pathology in PCA.
Three patients (no.1, 2 and 6) also underwent amyloid PET using 11C-PiB four, six, or 3 years, respectively, before 18F-FIBT (Table 1). Despite the long interscan interval both image modalities are shown in parallel for visual comparison in Supplementary Figure 9.
Associations between FIBT uptake and clinical severity
Plots of the CDR SOB against the results of the different analysis methods of the six subjects imaged with FIBT and of the 26 subjects imaged with PIB for comparison are provided in Fig. 5. Coefficients of the correlations between FIBT results of the five subjects within the continuum of AD, i.e. subjective cognitive impairment, MCI due to AD, mild and moderate AD dementia, and PCA due to AD, and of the PIB SUVR results of 26 subjects for comparison are provided in Table 3. SUVR 50–60 min analysis showed highest correlation with the CDR SOB. Plausibility analyses excluding single cases are provided in Supplementary Figure 8.
Discussion
First FIBT administration in a series of patients with different stages of dementia and different underlying pathologies as measured by biomarkers resulted in specific tracer uptake compatible with expectations. In progressed AD stages strongly elevated tracer uptake was detected, in milder AD stages only mildly elevated FIBT binding was detected, and in participants with non-AD pathologies or without evidence of neurodegeneration no specific tracer uptake was detected.
The preliminary evaluation of different methodologies for data analysis suggests that SUVRs is an easy and robust analyzing technique giving to imaging biomarkers correlated with the severity of the disease, even if it is needed a validation on a larger sample, suggesting FIBT a favorable amyloid marker for clinical routine. A higher retention of FIBT was demonstrated in hippocampus, amygdala and entorhinal cortex of patients 1–3 with evidence of the AD pathophysiological process compared with patient 4. FIBT demonstrated strong neocortical binding in patients with evidence of the AD pathophysiological process which was greater in precuneus/posterior cingulate and frontal cortex than in lateral temporal and parietal cortex, with relative sparing of occipital, sensorimotor and medial temporal cortex. The regional binding results of FIBT showed good similarity to distribution patterns of PiB and 18F-labeled amyloid-tracers such as flutemetamol [44] and florbetaben [42]. Three of the patients underwent amyloid imaging with PiB several years before FIBT PET. Comparability is certainly limited due to the long between scan interval and the low number of subjects. In the patient with mild dementia due to AD (patient no. 1) at FIBT uptake pattern was highly similar between both tracers. Despite the scan interval of 4 years there is no marked progression visible neither on MRI nor on amyloid PET. It is tempting to speculate that this might be due to the anti-amyloid active immunization the patient received for 3 years. The patient with the moderate dementia due to AD (patient no. 2) clearly progressed on MRI during the 6 years between PiB and FIBT imaging. Accordingly, higher uptake on FIBT could be due to the increased amyloid load or higher binding affinity of the tracer. All the regions that were marginally positive on the PiB scan are clearly positive on the later FIBT scan. The patient with the PCA due to AD (patient no. 6) showed a clearly positive amyloid scan with PiB. She progressed on MRI during the 3 years until FIBT. FIBT PET showed a highly comparable pattern as compared to PiB, and with regards to intensity a ceiling effect on the color scale.
The preliminary correlation with FIBT SUVRs showed very high correlation with the severity of dementia measured with the CDR SOB in the five patients of the AD spectrum again favoring SUVRs as analyzing technique. The high correlation coefficients should be interpreted with caution due to the small sample size.
Similar to our recent report, where the representative TACs, with VOI for frontal cortex, cerebellar cortex, and semioval center for AD and healthy subjects in analogy to a report on Florbetaben [45] were demonstrated [8], the 18F-FIBT uptake into frontal and cerebellar cortex peaked around 3 min p.i. for all subjects and initial uptake into cerebellum was always higher. For the patients with AD pathology, uptake in frontal cortex clearly went above cerebellar uptake around 10 min p.i. while this separation not only happened nearly 20 min later in the control subject but the frontal cortex uptake stayed close to cerebellar uptake.
We compared different calculation methods. SRTM, MRTM2, rLogan are theoretically the same and the binding potential and DVR theoretically has the relation BPnd = DVR-1. However, in practice, it is influenced by the following: (1) the bias of model assumptions considering the real situation; (2) different numerical stability of the models to noise in the data; (3) the bias of the derived arterial input function; 4) the bias of the selected reference tissue. More difference between BPnd and DVR has been observed for PiB than for FIBT, which can be explained due to higher noise in C-11 imaging. BPnd estimated from the three models are not identical. The difference is due to different numerical performance under the existence of noise. The difference between the different models of BPnd is less apparent compared to their difference to the DVR. As we do not have accurate arterial blood measurement for this study we applied recovery coefficient based on image-derived AIF. There might be still biases of the applied arterial input function. On the other side, cerebellum may still have some specific binding of the tracers. The existing biases of arterial input function and reference tissues may amplify the difference than theoretical expectations of BPnd = DVR-1.
SNR is an index for imaging quality; it may be useful for visual inspection to differentiate small structures. DVR has better SNR, it may favor the visual inspection. However, the acquisition is longer. It maybe also influenced by the errors of arterial input function. On the other side, there is not so much difference for the correlation with severity. Hence, we would prefer SUVR based on the comparisons between DVR and SUVR, between different time windows of SUVRs and between SUVRs and clinical severity, the time-window 40–60 min applying PVC showed highest SUVRs and correlated best with clinical severity.
18F-FIBT is a new compound as a promising Aβ-PET tracer with similar pharmacokinetics to PiB, superior binding affinity (Kd = 0.7 ± 0.2) and higher selectivity over tau and α-synuclein aggregates (Ki > 1000 nM). 18F-FIBT can be easily synthesized with high radiochemical yield and purity. Recently, 18F-FIBT was primarily compared with florbetaben and PiB in a group of APP/PS1 and control mice using same animals and quantification method. 18F-FIBT has shown an improved imaging and binding characteristics and suitable pharmacokinetics on its own as well as in comparison to PiB and florbetaben. Thus, we used it in a series of six patients with different clinical-pathophysiological phenotypes where we observed FIBT-PET correlated with disease severity. This finding and the good performance of FIBT encourage us to further investigate FIBT-PET in a larger group of patients and explore the qualities of FIBT-PET for dementia imaging preferably in relation to established ligands.
Limitations
While 18F-FIBT appears to be a very promising F18 labeled tracer for human amyloid beta imaging the small number of subjects and the lack of a healthy control group hampers generalizability of the current findings. Larger cohorts including different neurodegenerative diseases at stages need to be imaged to allow final assessment of tracer performance.
The absence of plasma input data could be considered a limitation for the assessment of the pharmacokinetics of FIBT. However, the measurement of plasma input function is an error sensitive procedure with limited time resolution. Hence, reference tissue model that implicitly utilizes the mutual plasma input to estimate pharmacokinetics [6, 7] and that is less sensitive to artifacts was used as an alternative. The so derived pharmacokinetic parameters are equivalent to those derived from plasma input function in our perspective. Furthermore, image-derived input function was used to calculate the DVR [9].
Conclusion
The preliminary results shows that 18F-FIBT with static imaging on PET/MR 40–60 min p.i. provides a clinical favorable routine, is not associated with any adverse events so far, and may aid PET for β-amyloid imaging in neurodegenerative diseases. A further strength of FIBT may be the better correlation with severity of AD pathology, which may favor its use for early diagnosis and therapy monitoring. These encouraging preliminary results support our plans to evaluate FIBT in larger samples.
Change history
08 February 2021
A Correction to this paper has been published: https://doi.org/10.1038/s41380-018-0302-3
References
Hardy J, Selkoe DJ. The amyloid hypothesis of Alzheimer’s disease: progress and problems on the road to therapeutics. Science. 2002;297:353–6.
Hardy JA, Higgins GA. Alzheimer’s disease: the amyloid cascade hypothesis. Science. 1992;256:184–5.
Klunk WE, Engler H, Nordberg A, Wang Y, Blomqvist G, Holt DP, et al. Imaging brain amyloid in Alzheimer’s disease with Pittsburgh Compound-B. Ann Neurol. 2004;55:306–19.
Jansen WJ, Ossenkoppele R, Knol DL, Tijms BM, Scheltens P, Verhey FR, et al. Prevalence of cerebral amyloid pathology in persons without dementia: a meta-analysis. JAMA. 2015;313:1924–38.
Ossenkoppele R, Jansen WJ, Rabinovici GD, Knol DL, van der Flier WM, van Berckel BN, et al. Prevalence of amyloid PET positivity in dementia syndromes: a meta-analysis. JAMA. 2015;313:1939–49.
Yousefi BH, Drzezga A, von Reutern B, Manook A, Schwaiger M, Wester HJ, et al. A Novel 18F-labeled imidazo[2,1-b]benzothiazole (IBT) for high-contrast PET imaging of β-amyloid plaques. ACS Med Chem Lett. 2011;2:673–7.
Yousefi BH, von Reutern B, Scherubl D, Manook A, Schwaiger M, Grimmer T, et al. FIBT versus florbetaben and PiB: a preclinical comparison study with amyloid-PET in transgenic mice. EJNMMI Res. 2015;5:20.
Yousefi BH, Manook A, Grimmer T, Arzberger T, von Reutern B, Henriksen G, et al. Characterization and first human investigation of FIBT, a novel fluorinated Abeta plaque neuroimaging PET radioligand. ACS Chem Neurosci. 2015;6:428–37.
Huang SC. Role of kinetic modeling in biomedical imaging. J Med Sci. 2008;28:57–63.
Gunn RN, Gunn SR, Cunningham VJ. Positron emission tomography compartmental models. J Cereb Blood Flow Metab. 2001;21:635–52.
Price JC, Klunk WE, Lopresti BJ, Lu X, Hoge JA, Ziolko SK, et al. Kinetic modeling of amyloid binding in humans using PET imaging and Pittsburgh Compound-B. J Cereb Blood Flow Metab. 2005;25:1528–47.
Zhou Y, Resnick SM, Ye W, Fan H, Holt DP, Klunk WE, et al. Using a reference tissue model with spatial constraint to quantify [11C]Pittsburgh compound B PET for early diagnosis of Alzheimer’s disease. Neuroimage. 2007;36:298–312.
Ito H, Shimada H, Shinotoh H, Takano H, Sasaki T, Nogami T, et al. Quantitative analysis of amyloid deposition in Alzheimer disease Using PET and the radiotracer (1)(1)C-AZD2184. J Nucl Med. 2014;55:932–8.
Logan J, Fowler JS, Volkow ND, Wolf AP, Dewey SL, Schlyer DJ, et al. Graphical analysis of reversible radioligand binding from time-activity measurements applied to [N-11C-methyl]-(-)-cocaine PET studies in human subjects. J Cereb Blood Flow Metab. 1990;10:740–7.
Lammertsma AA, Hume SP. Simplified reference tissue model for PET receptor studies. Neuroimage. 1996;4(3 Pt 1):153–8.
Gunn RN, Lammertsma AA, Hume SP, Cunningham VJ. Parametric imaging of ligand-receptor binding in PET using a simplified reference region model. Neuroimage. 1997;6:279–87.
Ikoma Y, Watabe H, Hayashi T, Miyake Y, Teramoto N, Minato K, et al. Quantitative evaluation of changes in binding potential with a simplified reference tissue model and multiple injections of [11C]raclopride. Neuroimage. 2009;47:1639–48.
Lammertsma AA, Bench CJ, Hume SP, Osman S, Gunn K, Brooks DJ, et al. Comparison of methods for analysis of clinical [11C]raclopride studies. J Cereb Blood Flow Metab. 1996;16:42–52.
Becker GA, Ichise M, Barthel H, Luthardt J, Patt M, Seese A, et al. PET quantification of 18F-florbetaben binding to beta-amyloid deposits in human brains. J Nucl Med. 2013;54:723–31.
Salinas CA, Searle GE, Gunn RN. The simplified reference tissue model: model assumption violations and their impact on binding potential. J Cereb Blood Flow Metab. 2015;35:304–11.
Logan J, Fowler JS, Volkow ND, Wang GJ, Ding YS, Alexoff DL. Distribution volume ratios without blood sampling from graphical analysis of PET data. J Cereb Blood Flow Metab. 1996;16:834–40.
Wong KP, Wardak M, Shao W, Dahlbom M, Kepe V, Liu J, et al. Quantitative analysis of [18F]FDDNP PET using subcortical white matter as reference region. Eur J Nucl Med Mol Imaging. 2010;37:575–88.
Cselenyi Z, Jonhagen ME, Forsberg A, Halldin C, Julin P, Schou M, et al. Clinical validation of 18F-AZD4694, an amyloid-beta-specific PET radioligand. J Nucl Med. 2012;53:415–24.
Bowen SL, Byars LG, Michel CJ, Chonde DB, Catana C. Influence of the partial volume correction method on (18)F-fluorodeoxyglucose brain kinetic modelling from dynamic PET images reconstructed with resolution model based OSEM. Phys Med Biol. 2013;58:7081–106.
Matsubara K, Ibaraki M, Shimada H, Ikoma Y, Suhara T, Kinoshita T, et al. Impact of spillover from white matter by partial volume effect on quantification of amyloid deposition with [11C]PiB PET. Neuroimage. 2016;143:316–24.
Erlandsson K, Buvat I, Pretorius PH, Thomas BA, Hutton BF. A review of partial volume correction techniques for emission tomography and their applications in neurology, cardiology and oncology. Phys Med Biol. 2012;57:R119–59.
Aklan B, Oehmigen M, Beiderwellen K, Ruhlmann M, Paulus DH, Jakoby BW, et al. Impact of point-spread function modeling on PET image quality in integrated PET/MR hybrid imaging. J Nucl Med. 2016;57:78–84.
Hofmann M, Pichler B, Scholkopf B, Beyer T. Towards quantitative PET/MRI: a review of MR-based attenuation correction techniques. Eur J Nucl Med Mol Imaging. 2009;36(Suppl 1):S93–104.
Morris JC, Heyman A, Mohs RC, Hughes JP, van Belle G, Fillenbaum G, et al. The Consortium to establish a registry for Alzheimer’s disease (CERAD). part i. clinical and neuropsychological assessment of Alzheimer’s disease. Neurology. 1989;39:1159–65.
Folstein MF, Folstein SE, McHugh PR. “Mini-mental state”. a practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res. 1975;12:189–98.
Morris JC. The Clinical Dementia Rating (CDR): current version and scoring rules. Neurology. 1993;43:2412–4.
Delso G, Furst S, Jakoby B, Ladebeck R, Ganter C, Nekolla SG, et al. Performance measurements of the Siemens mMR integrated whole-body PET/MR scanner. J Nucl Med. 2011;52:1914–22.
Drzezga A, Souvatzoglou M, Eiber M, Beer AJ, Furst S, Martinez-Moller A, et al. First clinical experience with integrated whole-body PET/MR: comparison to PET/CT in patients with oncologic diagnoses. J Nucl Med. 2012;53:845–55.
Muller-Gartner HW, Links JM, Prince JL, Bryan RN, McVeigh E, Leal JP, et al. Measurement of radiotracer concentration in brain gray matter using positron emission tomography: MRI-based correction for partial volume effects. J Cereb Blood Flow Metab. 1992;12:571–83.
Tzourio-Mazoyer N, Landeau B, Papathanassiou D, Crivello F, Etard O, Delcroix N, et al. Automated anatomical labeling of activations in SPM using a macroscopic anatomical parcellation of the MNI MRI single-subject brain. Neuroimage. 2002;15:273–89.
Su Y, Blazey TM, Owen CJ, Christensen JJ, Friedrichsen K, Joseph-Mathurin N, et al. Quantitative amyloid imaging in autosomal dominant Alzheimer’s disease: results from the DIAN Study Group. PLoS ONE. 2016;11:e0152082.
Brendel M, Hogenauer M, Delker A, Sauerbeck J, Bartenstein P, Seibyl J, et al. Improved longitudinal [(18)F]-AV45 amyloid PET by white matter reference and VOI-based partial volume effect correction. Neuroimage. 2015;108:450–9.
Chen K, Roontiva A, Thiyyagura P, Lee W, Liu X, Ayutyanont N, et al. Improved power for characterizing longitudinal amyloid-beta PET changes and evaluating amyloid-modifying treatments with a cerebral white matter reference region. J Nucl Med. 2015;56:560–6.
Landau SM, Fero A, Baker SL, Koeppe R, Mintun M, Chen K, et al. Measurement of longitudinal beta-amyloid change with 18F-florbetapir PET and standardized uptake value ratios. J Nucl Med. 2015;56:567–74.
Joshi AD, Pontecorvo MJ, Lu M, Skovronsky DM, Mintun MA, Devous MD, et al. for Quantification of F 18 Florbetapir PET Images. J Nucl Med. 2015;56:1736–41.
Wardak M, Schiepers C, Dahlbom M, Cloughesy T, Chen W, Satyamurthy N, et al. Discriminant analysis of (1)F-fluorothymidine kinetic parameters to predict survival in patients with recurrent high-grade glioma. Clin Cancer Res. 2011;17:6553–62.
Catafau AM, Bullich S, Seibyl JP, Barthel H, Ghetti B, Leverenz J, et al. Cerebellar amyloid-beta plaques: How frequent are they, and do they influence 18F-Florbetaben SUVR? J Nucl Med. 2016;57:1740–5.
Grimmer T, Henriksen G, Wester HJ, Forstl H, Klunk WE, Mathis CA, et al. Clinical severity of Alzheimer’s disease is associated with PIB uptake in PET. Neurobiol Aging. 2009;30:1902–9.
Mountz JM, Laymon CM, Cohen AD, Zhang Z, Price JC, Boudhar S, et al. Comparison of qualitative and quantitative imaging characteristics of [11C]PiB and [18F]flutemetamol in normal control and Alzheimer’s subjects. NeuroImage Clin. 2015;9:592–8.
Barthel H, Luthardt J, Becker G, Patt M, Hammerstein E, Hartwig K, et al. Individualized quantification of brain beta-amyloid burden: results of a proof of mechanism phase 0 florbetaben PET trial in patients with Alzheimer’s disease and healthy controls. Eur J Nucl Med Mol Imaging. 2011;38:1702–14.
Acknowledgements
We thank M. Herz, M. Beschorner, C. Meisinger, S. Swirczek, S. Schachoff, B. Dzewas, and C. Schaubitzer for their excellent support. This work was partially supported by grants DFG HE 4560/1–3 and FP7 MindView.
Author Contributions:
Study concept and design: TG, SF, MS, H-JW and BHY; data acquisition and analysis: TG, KS, IY, BHY; drafting the manuscript and figures: TG, KS, BHY; discussion and proofreading: all authors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
TG reported having received consulting fees from Actelion, Biogen, Lilly, MSD; Novartis, Quintiles, Roche Pharma; lecture fees from B. Braun, Biogen, Lilly, Parexel, Roche Pharma, and grants to his institution from Actelion and PreDemTech. The other authors declare that they have no conflict of interest.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Grimmer, T., Shi, K., Diehl-Schmid, J. et al. 18F-FIBT may expand PET for β-amyloid imaging in neurodegenerative diseases. Mol Psychiatry 25, 2608–2619 (2020). https://doi.org/10.1038/s41380-018-0203-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41380-018-0203-5
This article is cited by
-
Current radiotracers to image neurodegenerative diseases
EJNMMI Radiopharmacy and Chemistry (2019)