Abstract
Five sporadic Creutzfeldt–Jakob disease (CJD) strains have been identified to date, based on differences in clinicopathological features of the patients, the biochemical properties of abnormal prion proteins, and transmission properties. Recent advances in our knowledge about iatrogenic transmission of sporadic CJD have raised the possibility that the infectivity of sporadic CJD strains through peripheral routes is different from that of intracranial infection. To test this possibility, here we assessed systematically the infectivity of sporadic CJD strains through the peripheral route for the first time using a mouse model expressing human prion protein. Although the infectivity of the V2 and M1 sporadic CJD strains is almost the same in intracerebral transmission studies, the V2 strain infected more efficiently than the M1 strain through the peripheral route. The other sporadic CJD strains examined lacked infectivity. Of note, both the V2 and M1 strains showed preference for mice with the valine homozygosity at the PRNP polymorphic codon. These results indicate that the V2 strain is the most infectious sporadic CJD strain for infection through peripheral routes. In addition, these findings raise the possibility that individuals with the valine homozygosity at the PRNP polymorphic codon might have higher risks of infection through peripheral routes compared with the methionine homozygotes. Thus, preventive measures against the transmission of the V2 sporadic CJD strain will be important for the eradication of iatrogenic CJD transmission through peripheral routes.
Similar content being viewed by others
Introduction
Creutzfeldt–Jakob disease (CJD) is a lethal transmissible neurodegenerative disease caused by an abnormal isoform of prion protein (PrPSc) converted from the normal cellular isoform (PrPC) [1]. The conformational conversion of PrPC can occur due to either one of three causes: spontaneous conversion in sporadic CJD (sCJD), pathogenic mutations of the PRNP gene in genetic CJD, or infection with PrPSc in iatrogenic CJD (iCJD) and variant CJD [2]. In iCJD, transmission of PrPSc from sCJD patients occurs through medical procedures such as dura mater grafting, intramuscular/subcutaneous administration of human growth hormone derived from cadaveric pituitary, corneal transplantation, neurosurgical operation, and electroencephalogram using deep brain electrodes [3].
Recent advances in our knowledge about transmission of sCJD have suggested that the infectivity of sCJD strains through peripheral routes differs from that in intracranial infection [4]. sCJD is currently classified into six subtypes based on the genotype (methionine, M, or valine, V) at polymorphic codon 129 of the PRNP gene, the type (type 1, 21 kDa, or type 2, 19 kDa) of PrPSc accumulating in the brain, and neuropathologic features [5, 6]. The six sCJD subtypes have been proposed to harbor five sCJD strains: M1 (MM1/MV1 subtype corresponding to 40% of total sCJD patients), V2 (VV2 and MV2 subtypes, 23%), M2C (MM2C subtype, ≤1%), M2T (MM2T subtype, ≤1%), and V1 (VV1 subtype, ≤1%) [5,6,7,8]. In addition, co-occurrence of the M1 and M2C strains in the same patient has also been seen (MM1/MV1 + 2C subtype corresponding to 28% of total sCJD cases) [8]. Among the five sCJD strains, the M1 strain is the causative agent of the majority of dura mater graft-associated iCJD, reflecting the existing ratio of the sCJD subtypes in donors of the dura mater grafts [9, 10]. By contrast, in human growth hormone-associated iCJD, the M1 strain subgroup was underrepresented, and the V2 strain subgroup was overrepresented [11, 12]. In addition, the V2 strain subgroup was also predominant in kuru, which is caused by the transmission of sCJD through ritual cannibalism [13, 14]. The reason for the different proportions of the causative sCJD strains among acquired prion diseases has remained elusive. Recently, we established a quick bioassay to evaluate the infectivity of PrPSc by intraperitoneal inoculation and found that a M1 sCJD isolate did not infect efficiently through the peripheral route [15]. This finding has raised the possibility that the predominance of the V2 strain subgroup in human growth hormone-associated iCJD and kuru might have been due to the weak infectivity of the M1 strain through peripheral routes. To test this possibility, here we performed a systematic analysis of the infectivity of sCJD strains through the peripheral route using multiple sCJD isolates.
Materials and methods
Transmission experiments
The sCJD cases included in this study were from patients with clinically, genetically, and histopathologically proven sCJD, and brain tissues were obtained at autopsy from the patients after receiving written informed consent for research use. According to the current classification system of sCJD [5], these sCJD cases were classified as follows: VV2, 3 cases; MM1, 3 cases; MM2C, 3 cases; MM2T, 3 cases. Although two distinct MM2C subtypes harboring different prion strains have been recognized in Japan [16], the present study included the MM2C subtype showing large confluent vacuoles and perivacuolar PrPSc deposition. The production of knock-in mice expressing human PrPC with the 129 M/M genotype or 129 V/V genotype (Ki-Hu129M/M or Ki-Hu129V/V) has been reported previously [17]. Brain homogenates (10%) were prepared in sterile phosphate-buffered saline using glass homogenizers, and 50 μl of the homogenates were intraperitoneally inoculated into Ki-Hu129M/M or Ki-Hu129V/V mice as described previously [18]. The inoculated mice were sacrificed at 75 days post-inoculation, and the spleen was immediately frozen for western blotting. This bioassay system has been developed to evaluate rapidly the infectivity of PrPSc through the peripheral route [15, 17, 18].
Western blotting
Spleen tissues were homogenized in 2 ml of lysis buffer (100 mM Tris-HCl pH 8.0, 10 mM NaCl, 10 mM MgCl2, 2% Triton X-100, and 25 units/ml DNase I) and digested with collagenase (1 mg/200 mg tissue) overnight at room temperature. Collagenase digestion disrupts the connective tissue and improves the accessibility of detergents and/or proteinase K (PK) to PrPSc. The digested homogenates were ultracentrifuged at 323,000 × g for 30 min at 4 °C, and the pellets were resuspended and sonicated in 870 μl of PK-digestion buffer (100 mM Tris-HCl pH 8.0 and 5% Sarkosyl). The resuspended samples were centrifuged at 10,000 × g for 3 min to remove the cell debris, and the supernatants (800 μl) were digested with PK (4 μg/200 mg tissue) for 1 h at 37 °C. The PK-digested proteins were precipitated by adding 200 μl of 99.5% ethanol and ultracentrifugation at 100,000 × g for 60 min at 4 °C. The pellets were resuspended in Laemmli’s sample buffer (60 mM Tris-HCl pH 6.8, 5% glycerol, 2% SDS, and 0.01% bromophenol blue) (400 μl/200 mg tissue). Protein samples were subjected to SDS-PAGE using 13.5% Tris-glycine long gels of 15 cm length and western blotting. Anti-PrP monoclonal antibody 3F4 (BioLegend, SanDiego, CA, USA) and type 2 PrPSc-specific polyclonal antibody Tohoku 2 [19] were used as the primary antibodies. Goat anti-mouse immunoglobulin polyclonal antibody labeled with a peroxidase-conjugated dextran polymer, EnVsion+ (Agilent, Santa Clara, CA, USA) and anti-rabbit EnVision+ (Agilent) were used as the secondary antibodies. The blots were visualized with Clarity Max Western ECL Substrate (Bio-Rad, Hercules, CA, USA), and images were obtained by imaging device ImageQuant LAS 4000 mini (GE Healthcare, Chicago, IL, USA).
Results
The V2 sCJD strain was efficiently transmitted to both Ki-Hu129M/M and Ki-Hu129V/V mice through the peripheral route. Total 58% of the intraperitoneally inoculated mice (39% in Ki-Hu129M/M and 74% in Ki-Hu129V/V) showed PrPSc accumulation in the spleen at 75 days post-inoculation (Table 1). The V2 strain-inoculated Ki-Hu129M/M mice produced the intermediate type PrPSc (20 kDa), while the V2 strain-inoculated Ki-Hu129V/V mice produced type 2 PrPSc (19 kDa) (Fig. 1), as reported previously [15, 20]. In contrast, the M1 sCJD strain was not efficiently transmitted to the human PrPC knock-in mice by the peripheral route. One (H3) out of the three M1 isolates showed infectivity to both mouse lines [15], another isolate (I22) showed weak infectivity only to Ki-Hu129V/V mice, and the other isolate (J56) lacked infectivity. The total attack rate in the M1 strain-inoculated mice was 16% (6% in Ki-Hu129M/M and 26% in Ki-Hu129V/V). We have reported previously that the M1 isolate (H3)-inoculated Ki-Hu129M/M or Ki-Hu129V/V mice produced type 1 PrPSc (21 kDa) [15]. However, a single positive Ki-Hu129V/V mouse in the M1 isolate (I22)-inoculated group produced an unusual PrPSc, which migrated faster than the intermediate type PrPSc in the V2 isolate-inoculated Ki-Hu129M/M mice (Fig. 1). The M2C and M2T sCJD strains were not transmitted to both Ki-Hu129M/M and Ki-Hu129V/V mice by the peripheral route (Table 1).
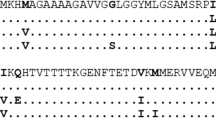
An antibody reactive for all PrPSc types, 3F4, and a type 2 PrPSc-specific antibody, Tohoku 2 [19], were used for the western blotting. After challenge with the M1 isolate (I22), Ki-Hu129M/M mice lacked PrPSc accumulation in the spleen, while a single positive Ki-Hu129V/V mouse produced an unusual PrPSc, which migrated faster than the intermediate type PrPSc in the V2 isolate (AK)-inoculated Ki-Hu129M/M mice. The production of the intermediate type PrPSc in the V2 isolate (AK)-inoculated Ki-Hu129M/M mice and that of type 2 PrPSc in the V2 isolate (AK)-inoculated Ki-Hu129V/V mice have already been reported [20]. The M2C isolate (H93)- or the M2T isolate (J86)-inoculated mice lacked PrPSc accumulation in the spleen. MM Ki-Hu129M/M mice, VV Ki-Hu129V/V mice.
Discussion
Here we investigated systematically the infectivity of sCJD strains through the peripheral route for the first time. The V2 strain showed stable infectivity, whereas the M1 strain showed only weak fluctuating infectivity. The M2C and M2T strains lacked infectivity. Of note, both the V2 and M1 strains showed relative preference for the mice with the 129 V/V genotype. These findings suggest that the V2 strain is the most infectious sCJD strain in infection through peripheral routes.
The infectivity of the M1 sCJD strain through the peripheral route was lower than that of the V2 sCJD strain. In intracerebral transmission studies, the M1 strain infected the human PrPC knock-in mice as efficiently as the V2 strain [6, 19]. Therefore, peripheral tissue-specific factor(s) might reduce the infectivity of the M1 strain. Peripherally infected PrPSc is taken up by dendritic cells and is transported to lymphoid follicles [21, 22]. In lymphoid follicles, PrPSc is passed from dendritic cells to follicular dendritic cells and is replicated in follicular dendritic cells before invasion into the peripheral nervous system [23,24,25]. In this neuroinvasion process, the M1 PrPSc may have different properties compared to the V2 PrPSc, e.g., stability in peripheral tissues, the efficiency of uptake by immune cells, or seeding activity for the replication in follicular dendritic cells. Recent reports suggest that spleen-derived PrPSc has more sialic acid on their N-linked glycans than brain-derived PrPSc [26], and that electrostatic repulsion between sialic acid molecules on N-linked glycans creates structural constraints controlling PrPSc replication rate [27]. Therefore, M1 PrPSc replication might be more affected by the sialylation of N-linked glycans than V2 PrPSc replication. Further studies will be needed in the future to unveil the reason for the reduced infectivity of the M1 strain through the peripheral route. On the other hand, in intracerebrally inoculated mice, the incubation period of the V2 strain was the shortest among the sCJD strains [6, 16, 19]. Therefore, the V2 strain might have the highest infectivity regardless of the route of infection. To address this point, further studies with quantitative infectivity titration will be needed.
The present study indicates that the underrepresentation of the M1 strain-originated subgroup and the overrepresentation of the V2 strain-originated subgroup in human growth hormone-associated iCJD may be due to the weak infectivity of the M1 strain through peripheral routes. It is noteworthy that the M2C strain did not infect through the peripheral route in the present study, despite the finding that four out of 21 human growth hormone-associated iCJD patients showed neuropathologic features of the MM2C subtype besides VV2/MV2 features [12]. The infectivity of the M2C strain is extremely low even in intracerebral transmission experiments [6, 10], and transmission of the M2C strain has not been observed in dura mater graft-associated iCJD [10]. In addition, the duration of illness of the MM2C subtype is the longest among the sCJD subtypes [8]. Therefore, the replication of the M2C PrPSc might occur much less efficiently and require longer time, resulting in the lack of positive transmission in the present study. The reason for the discrepancy between the lack of positive transmission in the present study and the presence of the MM2C-like neuropathologic features in a part of human growth hormone-associated iCJD patients needs to be addressed in the future.
Unexpectedly, the M1 sCJD strain showed relative preference for the host with the 129 V/V genotype in infection through the peripheral route. In intracerebral transmission experiments, the M1 and V2 strains are transmissible to both the 129 M/M and 129 V/V mice with almost 100% attack rates, but the incubation periods from inoculation to disease onset are prolonged in the codon 129 genotype-mismatched host [6, 19], suggesting that the homology of the codon 129 genotype between the inoculated sCJD strain and the host determines the efficiency of PrPSc replication. Indeed, the V2 strain showed preference for the host with the 129 V/V genotype also in the present study. Although these findings raise the possibility that individuals with the 129 V/V genotype might have higher potential risk of infection with CJD through peripheral routes compared with those with the 129 M/M genotype, human growth hormone-associated iCJD case with the 129 V/V genotype and MM1-like pathology or PrPSc type has not been recognized [11, 12]. In addition, the attack rates in the M1 strain-inoculated groups were variable, and the numbers of positive animals were very low in the present study. Furthermore, the present experimental design included neither an analysis of the spread of PrPSc from the spleen to the brain nor other peripheral routes such as subcutaneous inoculation. Therefore, further studies with more M1 isolates or other routes of inoculation will be required in the future to conclude that individuals with the 129 V/V genotype have higher potential risk of infection through peripheral routes. Furthermore, it will be of interest to investigate the pathology and PrPSc profiles in the brain of the intraperitoneally inoculated mice after disease onset. Particularly, in the present study, a single Ki-Hu129V/V mouse in the M1 isolate (I22)-inoculated group produced an unusual PrPSc migrating faster than the intermediate type PrPSc. This finding is not compatible with the data from the intracerebral transmission study of the M1 strain or those of the intraperitoneal transmission study of the other M1 isolate [6, 15, 20]. Therefore, it will be important to examine whether the change of PrPSc profiles could be reproduced in the brain of the Ki-Hu129V/V mice intraperitoneally inoculated with the M1 isolate (I22).
In conclusion, the present study shows that the V2 sCJD strain is more efficiently transmitted through the peripheral route than the M1 sCJD strain. Indeed, the V2 strain-originated subgroup has been reported not only in human growth hormone-associated iCJD patients but also in an occupational exposure-associated iCJD patient [28]. Since the current PrPSc disinfection/decontamination procedures had been developed using scrapie and CJD isolates before sCJD classification was established, the efficacy of these procedures against the V2 sCJD strain needs to be reconfirmed. In addition, as the presence of infectivity has been reported in a wide variety of peripheral tissues of sCJD MM1 patients including the lung, heart, kidney, pancreas, salivary gland, thyroid, adrenal gland, skeletal muscle, and lymphoid tissues [29], the distribution of infectivity in peripheral tissues should be investigated also in sCJD VV2 and MV2 patients. Furthermore, elucidation of the reason for the reduced infectivity of the M1 sCJD strain through the peripheral route will pave the way for the development of preventive measures against iCJD transmission through peripheral routes. Finally, as lymphoid tissues are more permissive than brain to cross-species transmission of chronic wasting disease and bovine spongiform encephalopathy [30], the bioassay system used in the present study will aid estimate the potential for PrPSc infection through peripheral routes in animal-to-human transmission as well as human-to-human transmission.
Data availability
The data that support the findings are available from the corresponding author upon request.
References
Prusiner SB, Scott MR, DeArmond SJ, Cohen FE. Prion protein biology. Cell. 1998;93:337–48.
Colby DW, Prusiner SB. Prions. Cold Spring Harb Perspect Biol. 2011;3:a006833.
Brown P, Brandel JP, Sato T, Nakamura Y, MacKenzie J, Will RG, et al. Iatrogenic Creutzfeldt-Jakob disease, final assessment. Emerg Infect Dis. 2012;18:901–7.
Kobayashi A, Kitamoto T, Mizusawa H. Iatrogenic Creutzfeldt-Jakob disease. Handb Clin Neurol. 2018;153:207–18.
Parchi P, Giese A, Capellari S, Brown P, Schulz-Schaeffer W, Windl O, et al. Classification of sporadic Creutzfeldt-Jakob disease based on molecular and phenotypic analysis of 300 subjects. Ann Neurol. 1999;46:224–33.
Bishop MT, Will RG, Manson JC. Defining sporadic Creutzfeldt-Jakob disease strains and their transmission properties. Proc Natl Acad Sci USA. 2010;107:12005–10.
Moda F, Suardi S, Di Fede G, Indaco A, Limido L, Vimercati C, et al. MM2-thalamic Creutzfeldt-Jakob disease: neuropathological, biochemical and transmission studies identify a distinctive prion strain. Brain Pathol. 2012;22:662–9.
Parchi P, Strammiello R, Giese A, Kretzschmar H. Phenotypic variability of sporadic human prion disease and its molecular basis: past, present, and future. Acta Neuropathol. 2011;121:91–112.
Kobayashi A, Matsuura Y, Mohri S, Kitamoto T. Distinct origins of dura mater graft-associated Creutzfeldt-Jakob disease: past and future problems. Acta Neuropathol Commun. 2014;2:32.
Kobayashi A, Matsuura Y, Iwaki T, Iwasaki Y, Yoshida M, Takahashi H, et al. Sporadic Creutzfeldt-Jakob disease MM1+2C and MM1 are identical in transmission properties. Brain Pathol. 2016;26:95–101.
Rudge P, Jaunmuktane Z, Adlard P, Bjurstrom N, Caine D, Lowe J, et al. Iatrogenic CJD due to pituitary-derived growth hormone with genetically determined incubation times of up to 40 years. Brain. 2015;138:3386–99.
Ritchie DL, Barria MA, Peden AH, Yull HM, Kirkpatrick J, Adlard P, et al. UK iatrogenic Creutzfeldt-Jakob disease: investigating human prion transmission across genotypic barriers using human tissue-based and molecular approaches. Acta Neuropathol. 2017;133:579–95.
McLean CA, Ironside JW, Alpers MP, Brown PW, Cervenakova L, Anderson RM, et al. Comparative neuropathology of kuru with the new variant of Creutzfeldt-Jakob disease: evidence for strain of agent predominating over genotype of host. Brain Pathol. 1998;8:429–37.
Parchi P, Cescatti M, Notari S, Schulz-Schaeffer WJ, Capellari S, Giese A, et al. Agent strain variation in human prion disease: insights from a molecular and pathological review of the National Institutes of Health series of experimentally transmitted disease. Brain. 2010;133:3030–42.
Munesue Y, Shimazaki T, Qi Z, Isoda N, Sawa H, Aoshima K, et al. Development of a quick bioassay for the evaluation of transmission properties of acquired prion diseases. Neurosci Lett. 2018;668:43–47.
Takeuchi A, Mohri S, Kai H, Tamaoka A, Kobayashi A, Mizusawa H, et al. Two distinct prions in fatal familial insomnia and its sporadic form. Brain Commun. 2019;1:fcz054.
Asano M, Mohri S, Ironside JW, Ito M, Tamaoki N, Kitamoto T. vCJD prion acquires altered virulence through trans-species infection. Biochem Biophys Res Commun. 2006;342:293–9.
Kitamoto T, Mohri S, Ironside JW, Miyoshi I, Tanaka T, Kitamoto N, et al. Follicular dendritic cell of the knock-in mouse provides a new bioassay for human prions. Biochem Biophys Res Commun. 2002;294:280–6.
Kobayashi A, Sakuma N, Matsuura Y, Mohri S, Aguzzi A, Kitamoto T. Experimental verification of a traceback phenomenon in prion infection. J Virol. 2010;84:3230–8.
Kobayashi A, Asano M, Mohri S, Kitamoto T. Cross-sequence transmission of sporadic Creutzfeldt-Jakob disease creates a new prion strain. J Biol Chem. 2007;282:30022–8.
Raymond CR, Aucouturier P, Mabbott NA. In vivo depletion of CD11c+ cells impairs scrapie agent neuroinvasion from the intestine. J Immunol. 2007;179:7758–66.
Cordier-Dirikoc S, Chabry J. Temporary depletion of CD11c+ dendritic cells delays lymphoinvasion after intraperitonal scrapie infection. J Virol. 2008;82:8933–6.
Kitamoto T, Muramoto T, Mohri S, Doh-Ura K, Tateishi J. Abnormal isoform of prion protein accumulates in follicular dendritic cells in mice with Creutzfeldt-Jakob disease. J Virol. 1991;65:6292–5.
Mabbott NA, Mackay F, Minns F, Bruce ME. Temporary inactivation of follicular dendritic cells delays neuroinvasion of scrapie. Nat Med. 2000;6:719–20.
McCulloch L, Brown KL, Bradford BM, Hopkins J, Bailey M, Rajewsky K, et al. Follicular dendritic cell-specific prion protein (PrP) expression alone is sufficient to sustain prion infection in the spleen. PLoS Pathog. 2011;7:e1002402.
Srivastava S, Makarava N, Katorcha E, Savtchenko R, Brossmer R, Baskakov IV. Post-conversion sialylation of prions in lymphoid tissues. Proc Natl Acad Sci USA. 2015;112:E6654–62.
Katorcha E, Makarava N, Savtchenko R, Baskakov IV. Sialylation of the prion protein glycans controls prion replication rate and glycoform ratio. Sci Rep. 2015;5:16912.
Kobayashi A, Parchi P, Yamada M, Brown P, Saverioni D, Matsuura Y, et al. Transmission properties of atypical Creutzfeldt-Jakob disease: a clue to disease etiology? J Virol. 2015;89:3939–46.
Douet JY, Huor A, Cassard H, Lugan S, Aron N, Arnold M, et al. Wide distribution of prion infectivity in the peripheral tissues of vCJD and sCJD patients. Acta Neuropathol. 2021;141:383–97.
Béringue V, Herzog L, Jaumain E, Reine F, Sibille P, Le Dur A, et al. Facilitated cross-species transmission of prions in extraneural tissue. Science. 2012;335:472–5.
Acknowledgements
We thank members of the Creutzfeldt–Jakob Disease Surveillance Committee in Japan, Creutzfeldt–Jakob disease specialists in the prefectures, and Creutzfeldt–Jakob disease patients and families for providing important clinical information. We thank Hiroko Kudo, Miyuki Yamamoto, and Ayumi Yamazaki for their excellent technical assistance.
Funding
This study was supported by JSPS KAKENHI Grant Number 18K05963 (AK), a grant from The Kato Memorial Trust for Nambyo Research (AK), The Kurata Grants from The Hitachi Global Foundation (AK), and a grant from Takeda Science Foundation (AK).
Author information
Authors and Affiliations
Contributions
Conceptualization: AK and T Kitamoto; Methodology: SM and T Kitamoto; Formal analysis and investigation: AK, YM, and TS; Writing—original draft preparation: AK; Writing—review and editing: KA, T Kimura, SM, T Kitamoto; Funding acquisition: AK; Resources: T Kitamoto, SM; Supervision: T Kimura, SM, and T Kitamoto.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
This study was approved by the Institutional Ethics Committee of Hokkaido University Faculty of Veterinary Medicine (approval number: VET1-5) and the Institutional Animal Care and Use Committees of Hokkaido University (approval number: 19-0025). All experiments using human materials are in compliance with the Helsinki Declaration. Animal experiments were performed in strict accordance with the Regulations for Animal Experiments and Related Activities at Hokkaido University.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kobayashi, A., Munesue, Y., Shimazaki, T. et al. Potential for transmission of sporadic Creutzfeldt–Jakob disease through peripheral routes. Lab Invest 101, 1327–1330 (2021). https://doi.org/10.1038/s41374-021-00641-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41374-021-00641-2
This article is cited by
-
Prion type 2 selection in sporadic Creutzfeldt–Jakob disease affecting peripheral ganglia
Acta Neuropathologica Communications (2021)