Abstract
BCORL1 is a transcriptional corepressor homologous to BCOR. We describe 12 BCORL1-altered uterine sarcomas with striking resemblance to BCOR-altered endometrial stromal sarcoma (BCOR-ESS), including 5 with BCORL1 rearrangements (JAZF1-BCORL1, EP300-BCORL1, or internal BCORL1 rearrangement), 5 with inactivating BCORL1 mutations (T513fs*22, P600fs*1, R945*, R1196*, or R1265fs*4) and 2 with homozygous BCORL1 deletion. The median patient age was 57.5 years (range 33–79). An association with aggressive clinical behavior was identified. Diagnoses assigned prior to genomic testing varied: 7 tumors were previously diagnosed as ESS, 2 as high-grade uterine sarcomas, 2 as myxoid uterine leiomyosarcomas, and 1 as a uterine spindle cell neoplasm consistent with leiomyosarcoma. Tumors harbored frequent gelatinous, mucomyxoid-like appearance by gross examination and unique histology with morphological overlap with BCOR-ESS. Key microscopic features included (1) a spindle cell appearance, most often with at least focal myxoid stroma, (2) variable amounts of hypocellular fibromyxoid spindle areas with lower grade atypia and/or (3) variable amounts of epithelioid areas with higher grade atypia. Specifically, spindle and epithelioid components were present in 100 and 75% of sarcomas, respectively; myxoid stroma was identified in 83%, collagen plaques or fibrosis in 50%, and high-grade nuclear atypia was present in 42%. Like BCOR-ESS, 50% of BCORL1-altered sarcomas exhibited CDK4 amplification or CDKN2A loss. In contrast, 33% harbored NF1 alterations, while 25% had other alterations in the NF2-mTOR pathway, expanding potential therapeutic targets. In conclusion, inactivating BCORL1 genomic alterations may define a distinct subset of high-grade endometrial stromal sarcomas with biological overlap with BCOR-ESS, both of which may mimic myxoid leiomyosarcomas.
Similar content being viewed by others
Introduction
Rare cases of endometrial stromal sarcomas with BCORL1 alterations have been reported [1, 2]; however, the clinicopathological features and mutational landscape of BCORL1-altered uterine sarcomas have not been systematically investigated. BCORL1 is a transcriptional corepressor homologous to BCOR; both have related biological functions during transcriptional regulation as they may interchangeably form polycomb repression complex 1 (PRC1) variants [3]. Recently, uterine sarcomas with genomic alterations in BCOR via rearrangements or internal tandem duplication (ITD) have become a newly recognized, distinct subtype of high-grade endometrial stromal sarcomas with aggressive behavior and unique morphology [4,5,6]. Because of its homology and related biological function to BCOR, we hypothesized that molecular alterations of BCORL1 may also similarly define a distinct subset of uterine sarcomas.
Low-grade endometrial stromal sarcomas are generally considered to be indolent tumors with late recurrences, while high-grade endometrial sarcomas exhibit more aggressive behavior with poorer outcomes [7]. Low-grade endometrial stromal sarcomas are often associated with JAFZ1 or MEAF6, EPC1, EPC2 rearrangements partnering with either SUZ12 or PHF1, both of which are subunits of polycomb repressive complex 2 (PRC2), a histone methyltransferase [8]. These rearrangements give rise to fusion products such as JAZF1-SUZ12, JAZF1-PHF1, MEAF6-PHF1, EPC1-PHF1, or EPC2-PHF1 [9,10,11]. Morphologically, low-grade endometrial sarcoma is classically characterized by islands of cells resembling endometrial stroma with oval to fusiform nuclei, scant cytoplasm, low grade nuclear atypia, variable mitotic activity, with infiltration into the myometrium in a tongue-like fashion. In contrast, high-grade endometrial stromal sarcomas exhibit poor prognosis and may harbor YWHAE or BCOR rearrangements, or alternatively, BCOR ITD, as well as high-grade features. YWHAE-NUTM2A/B fusion-positive uterine sarcomas demonstrate round cells with eosinophilic cytoplasm and high-grade nuclear atypia in the form of irregular nuclear contours, vesicular chromatin, and variably distinct nucleoli.
BCOR-altered endometrial stromal sarcomas exhibit unique morphology, which may be dependent on the type of BCOR molecular alteration. ZC3H7B-BCOR fusion is the most common BCOR genomic alteration in uterine sarcomas, and most tumors with ZC3H7B-BCOR fusions feature spindle cells in a fascicular growth pattern with uniform nuclei, mild-to-moderate atypia, and frequent myxoid and collagenous stromal changes [4, 12]. In addition, tumors with rare BCOR rearrangements involving non-ZC3H7B partners or with BCOR ITD demonstrate a mix of spindle, epithelioid or small round cell components and varying degrees of fascicular growth and myxoid or collagenous stroma [6]. Because of prominent myxoid features, a subset of BCOR-altered endometrial stromal sarcoma has previously masqueraded as myxoid uterine leiomyosarcomas [4, 12]. Molecularly, BCOR-rearranged endometrial stromal sarcomas harbor frequent MDM2 amplification and activation of the cyclin D1-CDK4 pathway via CDK4 amplification, CDKN2A deletion or cyclin D1 protein overexpression [6].
Similar to BCOR, disruption of PRC1 via genomic alterations of the homologous BCORL1 has been reported in several cancer types, such as ossifying fibromyxoid tumor with CREBBP-BCORL1 fusion [13], myelodysplastic syndrome (MDS) or acute myeloid leukemia (AML) with inactivating BCORL1 truncating mutations[14], and hepatocellular carcinoma with BCORL1-ELF4 fusion [15]. In addition, JAZF1-BCORL1 fusions have been previously reported in a case of uterine adenosarcoma [16] as well as in rare cases of endometrial stromal sarcoma [1, 17]. However, the morphological spectrum and biological behavior of BCORL1-altered uterine sarcomas are not well defined.
While inactivating truncating BCORL1 short variant mutations, such as nonsense and frameshift mutations, are present in MDS and AML, they have not been previously reported in uterine sarcoma. Due to the identification of inactivating mutations in leukemia, BCORL1 has been postulated to be a tumor suppressor gene, whose loss of function leads to disruption of the PRC1 complex and dysregulation of proper transcription control [18]. Considering the homology and related biological functions of BCOR and BCORL1, we aimed to (1) investigate the mutational landscape of BCORL1 in uterine sarcomas, (2) determine whether molecular alterations of BCORL1 drive the oncogenesis of a distinct subset of endometrial stromal sarcomas, and (3) compare the clinicopathological and molecular features of BCOR- versus BCORL1-altered uterine sarcomas.
Methods
BCORL1-altered uterine sarcoma cohorts
Approval for this study was obtained from the Western Institutional Review Board (Protocol No. 20152817). A retrospective database search of a CLIA-certified and CAP-accredited reference molecular laboratory was performed for uterine sarcomas with genomic profiles containing all classes of pathogenic BCORL1 genomic alterations. The samples were previously sent to Foundation Medicine from medical care facilities across North America for detection of targetable genetic alterations during routine clinical and oncologic care at various institutions. At the time of the study, the Foundation Medicine research database originally contained 1,445 uterine sarcomas (278 endometrial stromal sarcomas, 963 uterine leiomyosarcoma, and 204 uterine sarcomas NOS). The tumors were previously assayed with comprehensive genomic profiling (CGP) via both DNA- and RNA-based targeted next-generation sequencing (NGS). Clinicopathological data including age, sites of metastases, and immunohistochemistry were extracted from pathology reports and available clinical documents. The pathologic diagnosis of uterine sarcoma, endometrial stromal sarcoma and associated morphological features were centrally re-evaluated on routine H&E slides of tissue sections submitted for genomic profiling by Dr. Douglas Lin.
Genomic profiling and biomarker analysis
NGS-based CGP was performed on hybridization-captured, adaptor ligation–based libraries using DNA and RNA extracted from formalin-fixed paraffin-embedded tumor tissue. All samples forwarded for DNA and RNA extraction contained a minimum of 20% tumor cells. The samples were assayed for all coding exons from up to 406 cancer-related genes, plus select introns from up to 31 genes. Patient samples were sequenced and evaluated for all classes of genomic alterations including base substitutions, insertions, deletions, copy number alterations (amplifications and homozygous deletions), and for fusions/rearrangements, as previously described [19,20,21]. Oncoprints of genomic data were generated with the cBio portal [22, 23]. Tumor mutational burden (TMB) was determined on 0.79–1.14 Mb of sequenced DNA using a mutation burden estimation algorithm [24]. In this study, low TMB was defined as <10 mut/Mb, since a score of at least 10 mut/Mb is currently FDA-approved companion diagnostic biomarker for immunotherapy [25]. Assessment of microsatellite instability was performed from DNA next-generation sequencing across 114 loci [24].
Statistical analysis
Differences among categorical variables were assessed using Fisher’s exact test. Statistical tests were 2-sided and used a significance threshold of p < 0.05. Reported p values were not adjusted for multiple testing.
Results
BCORL1-altered endometrial stromal sarcomas
We retrospectively identified 12 uterine sarcomas with BCORL1 genomic alterations from the archives of Foundation Medicine. The 12 BCORL1-mutated uterine sarcomas comprised 1% of all uterine sarcomas in our research database, which originally contained 1,445 uterine sarcomas (278 endometrial stromal sarcomas, 963 uterine leiomyosarcomas and 204 uterine sarcomas NOS) with comprehensive genomic profiling data between 2010 and 2020. The 12 uterine sarcomas harbored different classes of BCORL1 genomic alterations, which included 5 tumors with BCORL1 rearrangements or fusions (JAZF1-BCORL1, EP300-BCORL1, or internal BCORL1 rearrangement), 5 with truncating BCORL1 short variant, nonsense or frameshift mutations (T513fs*22, P600fs*1, R945*, R1196*, or R1265fs*4) and 2 tumors with homozygous BCORL1 gene deletion (Fig. 1 and Table 1). The sarcoma with internal BCORL1 alteration exhibited a chromosome X deletion, resulting in loss of exon 8 and intron 9 of BCORL1. Out of the uterine sarcomas with BCORL1 fusions, three harbored recurrent JAZF1-BCORL1 or reciprocal BCORL1-JAZF1-fusions and one harbored a novel EP300-BCORL1 fusion (Fig. 1 and Table 1).
A Top: schematic representation of BCORL1 and positions and types of mutations in BCORL1 (NM_021946) identified in our uterine sarcomas. Protein domains: CTBP1 binding site, NLS (nuclear localization signal), LXXL (Leu-Xaa-Xaa-Leu-Leu motif), ANK (ankyrin repeats) and PUFD domains. Bottom: Homologous BCOR protein domains for comparison and location of previously described BCOR internal tandem duplications (ITD, denoted by arrow, BBD: BCL6 binding domain). B Diagrams demonstrating BCORL1 fusions identified in our cohort. Reference sequences: JAZF1 (NM_175061), BCORL1 (NM_021946), EP300 (NM_001429), BCOR (NM_017745). Two additional uterine sarcomas also harbored homozygous BCORL1 gene deletion and one additional tumor had an internal BCORL1 rearrangement, which are not represented in this figure.
In our cohort of 12 BCORL1-altered sarcomas, the median patient age was 57.5 years (range 33–79) (Table 1). There were 3 primary uterine resections, 2 initial non-uterine diagnostic biopsies (1 vagina, 1 pelvis) and 7 recurrences. Most tumors were aggressive with extra-uterine spread and/or recurrences after total hysterectomy (Table 1). Available tumor staging is provided on Table 1. Sites of metastasis or recurrences included peri-aortic and obturator lymph nodes, pelvic sidewall, omentum, pancreas, small and large intestine, bladder, rectum and vagina. Of patients with available clinical follow-up, 4 died of disease at 11, 11, 64 and 73 months after total abdominal hysterectomy, respectively (Table 1, case #1, #5, #7 and #12).
Primary tumor size was available for 3 of 12 tumors (3.0, 7.4 and 14.5 cm). Size of recurrences was available for 6 cases and ranged from 4.5 to 25 cm. Gross descriptions were available for 9 tumors (3 primary samples and 6 metastasis) and not available for 3 samples since they were core biopsies or fragmented specimens. Two of three primary tumors exhibited gross necrosis and one of three had gelatinous, mucomyxoid-like areas. Of the 6 metastasis, 4 cases exhibited gross hemorrhage and necrosis, and 2 cases had mucomyxoid-like gelatinous areas. Lymph node metastasis was identified in one sample; however, not all patients underwent a lymph node dissection.
Microscopically, of the 3 primary tumors, 2 had evaluable borders. 1 case exhibited tongue-like infiltration, dissecting into the myometrium (Fig. 2A), while the second tumor had a broad front, pushing border (Fig. 2B). One of three sarcomas extended into the cervical stroma. When it could be evaluated, tongue-like pattern of infiltration into adjacent pelvic soft tissue structures was also identified in one recurrence.
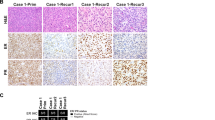
Histologically, most tumors were characterized by spindle cells and variable amounts of epithelioid cells. No small cell morphology was identified in any tumor. (Table 2). A spindle cell component was present in 100% (12 of 12) of tumors and was characterized by either cellular areas some of which were interspersed with collagen plaques, fibrosis or by hypocellular spindle areas with myxoid stroma (Figs. 3, 4 and 5). Spindle cells had scant cytoplasm, predominantly uniform nuclei, even chromatin pattern and inconspicuous nucleoli (Figs. 3, 4 and 5). Occasionally, focal areas with increased atypia and pleomorphism were noted in the spindle and myxoid components (Fig. 3F). In contrast, an epithelioid component was identified in 75% (9 of 12) tumors and featured cells with abundant clear to pale cytoplasm, round nuclei, often with increased nuclear atypia, coarser chromatin and conspicuous nucleoli (Figs. 3, 4 and 5). Rhabdoid morphology was also identified within the epithelioid component in 42% (5 of 12) of sarcomas (Fig. 5E) with the amount of rhabdoid component ranging from 5 to 80% of the tumors. No sex cord stromal elements were identified in any tumor.
A-D: Case #1 with JAZF1-BCORL1 demonstrating (A) alternating hypercellular and hypocellular areas and myxoid stroma on low power view, (B) hypercellular area with spindle cells and mild to moderate atypia, (C) hypocellular and spindle cell area with low-grade atypia and myxoid stromal change, and (D) hypercellular, epithelioid area with high-grade nuclear atypia and prominent nucleoli. E-F: Case #2 with EP300-BCORL1 rearrangement characterized by (E) epithelioid morphology with pink cytoplasm and (F) hypocellular myxoid and spindle cell areas.
A-B-C) Case #7 with epithelioid and spindle areas, spiral arterioles, myxoid stroma and mild to moderate atypia. D-E-F) Case #9 with hypercellular spindle cell area with collagen plaques, hypocellular fibromyxoid areas, and focal epithelioid areas with high-grade atypia adjacent to spindle cell and myxoid areas.
A-C: Case #5 with BCORL1 R1265fs*4 frameshift mutation, previously diagnosed as low grade endometrial stromal sarcoma, characterized by (A) epithelioid morphology with clear to pale cytoplasm and moderate to high grade atypia, (B) spindle to epithelioid areas with collagen plaques, and (C) spindle cell areas with myxoid stroma and lower grade atypia. D. Case #10, previously diagnosed as myxoid leiomyosarcoma, harboring BCORL1 T513fs*22 frameshift mutation as the only oncogenic genomic alteration, exhibiting spindle cell morphology with myxoid stroma and stromal fibrosis. E-F: Case #12, previously diagnosed as spindle cell neoplasm consistent with leiomyosarcoma with BCORL1 P600fs*1 frameshift mutation, featuring epithelioid and rhabdoid areas with high grade atypia, and (E) spindle cell area with lower grade atypia and myxoid change.
Overall, 58% of tumors had uniform nuclei and mild to moderate atypia, while 42% harbored severe nuclear atypia, characterized by nuclear enlargement, condensed chromatin and prominent nucleoli, more often associated with the epithelioid component (Figs. 3, 4 and 5). Myxoid stroma was present in 83% of tumors, while stromal fibrosis or collagen plaques were present in 50%. Tumor necrosis and spiral arterioles were present in 42 and 25% of sarcomas, respectively (Table 2). One sarcoma exhibited perivascular tumor cell whorling; and none demonstrated hemangiopericytoma-like vessels or larger thick-walled vessels. Ossification, characterized by benign bone formation, was present in one tumor. Mitotic count across the twelve uterine sarcomas with BCORL1 alterations varied and ranged from 2 to 25 mitoses per 10 high power fields (mean = 9.5, median = 8.5 mitoses, range 2–25 mitoses per 10 HPF). 42% (5 out of 12) of tumors had mitotic indices of at least 10 mitotic figures/10 HPF (Table 2). One uterine sarcoma (8%, 1 of 12) with a BCORL1 R1196* mutation (case #6) exhibited classic, conventional low-grade endometrial stromal sarcoma morphology with low-grade spindle cells in a tongue-like pattern of infiltration and devoid of myxoid, collagen plaques or epithelioid components. Interestingly, this case also harbored an EPC1-PHF1 fusion, which is characteristic of low-grade endometrial stromal sarcoma.
By stratifying according to the type of BCORL1 genomic alteration (i.e. fusion/rearrangement, short variant alteration or homozygous gene deletion), 100% of uterine sarcomas (7 of 7) harboring BCORL1 fusions or homozygous gene deletion exhibited an epithelioid component with at least moderate nuclear atypia (Fig. 3, Fig. 4 and Table 2). In contrast, tumors featuring BCORL1 short variant alterations (i.e. frameshift or nonsense mutations) exhibited predominant spindle cell morphology with uniform nuclei, and an epithelioid component was present in 40% (2 of 5) (Fig. 5 and Table 2). In addition, 40% (2 of 5) of tumors with BCORL1 short variant alterations also harbored fusions classically associated with low-grade endometrial stromal sarcoma, such as JAFZ1-SUZ12 or EPC1-PHF1, in contrast to 0% of tumors containing BCORL1 fusion or homozygous gene deletion. The case with a JAFZ1-SUZ12 fusion had a co-occurring BCORL1 R1265fs*4 frameshift mutation and demonstrated an associated epithelioid component with higher grade nuclear atypia as well as myxoid and stromal fibrosis components (Figs. 5A, 5B and 5C).
The submitted diagnoses from referring outside institutions included 7 endometrial stromal sarcomas, 2 high-grade uterine sarcomas, 2 myxoid uterine leiomyosarcomas and 1 uterine spindle cell neoplasm consistent with leiomyosarcoma (Table 1). Based on genomics and morphological similarity to BCOR-altered endometrial stromal sarcomas, the 3 cases with prior diagnoses of leiomyosarcoma were reclassified as BCORL1-altered endometrial stromal sarcoma, as previously described for BCOR-altered ESS that mimicked myxoid leiomyosarcomas [4, 6], based on morphology and lack of known driver mutations commonly found in either conventional uterine leiomyosarcoma (i.e., lack of TP53, ATRX, PTEN or MED12 alterations [26,27,28]) or in myxoid uterine leiomyosarcoma (i.e., lack of TP53 and BRCA2 mutations or PLAG1 rearrangements [29,30,31]). For instance, case #10 was previously diagnosed as myxoid uterine leiomyosarcoma; however, the frameshift BCORL1 T513fs*22 short variant mutation was the only genomic alteration identified with no other co-occurring molecular alterations.
Comprehensive genomic profiling revealed that BCORL1-altered uterine sarcomas were microsatellite stable and had a low tumor mutation burden. In addition to BCORL1 alterations, high frequency of homozygous deletion of CDKN2A and CDKN2B was present in 33% of BCORL1-altered uterine sarcomas, while CDK4 and MDM2 gene amplification was present in 17% of cases (Fig. 6). Therefore, overall genomic alterations leading to the activation of the CDK4 kinase pathway, via CDK4 amplification or CDKN2A loss, occurred in 6 of 12 (50%) BCORL1-altered uterine sarcoma cases, at a comparable rate as seen with previously described BCOR-rearranged endometrial stromal sarcomas by our group using the same methodology [6] (50% vs. 65%, p = 0.5).
Other targetable alterations in BCORL1-altered uterine sarcomas included homozygous deletion or inactivating truncating mutations of NF1 (W267* and E291*) in 33% of tumors (Fig. 6). The frequency of NF1 alterations in BCORL1-mutated uterine sarcoma was higher than that seen in BCOR-rearranged endometrial stromal sarcoma (33% vs. 5%, p = 0.02). Another notable difference was a higher frequency of alterations in the NF2-mTOR pathway in 25% of BCORL1-altered uterine sarcoma compared with 2.5% in BCOR-rearranged endometrial stromal sarcoma (p = 0.03) (Fig. 6). Specific alterations in the NF2-mTOR pathway included NF2 K550fs*2 and PIK3R1 D560Y in one case as well as mTOR C1483Y or AKT1 E17K in two additional cases.
BCORL1 alterations in other uterine mesenchymal tumors
In contrast to BCORL1 fusions or rearrangements which were specific to uterine sarcomas in the Foundation Medicine genomic database, two short variant BCORL1 mutations were also identified in one conventional uterine leiomyosarcoma and one uterine inflammatory myofibroblastic tumor. The conventional uterine leiomyosarcoma had a BCORL1 V1096I missense mutation, as well as TP53, RB1, and MED12 co-alterations and classic leiomyosarcoma morphology and without myxoid features. The uterine inflammatory myofibroblastic tumor harbored a BCORL1 R1196* nonsense mutation, a co-occurring RANBP2-ALK1 fusion and myxoid features. In these two cases, the morphology and genomic profiles were compatible with conventional leiomyosarcoma and inflammatory myofibroblastic tumors with co-occurring BCORL1 short variant mutations. Notably, the type of BCORL1 mutation identified in the conventional uterine leiomyosarcoma was a point mutation, BCORL1 V1096I, in contrast to the nonsense and frameshift mutations that were identified in the other BCORL1-mutated uterine sarcomas with myxoid features.
Reported immunohistochemistry
We extracted immunohistochemistry (IHC) results that were performed at outside institutions, if they were available, from the submitting pathology reports. All tested tumors were reportedly negative for cytokeratins, while all tumors (100%, 6 of 6) were positive for CD10. The majority was positive for ER (71%, 5 of 7) and PR (83%, 5 of 6). BCOR IHC was not available for any tumor. One of three tumors (33%) was positive for cyclin D1, and there was variable staining for muscle markers, including for desmin (75%, 3 of 4), smooth muscle actin (75%, 3 of 4). Tumors were also reportedly negative for caldesmon (n = 2), CD117 (n = 2), DOG1 (n = 2), S100 (n = 2) and CD34 (n = 1). However, these results need to be confirmed in a larger series and in a centralized laboratory.
Discussion
BCORL1 is a transcriptional corepressor that is homologous to BCOR. We assessed whether molecular alterations in BCORL1 defined a distinct subset of uterine sarcomas similar to BCOR. To test this hypothesis, we examined the molecular landscape of BCORL1 genomic alterations across various uterine mesenchymal malignancies and characterized the clinicopathological and genomic features of uterine sarcomas that are driven by BCORL1 molecular alterations. The identified cohort of BCORL1-altered uterine sarcomas had several similarities to the findings previously described for BCOR-rearranged high grade endometrial stromal sarcomas [4, 6] including: (1) similar median age of mid-50 years and age range of 20–30 s to 70 s in both types, (2) aggressive behavior with refractory recurrences, (3) striking morphological resemblance with hypocellular fibromyxoid areas, collagen plaques, stromal fibrosis, and high-grade epithelioid components, (4) frequent myxoid stroma, some of which mimicked and were previously diagnosed as myxoid uterine leiomyosarcoma, and (5) frequent co-occurring genomic alterations leading to the activation of the CDK4 kinase pathway, via CDK4 amplification or CDKN2A loss.
Overall, 11 of 12 cases demonstrated striking resemblance to previously described BCOR-altered endometrial stromal sarcomas in the form of myxoid stromal change, collagen plaques or stromal fibrosis and/or epithelioid components [4, 6]. One exception resembled low-grade ESS and harbored a BCORL1 frameshift mutation and an EPC1-PH1 fusion. It remains to be determined whether low-grade tumors with BCORL1 short variant mutations and EPC1-PH1 fusions have a worse prognosis or increased propensity to high grade transformation than typical low-grade ESS. A limitation to our study is lack of long-term follow-up for the entire cohort to assess prognosis and survival. Nevertheless, four patients succumbed to disease within six years following hysterectomy, two of which died <1 year following hysterectomy. Our results suggest that similarly to BCOR, genomic alterations in BCORL1 drive the pathogenesis of a distinct subset of high-grade endometrial stromal sarcomas. The high-grade nature of this new uterine sarcoma subtype is supported by both morphological features and short patient survival following hysterectomy when follow-up was available. The endometrial stromal origin is supported by positivity for CD10, which is traditionally viewed as a marker of endometrial stromal differentiation.
Identification of BCORL1-altered high-grade endometrial stromal sarcomas may be difficult without performing molecular analysis, as highlighted by the fact that 25%, 3 of 12 tumors were previously diagnosed as leiomyosarcomas, all of which exhibited myxoid features on re-review. Key clues that may raise an index of suspicion for a BCORL1-endometrial stromal sarcoma are: 1) frequent gelatinous, mucomyxoid-like appearance by gross examination, 2) microscopic spindle cell appearance, most often with at least focal myxoid stromal change, 3) variable amounts of hypocellular fibromyxoid spindle areas with lower grade atypia and/or 4) variable amounts of epithelioid areas with higher grade nuclear atypia. Notably, epithelioid or rhabdoid components were never identified in isolation and were always associated with a spindle cell component.
Most of the short variant BCORL1 alterations in the uterine sarcomas were nonsense mutations or out-of-frame insertions that are predicted to encode truncated proteins lacking the last C-terminal LXXLL nuclear receptor recruitment motif and ANK and PUFD domains, which are involved in protein-protein interactions and transcriptional regulation [32] (Fig. 1). In addition, we identified 2 uterine sarcomas with homozygous BCORL1 deep gene deletion. These results suggest that a subset of uterine sarcomas is driven by BCORL1 loss of function within the PRC1 transcriptional complex, leading to transcriptional de-regulation. Supporting this notion, a BCORL1-mutated tumor in our study also exhibited co-occurring BCOR alteration (Fig. 6), likely further contributing to PRC1 transcriptional dysfunction. In addition, none of the BCORL1 short variant alterations were subclonal (defined as BCORL1 next-generation sequencing variant allele frequency less than 10% of tumor purity) and therefore were consistent with driver alterations. While loss of heterozygosity of second the BCORL1 could not be determined, it is worth noting that BCORL1 is located on the X chromosome, and the second BCORL1 allele may be subject to complete chromosome X inactivation in females [33].
While JAFZ1-BCORL1 or EP300-BCORL1 fusions and homozygous BCORL1 deletion appeared specific to uterine sarcomas, a BCORL1 point mutation and a BCORL1 nonsense mutation were also identified in one conventional uterine leiomyosarcoma and one myxoid inflammatory myofibroblastic tumor. However, the morphology and genomic profiles of these 2 tumors were different than BCORL1-altered uterine sarcomas and compatible with the submitting diagnoses. In addition, short variant BCORL1 frameshift or nonsense mutations were also identified in endometrial carcinomas and carcinosarcomas, especially in a microsatellite instability setting (not shown). Defects in the mismatch repair system often results in frameshift mutations, and therefore most of the BCORL1 short variant mutations in endometrial carcinoma or carcinosarcoma were likely secondary mutations due to microsatellite instability rather than primary driver mutations as seen in microsatellite stable uterine sarcomas.
In this study, we report novel BCORL1 rearrangements in uterine sarcomas such as EP300-BCORL1 and as well as a case with internal BCORL1 gene rearrangement without another gene partner. Interestingly, similar EP300-BCOR and internal BCOR rearrangements have also been previously identified in BCOR-rearranged high-grade endometrial stromal sarcomas, which also demonstrated similar spindle, epithelioid and myxoid morphology [6].
Like BCOR-rearranged endometrial stromal sarcomas, BCORL1-altered uterine sarcomas exhibited high frequencies of CDK4 amplification and CDKN2A homozygous gene deletion. Genomic alterations leading to the activation of the CDK4 kinase, via CDK4 amplification or CDKN2A loss, occurred in 50% BCORL1-altered uterine sarcomas. Future investigation would be of interest to determine whether refractory tumors with CDK4 and CDKN2A alterations are responsive to CDK4 inhibitors, such as palbociclib, ribociclib, and abemaciclib, in patients that fail conventional therapy and in which current standard treatments may no longer be clinically effective. CDK4 inhibitors are currently FDA-approved for the treatment of ER-positive and HER2-negative breast carcinomas with minimal side effects [34, 35].
In contrast to BCOR-rearranged uterine sarcomas in which only a small minority exhibited NF1 (5%) or NF2 (2.5%) alterations [6], BCORL1-altered uterine sarcomas harbored inactivating NF1 mutations or alterations of the mTOR-NF2-AKT pathway in 33 and 25% of cases, respectively. For these reasons, based on response in other tumor types [36, 37], off-label use of MEK inhibitors or mTOR inhibitors as single agents or in combination with other therapies could also be considered in a subset of BCORL1-mutated uterine sarcomas that are refractory to conventional cytotoxic chemotherapy.
Our study demonstrates the value of next-generation sequencing-based comprehensive genomic profiling for identifying BCORL1-mutated uterine sarcomas since the BCORL1 nonsense and frameshift mutations seen in our study would not have been detected by either FISH or sarcoma fusion panels, similarly to the previously described BCOR internal tandem duplications within exon 15 of BCOR in high-grade endometrial stromal sarcomas. Therefore, during the pathological work-up of an uterine sarcoma case, a negative result for either FISH or sarcoma fusion panel for BCOR or BCORL1 may warrant reflex testing to a next-generation sequencing-based assay for a more comprehensive evaluation of either BCOR or BCORL1.
In conclusion, we evaluated the clinicopathological and molecular features of uterine sarcomas driven by BCORL1 genomic alterations. BCORL1-altered uterine sarcomas had striking morphological resemblance to previously described BCOR-rearranged high-grade endometrial stromal sarcomas. Given homologous gene structures, related biological functions of BCORL1 and BCOR and similar clinicopathological features, BCORL1-altered uterine sarcomas may represent a new subtype of high-grade endometrial stromal sarcomas. This new group of aggressive BCORL1-mutated uterine sarcomas may mimic myxoid leiomyosarcomas and should also be considered in the differential diagnosis of myxoid uterine mesenchymal neoplasms.
Data availability
All data generated or analyzed during this study are included in this published article.
References
Allen AJ, Ali SM, Gowen K, Elvin JA, Pejovic T. A recurrent endometrial stromal sarcoma harbors the novel fusion JAZF1-BCORL1. Gynecol Oncol Rep. 2017;20:51–53.
Brahmi M, Franceschi T, Treilleux I, Pissaloux D, Ray-Coquard I, Dufresne A, et al. Molecular classification of endometrial stromal sarcomas using RNA sequencing defines nosological and prognostic subgroups with different natural history. Cancers (Basel). 2020;12:1–12.
Wong SJ, Senkovich O, Artigas JA, Gearhart MD, Ilangovan U, Graham DW, et al. Structure and Role of BCOR PUFD in Noncanonical PRC1 Assembly and Disease. Biochemistry. 2020;59:2718–28.
Lewis N, Soslow RA, Delair DF, Park KJ, Murali R, Hollmann TJ, et al. ZC3H7B-BCOR high-grade endometrial stromal sarcomas: a report of 17 cases of a newly defined entity. Mod Pathol. 2018;31:674–84.
Juckett LT, Lin DI, Madison R, Ross JS, Schrock AB, Ali SM. A pan-cancer landscape analysis reveals a subset of endometrial stromal and pediatric tumors defined by internal tandem duplications of BCOR. Oncology. 2018;96:101–9.
Lin DI, Hemmerich A, Edgerly C, Duncan D, Severson EA, Huang RSP, et al. Genomic profiling of BCOR-rearranged uterine sarcomas reveals novel gene fusion partners, frequent CDK4 amplification and CDKN2A loss. Gynecol Oncol. 2020;157:357–66.
Benson C, Miah AB. Uterine sarcoma - current perspectives. Int J Women’s Health. 2017;9:597–606.
Margueron R, Reinberg D. The Polycomb complex PRC2 and its mark in life. Nature. 2011;469:343–9.
Koontz JI, Soreng AL, Nucci M, Kuo FC, Pauwels P, van Den Berghe H, et al. Frequent fusion of the JAZF1 and JJAZ1 genes in endometrial stromal tumors. Proc Natl Acad Sci USA. 2001;98:6348–53.
Micci F, Gorunova L, Gatius S, Matias-Guiu X, Davidson B, Heim S, et al. MEAF6/PHF1 is a recurrent gene fusion in endometrial stromal sarcoma. Cancer Lett. 2014;347:75–78.
Brunetti M, Gorunova L, Davidson B, Heim S, Panagopoulos I, Micci F. Identification of an EPC2-PHF1 fusion transcript in low-grade endometrial stromal sarcoma. Oncotarget. 2018;9:19203–8.
Hoang LN, Aneja A, Conlon N, Delair DF, Middha S, Benayed R, et al. Novel High-grade Endometrial Stromal Sarcoma. Am J Surg Pathol. 2017;41:12–24.
Kao Y-C, Sung Y-S, Zhang L, Chen CL, Huang SC, Antonescu CR. Expanding the molecular signature of ossifying fibromyxoid tumors with two novel gene fusions: CREBBP-BCORL1 and KDM2A-WWTR1. Genes, Chromosom Cancer. 2017;56:42–50.
Li M, Collins R, Jiao Y, Ouillette P, Bixby D, Erba H, et al. Somatic mutations in the transcriptional corepressor gene BCORL1 in adult acute myelogenous leukemia. Blood. 2011;118:5914–7.
Totoki Y, Tatsuno K, Yamamoto S, Arai Y, Hosoda F, Ishikawa S, et al. High-resolution characterization of a hepatocellular carcinoma genome. Nat Genet. 2011;43:464–71.
Muthukumarana V, Fix DJ, Stolnicu S, Stolnicu S, Park KJ, Soslow RA, et al. BCOR Expression in Mullerian Adenosarcoma: A Potential Diagnostic Pitfall. Am J Surg Pathol. 2020;44:765–70.
Mohammad N, Stewart CJR, Chiang S, Turashvili G, Dickson BC, Ng TL, et al. P53 Immunohistochemical Analysis of Fusion‐Positive Uterine Sarcomas. Histopathology. 2020;78:805–13.
Yamamoto Y, Abe A, Emi N. Clarifying the impact of polycomb complex component disruption in human cancers. Mol Cancer Res. 2014;12:479–84.
Lipson D, Capelletti M, Yelensky R, Otto G, Parker A, Jorosz M, et al. Identification of new ALK and RET gene fusions from colorectal and lung cancer biopsies. Nat Med. 2012;18:382–4.
He J, Abdel-Wahab O, Nahas MK, Wang K, Rampal RK, Intlekofer AM, et al. Integrated genomic DNA/RNA profiling of hematologic malignancies in the clinical setting. Blood. 2016;127:3004–14.
Frampton GM, Fichtenholtz A, Otto GA, Wang K, Downing SR, He J, et al. Development and validation of a clinical cancer genomic profiling test based on massively parallel DNA sequencing. Nat Biotechnol. 2013;31:1023–31.
Gao J, Aksoy BA, Dogrusoz U, Dresdner G, Gross B, Sumer SO, et al. Integrative analysis of complex cancer genomics and clinical profiles using the cBioPortal. Sci Signal. 2013;6:pl1.
Cerami E, Gao J, Dogrusoz U, Gross BE, Sumer SO, Aksoy BA, et al. The cBio cancer genomics portal: an open platform for exploring multidimensional cancer genomics data. Cancer Disco. 2012;2:401–4.
Chalmers ZR, Connelly CF, Fabrizio D, Gay L, Ali SM, Ennis R, et al. Analysis of 100,000 human cancer genomes reveals the landscape of tumor mutational burden. Genome Med. 2017;9:34.
Marabelle A, Fakih M, Lopez J, Shah M, Shapira-Frommer R, Nakagawa K, et al. Association of tumour mutational burden with outcomes in patients with advanced solid tumours treated with pembrolizumab: prospective biomarker analysis of the multicohort, open-label, phase 2 KEYNOTE-158 study. Lancet Oncol. 2020;10:1353–1365.
Mäkinen N, Aavikko M, Heikkinen T, Taipale M, Taipale J, Koivisto-Korander R, et al. Exome sequencing of uterine leiomyosarcomas identifies frequent mutations in TP53, ATRX, and MED12. PLOS Genet. 2016;12:e1005850.
Cuppens T, Moisse M, Depreeuw J, Annibali D, Colas E, Gil-Moreno A, et al. Integrated genome analysis of uterine leiomyosarcoma to identify novel driver genes and targetable pathways. Int J Cancer. 2018;142:1230–43.
Elvin JA, Gay LM, Ort R, Shuluk J, Long J, Shelley L, et al. Clinical benefit in response to palbociclib treatment in refractory uterine leiomyosarcomas with a common CDKN2A alteration. Oncologist. 2017;22:416–21.
Schaefer IM, Hornick JL, Sholl LM, Quade BJ, Nucci MR, Parra-Herran C. Abnormal p53 and p16 staining patterns distinguish uterine leiomyosarcoma from inflammatory myofibroblastic tumour. Histopathology. 2017;70:1138–46.
Arias-Stella JA, Benayed R, Oliva E, Young RH, Hoang LN, Lee CH, et al. Novel PLAG1 gene rearrangement distinguishes a subset of uterine myxoid leiomyosarcoma from other uterine myxoid mesenchymal tumors. Am J Surg Pathol. 2019;43:382–8.
Yoon J-Y, Mariño-Enriquez A, Stickle N, de Borja RJ, Ismiil N, Djordjevic B, et al. Myxoid smooth muscle neoplasia of the uterus: comprehensive analysis by next-generation sequencing and nucleic acid hybridization. Mod Pathol. 2019;32:1688–97.
Plevin MJ, Mills MM, Ikura M. The LxxLL motif: a multifunctional binding sequence in transcriptional regulation. Trends Biochem Sci. 2005;30:66–69.
Carrel L, Willard HF. X-inactivation profile reveals extensive variability in X-linked gene expression in females. Nature. 2005;434:400–4.
Turner NC, Ro J, André F, Loi S, Verma S, Iwata H, et al. Palbociclib in hormone-receptor–positive advanced breast cancer. N Engl J Med. 2015;373:209–19.
Turner NC, Slamon DJ, Ro J, Bondarenko I, Im SA, Masuda N, et al. Overall survival with palbociclib and fulvestrant in advanced breast cancer. N Engl J Med. 2018;379:1926–36.
See WL, Tan I-L, Mukherjee J, Nicolaides T, Pieper RO. Sensitivity of glioblastomas to clinically available MEK inhibitors is defined by neurofibromin 1 deficiency. Cancer Res. 2012;72:3350–9.
Ali SM, Miller VA, Ross JS, Pal SK. Exceptional response on addition of everolimus to taxane in urothelial carcinoma bearing an NF2 mutation. Eur Urol. 2015;67:1195–6.
Acknowledgements
We thank Foundation Medicine’s lab assistants, histology and molecular technologists for assistance with tumor processing and sample preparation for genomic sequencing.
Funding statement
No external funding sources were available for this study.
Author information
Authors and Affiliations
Contributions
DIL performed study concept, design, analysis, data acquisition, and manuscript writing. ND performed analysis and acquired part of the data. All authors interpreted data and provided the study materials; and all authors read, revised, and approved the paper.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Approval for this study was obtained from the Western Institutional Review Board Protocol No. 20152817.
Competing interests
All authors are full-time employees of Foundation Medicine, Inc., which is a whole subsidiary of Roche, and these employees have equity interest in Roche.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lin, D.I., Huang, R.S.P., Mata, D.A. et al. Clinicopathological and genomic characterization of BCORL1-driven high-grade endometrial stromal sarcomas. Mod Pathol 34, 2200–2210 (2021). https://doi.org/10.1038/s41379-021-00873-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41379-021-00873-z
This article is cited by
-
Differential diagnosis of uterine adenosarcoma: identification of JAZF1-BCORL1 rearrangement by comprehensive cancer genomic profiling
Diagnostic Pathology (2023)
-
Diffusely infiltrating glioma with CREBBP–BCORL1 fusion showing overexpression of not only BCORL1 but BCOR: A case report
Brain Tumor Pathology (2022)