Abstract
The wound-healing process is a natural response to burn injury. Resveratrol (RES) may have potential as a therapy for wound healing, but how and whether RES regulates skin repair remains poorly understood. Human epidermal keratinocyte (HaCaT) cells were treated with lipopolysaccharide (LPS), and a mouse skin wound-healing model was established. Cell viability and apoptosis were analyzed by 3-(4,5-dimethyl-2-thiazolyl)−2,5-diphenyl-2-H-tetrazolium bromide or flow cytometry. Cell proliferation was assessed by cell viability and colony-formation analyses. Cell migration was tested by wound-healing analysis. The microRNA-212 (miR-212) and caspase-8 (CASP8) levels were determined by quantitative reverse transcription polymerase chain reaction and western blotting. The correlation between miR-212 and CASP8 was analyzed by dual-luciferase reporter analysis. Skin wound healing in mice was assessed by measuring the wound area and gap after hematoxylin–eosin (HE) staining. RES reduced the LPS-induced reduction in viability and apoptosis in HaCaT cells. miR-212 expression was reduced by LPS and increased by exposure to RES. RES promoted cell proliferation and migration after LPS treatment by increasing miR-212 levels. CASP8 was a target of miR-212. CASP8 silencing promoted cell proliferation and migration, which was reversed by miR-212 knockdown in LPS-treated HaCaT cells. RES promoted skin wound healing in mice, which was reduced by miR-212 knockdown. Thus, RES facilitates cell proliferation and migration in LPS-treated HaCaT cells and promotes skin wound-healing in a mouse model by regulating the miR-212/CASP8 axis.
Similar content being viewed by others
Introduction
Burns from fire or scalds are a prevalent problem, and large improvements have been made in burn wound care and treatment [1]. Burn wound healing is a complex process, and keratinocytes are used as skin substitutes for wound treatment [2]. Keratinocyte proliferation and migration contribute to burn wound healing [3]. Thus, exploring the mechanism of keratinocyte proliferation and migration may help to find new strategies for burn wound healing.
There are various natural treatments available for treating wounds, such as bee products and its active monomer, used in Chinese medicine [4, 5]. Resveratrol (RES) is a polyphenol with protective effects in human cancers and diseases that has been identified in 185 plants [6]. RES can promote incisional, cutaneous, uterine, and diabetic wound healing [7,8,9,10]. Furthermore, RES can mitigate lipopolysaccharide (LPS)-induced keratinocyte injury by regulating microRNA-17 [11]. Nevertheless, the mechanism of RES in burn wound healing is complex and requires additional investigation.
MicroRNAs (miRNAs) are a group of noncoding RNAs interacting with targeted mRNAs to regulate wound healing [12]. Multiple studies suggest that miRNAs, such as miR-126, miR-17, and miR-155, are relevant to keratinocyte proliferation and migration [13,14,15]. Previous evidence indicates that miRNAs are related to the function of RES in human disorders [16]. miR-212 promotes cell proliferation and migration in non-small cell lung cancer and pancreatic cancer [17, 18]. Nevertheless, whether miR-212 is required for RES in regulating wound healing is unknown.
Human epidermal keratinocytes (HaCaTs) are widely used for research on burn injury in vitro [11, 19]. In bacterial infection, LPS triggers an inflammatory state, and it can be used to induce an inflammatory injury in skin [20]. Hence, we employed LPS to stimulate HaCaT cells to mimic burn injury in vitro, as previously reported [11, 21]. Moreover, a mouse skin wound healing model has been widely established using biopsy punches [22, 23]. In this study, we established LPS-treated keratinocytes (HaCaT) and a skin wound-healing model in mice. Moreover, we explored the function of RES in HaCaT cell proliferation and migration and wound healing in mice. In addition, we analyzed whether RES modulated HaCaT cell processes by the miR-212/caspase-8 (CASP8) axis. Our study aimed to provide new insights into the pathogenesis and treatment of burns.
Materials and methods
Cell culture and exposure to LPS and RES
The human immortalized keratinocyte HaCaT cell line (CL-0090; Procell, Wuhan, China) was grown in minimum Eagle’s medium (MEM; Procell) with 15% fetal bovine serum (Thermo Fisher, Waltham, MA, USA) and 1% penicillin/streptomycin (Thermo Fisher) in 5% CO2 at 37 °C.
To mimic skin wounds in vitro, HaCaT cells were stimulated with various doses (0, 2, 4, 6, 8 and 10 μg/mL) of LPS (Solarbio, Beijing, China) for 6 h, as previously reported [24]. To analyze the function of RES in LPS-induced HaCaT cell injury, the cells were exposed to various doses (0, 2, 5 and 10 μM) of RES (C14H12O3; Santa Cruz Biotechnology, Santa Cruz, CA, USA) for 24 h and then treated with 6 μg/mL LPS for 6 h.
Cell viability
Cell viability was analyzed by 3-(4,5-dimethyl-2-thiazolyl)−2,5-diphenyl-2-H-tetrazolium bromide (MTT). A total of 1 × 104 HaCaT cells per well were placed into 96-well plates overnight and exposed to various doses of LPS for 6 h or RES for 24 h; after the indicated transfection or treatment with RES or LPS, 1 × 104 HaCaT cells per well were added to 96-well plates and then incubated for 0, 24, 48, or 72 h. Next, the cell medium was changed to fresh medium supplemented with 0.5 mg/mL MTT (Solarbio), and the cells were cultured for another 4 h. Subsequently, the medium was discarded and replaced with 100 μL of dimethyl sulfoxide (Solarbio). The absorbance was examined by use of a microplate reader (Molecular Devices, Sunnyvale, CA, USA) at a 570-nm wavelength. The cell viability was normalized to the control group (100%).
Cell apoptosis
Cell apoptosis was tested with an Annexin V-FITC Apoptosis Detection Kit (Abcam, Cambridge, MA, USA) by flow cytometry. After exposure to 6 μg/mL LPS for 6 h with or without pretreatment with 10 μM RES for 24 h, 1 × 105 HaCaT cells were harvested, incubated with annexin V binding buffer, and then stained with annexin V-FITC and propidium iodide (PI) for 5 min. Apoptotic cells were examined with a flow cytometer (Agilent, Hangzhou, China). The apoptotic rate was expressed as the percentage of cells in the upper- and lower-right quadrants.
Colony formation
After the indicated transfection or treatment with RES or LPS, 1000 HaCaT cells per well in Eagle’s medium supplemented with 15% fetal bovine serum were added to 6-well plates and then cultured for 14 days. After incubation, the cells were fixed with 4% paraformaldehyde (Beyotime, Shanghai, China) and stained with 0.2% crystal violet (Beyotime). The cellular colonies were photographed and counted under a microscope (Olympus, Tokyo, Japan).
Wound-healing analysis
The migratory abilities of HaCaT cells were analyzed by wound-healing analysis. After stimulation with 6 μg/mL LPS for 6 h with or without pretreatment with 10 μM RES for 24 h, 2 × 105 HaCaT cells per well were placed into 12-well plates. When the cells reached 95% confluence, a straight scratch was made by use of a 200-μL pipette tip. Images of the wound healing were obtained at 0 h and 24 h. The migrated cell number was counted in three random fields.
Quantitative reverse transcription polymerase chain reaction (qRT-PCR)
RNA was extracted with TRIzol reagent (Thermo Fisher) using the acid guanidinium thiocyanate–phenol–chloroform method [25]. RNA (500 ng) was reverse transcribed to cDNA using a specific reverse transcription kit (iGene Biotechnology, Guangzhou, China). The cDNA was mixed with SYBR (Thermo Fisher) and specific primers (Sangon, Shanghai, China), and then the mixture was used for qRT-PCR under the following conditions: 95 °C for 5 min, 40 cycles of 95 °C for 20 s, and 60 °C for 1 min. The primers are shown in Table 1. U6 or β-actin was used as a reference for miR-212 and CASP8. The relative RNA expression was tested with the 2−ΔΔCt method [26].
Oligonucleotide constructs and cell transfection
The miR-212 mimic (5′-UAACAGUCUCCAGUCACGGCC-3′), negative control mimic (NC mimic) (5′-CGAUCGCAUCAGCAUCGAUUGC-3′), siRNA for CASP8 (si-CASP8) (5′-AAACUUUGUCCAAAGUCUGUG-3′), negative control siRNA (si-NC) (5′-AACAGUCGCGUUUGCGACUGG-3′), miR-212 inhibitor (5′-GGCCGUGACUGGAGACUGUUA-3′), and negative control inhibitor (NC inhibitor) (5′-UGAGCUGCAUAGAGUAGUGAUUA-3′) were generated by RiboBio (Guangzhou, China). HaCaT cells at 70% confluence were transfected with 20 nM oligonucleotides with Lipofectamine 2000 (Thermo Fisher) for 24 h. The transfection efficacy was confirmed by qRT-PCR.
Western blot
Total protein was isolated using RIPA buffer (Beyotime), and the concentration was measured using a BCA kit (Thermo Fisher). The protein (20 μg) was separated using sodium dodecyl sulfate–polyacrylamide gel electrophoresis and transferred to polyvinylidene difluoride membranes (Solarbio). Next, the membranes were blocked in 5% BSA (Solarbio) for 1 h and then incubated with specific anti-CASP8 (ab25901, 1:2000 dilution, Abcam) or anti-β-actin (ab8227, 1:3000 dilution, Abcam) primary antibodies and a horseradish peroxidase-conjugated IgG (ab6721, 1:10000 dilution, Abcam) secondary antibody. β-actin functioned as a loading reference. After exposure to ECL reagent (Solarbio), the blots were analyzed with ImageJ software (NIH, Bethesda, MD, USA). The relative protein expression was normalized to the corresponding control group.
Dual-luciferase reporter analysis
The target relationship of miR-212 and CASP8 was predicted by the use of starBase. The luciferase reporter vectors WT-CASP8 and MUT-CASP8 were generated by inserting the wild-type or mutant 3′UTR sequence of CASP8 containing the miR-212-binding sites into the pMIR reporter (Thermo Fisher). HaCaT cells were cotransfected with WT-CASP8, MUT-CASP8, or control vector and NC mimic, miR-212 mimic, NC inhibitor or miR-212 inhibitor for 24 h. Luciferase activity was detected with a dual-luciferase analysis kit (Promega, Madison, WI, USA).
Animals and treatment
Ten-week-old male C57/BL6 mice were provided by the Shanghai SLAC Laboratory Animal Co., Ltd. (Shanghai, China) and arbitrarily divided into 5 groups (n = 5/group): normal group (nonwound), control (wound) group (full-thickness wounds in the dorsal skin using a 5-mm biopsy punch, as described in a previous report [22]), RES group (full-thickness wounds in the dorsal skin using a 5-mm biopsy punch, and the wounds were covered with 10 μM RES), RES + NC inhibitor group (full-thickness wounds in the dorsal skin using a 5-mm biopsy punch, and the wounds were covered with 10 μM RES, together with intradermal injection of NC inhibitor (iGene Biotechnology) into the wound edges), and RES + miR-212 inhibitor group (full-thickness wounds in the dorsal skin using a 5-mm biopsy punch, and the wounds were covered with 10 μM RES, together with intradermal injection of miR-212 inhibitor (iGene Biotechnology) into the wound edges). The related procedures were performed under anesthesia using 1.5% isoflurane. Photographs were taken of the wound site at 0, 4, 7 or 10 days postwounding, and the wound area was analyzed. The skin was harvested at the indicated times for histological examination with hematoxylin–eosin (HE) staining, and the wound gap was examined. miR-212 and CASP8 abundances were assessed in skin in the normal and wound groups. The animal experiments were approved by the Animal Ethical Committee of The Third Xiangya Hospital and conducted in line with the guidelines of the National Institutes of Health.
HE staining
The wound tissues were subjected to paraffin embedding, cut into 5-µm sections, and then subjected to hematoxylin and eosin staining (Beyotime). The sections were observed under a microscope.
Statistical analysis
The data were analyzed using GraphPad Prism 7 (GraphPad Inc., La Jolla, CA, USA). Three independent experiments were repeated, and the results are displayed as the mean ± SD. The difference between groups was tested via Student’s t-test or ANOVA followed by Tukey’s test. P < 0.05 indicated a significant difference.
Results
RES attenuates LPS-induced damage in HaCaT cells
The skin wound model was established using LPS-treated HaCaT cells in vitro. After exposure to various doses of LPS for 6 h, HaCaT cell viability was decreased in a dose-dependent manner (Fig. 1A). LPS (6 μg/mL) that induced an approximately 50% viability reduction was chosen for subsequent experiments. As displayed in Fig. 1B, stimulation with LPS caused evident apoptosis in HaCaT cells. To analyze the function of RES in the LPS-induced damage in HaCaT cells, cells were treated with various doses of RES for 24 h. As shown in Fig. 1C, RES showed little effect on HaCaT cell viability, except when its concentration reached 20 μM. Thus, nontoxic doses (0–10 μM) of RES were used for subsequent experiments. The results showed that pretreatment with RES protected against LPS-induced viability reduction in a dose-dependent manner (Fig. 1D). Ten micromolar RES, with the largest relative influence, was used as the exposure dose for subsequent experiments. As shown in Fig. 1E, treatment with RES reduced LPS-induced HaCaT cell apoptosis. Taken together, RES mitigated LPS-induced HaCaT cell injury.
A HaCaT cell viability was detected after exposure to various doses of LPS for 6 h by MTT. B HaCaT cell apoptosis was measured after stimulation with LPS by flow cytometry. C HaCaT cell viability was examined after treatment with different doses of RES for 24 h by MTT. D HaCaT cell viability was measured after exposure to different doses of RES and LPS by MTT. E HaCaT cell apoptosis was detected after stimulation with RES and LPS by flow cytometry. *P < 0.05, **P < 0.01, ***P < 0.001.
RES regulates miR-212 to promote cell proliferation and migration in LPS-treated HaCaT cells
As shown in Fig. 2A, the miR-212 abundance was evidently reduced in HaCaT cells after LPS treatment, while it was progressively upregulated after exposure to RES in a dose-dependent manner. To analyze whether miR-212 was relevant to RES-mediated regulation of LPS-induced damage, HaCaT cells were transfected with miR-212 inhibitor or NC inhibitor and then exposed to RES and LPS. As displayed in Fig. 2B, the miR-212 abundance in HaCaT cells was effectively decreased after transfection with the miR-212 inhibitor compared to the NC inhibitor. Furthermore, HaCaT cell proliferation was obviously inhibited by LPS treatment. In addition, exposure to RES reversed the LPS-induced inhibition of HaCaT cell proliferation, and this effect was reduced by miR-212 knockdown (Figs. 2C and 2D). In addition, cell migration was suppressed by LPS treatment and restored with the introduction of RES, which was mitigated by miR-212 knockdown (Fig. 2E). These results suggested that RES promoted cell proliferation and migration due to the upregulation of miR-212 in LPS-treated HaCaT cells.
A miR-212 abundance was detected in HaCaT cells after exposure to different doses of RES and LPS by qRT-PCR. B miR-212 abundance was detected in HaCaT cells transfected with miR-212 inhibitor or NC inhibitor. C–E Cell viability C, colony-formation ability D, and migration E were measured in HaCaT cells transfected with miR-212 inhibitor or NC inhibitor after exposure to RES and LPS by MTT, colony formation, and wound-healing analyses. *P < 0.05, **P < 0.01, ***P < 0.001.
miR-212 targets CASP8
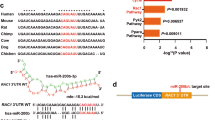
To analyze the regulatory network of miR-212, its targets were predicted by starBase. CASP8 was a candidate target of miR-212, and the target sequence is shown in Fig. 3A. To identify their interaction, we constructed the luciferase reporter vectors WT-CASP8 and MUT-CASP8. The dual-luciferase reporter analysis data showed that miR-212 overexpression evidently decreased the luciferase activity of WT-CASP8 and that miR-212 knockdown obviously elevated the luciferase activity of WT-CASP8, while neither miR-212 overexpression nor knockdown affected the luciferase activity of MUT-CASP8 (Fig. 3B). Furthermore, the influence of miR-212 on CASP8 was assessed in HaCaT cells transfected with an NC mimic, miR-212 mimic, NC inhibitor, and miR-212 inhibitor. The transfection efficacy of the miR-212 mimic and inhibitor was validated, as shown in Fig. 3C. In addition, CASP8 abundance was evidently reduced by miR-212 overexpression and enhanced by miR-212 knockdown (Figs. 3D and 3E). These data indicated that CASP8 was targeted by miR-212.
A The binding sequence of miR-212 and CASP8 predicted by starBase. B Luciferase activity was detected in HaCaT cells cotransfected with WT-CASP8 or MUT-CASP8 and NC mimic, miR-212 mimic, NC inhibitor or miR-212 inhibitor. C–E HaCaT cells transfected with NC mimic, miR-212 mimic, NC inhibitor, or miR-212 inhibitor. C miR-212 and D CASP8 abundances were examined by qRT-PCR. E The protein level of CASP8 was evaluated by western blot. *P < 0.05, **P < 0.01, ***P < 0.001.
miR-212 targets CASP8 to regulate cell proliferation and migration in LPS-treated HaCaT cells
To explore the function of the miR-212/CASP8 axis in LPS-induced HaCaT cell damage, HaCaT cells were transfected with si-NC, si-CASP8, si-CASP8 + miR-212 inhibitor, or NC inhibitor and then treated with LPS. After transfection, miR-212 expression was evidently reduced by transfection with the miR-212 inhibitor but was not affected after transfection with si-CASP8 (Fig. 4A). Moreover, CASP8 expression was markedly decreased due to transfection with si-CASP8, which was restored as a result of transfection of the miR-212 inhibitor (Fig. 4B). Furthermore, CASP8 silencing reversed the LPS-induced reduction in proliferation, and this effect was reduced due to the miR-212 knockdown (Figs. 4C and 4D). In addition, CASP8 interference promoted cell migration in LPS-treated HaCaT cells, which was reduced by miR-212 downregulation (Fig. 4E). These results suggested that miR-212 targeted CASP8 to regulate cell proliferation and migration in LPS-treated HaCaT cells.
A and B miR-212 and CASP8 abundances were detected in HaCaT cells transfected with si-NC, si-CASP8, si-CASP8 + miR-212 inhibitor, or NC inhibitor. C–E Cell viability C, colony-formation ability D, and migration E were examined in HaCaT cells transfected with si-NC, si-CASP8, si-CASP8 + miR-212 inhibitor, or NC inhibitor after exposure to LPS. *P < 0.05, **P < 0.01, ***P < 0.001.
RES promotes skin wound healing in mice through the miR-212/CASP8 axis
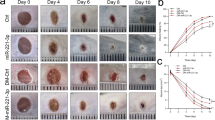
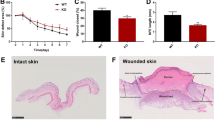
To further explore the function of RES in skin wound healing, a skin wound-healing model was established in mice, and then the mice were treated with RES alone or together with NC inhibitor or miR-212 inhibitor. As displayed in Figs. 5A and 5B, miR-212 expression was evidently reduced in wound tissues compared with the normal group, and CASP8 abundance was enhanced. Furthermore, treatment with RES markedly promoted skin wound healing from day 7 to day 10, as revealed by the reduced wound area, and this effect was reversed through miR-212 knockdown (Fig. 5C). In addition, the HE staining data showed that treatment with RES evidently reduced the wound at day 10, and this event was abolished by miR-212 downregulation (Fig. 5D). These data suggested that RES contributed to skin wound healing in mice by regulating the miR-212/CASP8 axis.
Discussion
Burn injuries can affect nearly all organ systems, and slow wound healing remains a challenge for burn treatment [27]. Skin is the main organ in response to burn injury, and skin healing contributes to improving burn treatment [28]. In this research, we found that RES promoted skin wound healing. Moreover, this study was the first to propose that the protective function of RES was associated with the miR-212/CASP8 axis.
LPS could induce keratinocyte injury by reducing cell viability and increasing apoptosis [24, 29]. Similarly, we also established an LPS-induced keratinocyte injury model using HaCaT cells. Previous reports suggested the promoting role of RES in incisional wound healing, cutaneous wound healing, uterine wound healing, and diabetic wound healing [7,8,9,10]. However, the benefits of RES in human health are dose-dependent, with a protective role at low doses and a proapoptotic role at high doses [30]. Pastore et al. reported that 50 μM RES obviously promoted apoptotic protein expression in keratinocytes [31]. In our study, HaCaT cells were exposed to a series of doses of RES, and we found that 0–10 μM RES showed little effect on cell viability, but 5 or 10 μM RES restored cell viability after LPS treatment, suggesting the potential protective function of RES in LPS-induced keratinocyte injury. Keratinocyte proliferation is related to the skin wound-healing process [32]. Here, we found that RES promoted keratinocyte proliferation after LPS exposure, which was also in agreement with a previous study [11]. Moreover, keratinocyte migration is also repressed by LPS [33]. Our work also indicated that RES could facilitate keratinocyte migration in the presence of LPS. Taken together, RES might play a protective role in wound healing by increasing keratinocyte proliferation and migration.
Abnormally expressed miRNAs are involved in skin wound healing [12]. Moreover, miRNAs can act as targets of RES in many conditions [34, 35]. A previous study reported that RES can upregulate miR-17 to mitigate LPS-induced keratinocyte injury [11]. Our study found that miR-212 was expressed at low levels in LPS-treated keratinocytes, and its abundance was restored by RES treatment, indicating that miR-212 might be required for RES-mediated function. In this study, miR-212 knockdown repressed keratinocyte proliferation and migration after LPS and RES exposure, which was also in agreement with that observed in non-small-cell lung cancer and pancreatic cancer, but opposite to that seen in hepatocellular carcinoma [17, 18, 36]. We hypothesized that the opposite function resulted from the different microenvironments. Furthermore, miR-212 knockdown attenuated the function of RES in keratinocyte proliferation and migration, suggesting that RES promotes keratinocyte proliferation and migration after LPS treatment by upregulating miR-212.
miRNAs affect the expression of targeted genes to regulate skin wound healing [37]. Here, we were the first to identify the target relationship between miR-212 and CASP8. CASP8 is an initiator caspase of the extrinsic apoptotic pathway [38]. A previous study indicated that CASP8 contributed to cyclodextrin-induced keratinocyte apoptosis [39]. Furthermore, CASP8 knockdown can facilitate cell proliferation and migration in oral squamous cell carcinoma [40]. Similarly, our study validated that CASP8 silencing promoted keratinocyte proliferation and migration after LPS treatment. Additionally, miR-212 knockdown attenuated this effect, indicating that CASP8 was implicated in RES-mediated regulation of the keratinocyte process via the crosstalk of miR-212. In addition, we further confirmed that RES could promote skin wound healing in mice through the miR-212/CASP8 axis.
In conclusion, RES promotes HaCaT cell proliferation and migration after LPS exposure and accelerates skin wound healing in mice, possibly by regulating the miR-212/CASP8 axis. This study provides a novel mechanism for understanding the function of RES in burn wound healing.
Data availability
All data generated or analyzed during this study are included in this article. The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Rowan MP, Cancio LC, Elster EA, Burmeister DM, Rose LF, Natesan S, et al. Burn wound healing and treatment: review and advancements. Crit Care. 2015;19:243.
Shakespeare P. Burn wound healing and skin substitutes. Burns. 2001;27:517–22.
Zhu C, Liu J, He B, Qu X, Peng D. The role of human immortal skin keratinocytes-acellular dermal matrix scaffold in skin repair and regeneration. J Cell Biochem. 2019;120:12182–91.
Liu Y, Sun J, Ma X, Li S, Ai M, Xu F. et al. Vaccarin regulates diabetic chronic wound healing through FOXP2/AGGF1 pathways. Int J Mol Sci. 2020;21:1966
Filipic B, Mazija H. Bee product apiderm royal gel in pressure injuries and wound care - case report. MOJ Toxicol. 2017;3:44–45.
Huminiecki L, Horbanczuk J. The functional genomic studies of resveratrol in respect to its anti-cancer effects. Biotechnol Adv. 2018;36:1699–708.
Yaman I, Derici H, Kara C, Kamer E, Diniz G, Ortac R, et al. Effects of resveratrol on incisional wound healing in rats. Surg Today. 2013;43:1433–8.
Zhao P, Sui BD, Liu N, Lv YJ, Zheng CX, Lu YB, et al. Anti-aging pharmacology in cutaneous wound healing: effects of metformin, resveratrol, and rapamycin by local application. Aging Cell. 2017;16:1083–93.
Sayin O, Micili SC, Goker A, Kamaci G, Ergur BU, Yilmaz O, et al. The role of resveratrol on full - Thickness uterine wound healing in rats. Taiwan J Obstet Gynecol. 2017;56:657–63.
Huang X, Sun J, Chen G, Niu C, Wang Y, Zhao C, et al. Resveratrol promotes diabetic wound healing via SIRT1-FOXO1-c-Myc signaling pathway-mediated angiogenesis. Front Pharmacol. 2019;10:421.
Wang X, Zhang Y. Resveratrol alleviates LPS-induced injury in human keratinocyte cell line HaCaT by up-regulation of miR-17. Biochem Biophys Res Commun. 2018;501:106–12.
Li P, He Q, Luo C, Qian L. Differentially expressed miRNAs in acute wound healing of the skin: a pilot study. Medicine. 2015;94:e458.
Chang L, Liang J, Xia X, Chen X. miRNA-126 enhances viability, colony formation, and migration of keratinocytes HaCaT cells by regulating PI3 K/AKT signaling pathway. Cell Biol Int. 2019;43:182–91.
Yan H, Song K, Zhang G. MicroRNA-17-3p promotes keratinocyte cells growth and metastasis via targeting MYOT and regulating Notch1/NF-kappaB pathways. Pharmazie. 2017;72:543–9.
Yang L, Zheng Z, Zhou Q, Bai X, Fan L, Yang C, et al. miR-155 promotes cutaneous wound healing through enhanced keratinocytes migration by MMP-2. J Mol Histol. 2017;48:147–55.
Lancon A, Kaminski J, Tili E, Michaille JJ, Latruffe N. Control of microRNA expression as a new way for resveratrol to deliver its beneficial effects. J Agric Food Chem. 2012;60:8783–9.
Li Y, Zhang D, Chen C, Ruan Z, Li Y, Huang Y. MicroRNA-212 displays tumor-promoting properties in non-small cell lung cancer cells and targets the hedgehog pathway receptor PTCH1. Mol Biol Cell. 2012;23:1423–34.
Ma C, Nong K, Wu B, Dong B, Bai Y, Zhu H, et al. miR-212 promotes pancreatic cancer cell growth and invasion by targeting the hedgehog signaling pathway receptor patched-1. J Exp Clin Cancer Res. 2014;33:54.
Wang Y, Maitz PKM. Advances and new technologies in the treatment of burn injury. Adv Drug Deliv Rev. 2018;123:1–2.
Crompton R, Williams H, Ansell D, Campbell L, Holden K, Cruickshank S, et al. Oestrogen promotes healing in a bacterial LPS model of delayed cutaneous wound repair. Lab Invest. 2016;96:439–49.
Zhang J, Zheng Q, Lu H, Jin F, Li Y, Bi F. et al. Notoginsenoside R1 protects human keratinocytes HaCaT from LPS-induced inflammatory injury by downregulation of Myd88. Int J Immunopathol Pharmacol. 2019;33:2058738419857550
Zhang J, Li L, Zhang Q, Wang W, Zhang D, Jia J, et al. Microtubule-associated protein 4 phosphorylation regulates epidermal keratinocyte migration and proliferation. Int J Biol Sci. 2019;15:1962–76.
Nagae K, Uchi H, Morino-Koga S, Tanaka Y, Oda M, Furue M. Glucagon-like peptide-1 analogue liraglutide facilitates wound healing by activating PI3K/Akt pathway in keratinocytes. Diabetes Res Clin Pract. 2018;146:155–61.
Hou XM, Zhang T, Da Z, Wu XA. CHPF promotes lung adenocarcinoma proliferation and anti-apoptosis via the MAPK pathway. Pathol Res Pract. 2019;215:988–94.
Chomczynski P, Sacchi N. The single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction: twenty-something years on. Nat Protoc. 2006;1:581–5.
Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods. 2001;25:402–8.
Wang Y, Beekman J, Hew J, Jackson S, Issler-Fisher AC, Parungao R, et al. Burn injury: challenges and advances in burn wound healing, infection, pain and scarring. Adv Drug Deliv Rev. 2018;123:3–17.
Shpichka A, Butnaru D, Bezrukov EA, Sukhanov RB, Atala A, Burdukovskii V, et al. Skin tissue regeneration for burn injury. Stem Cell Res Ther. 2019;10:94.
He Y, Kim BG, Kim HE, Sun Q, Shi S, Ma G. et al. The protective role of feruloylserotonin in LPS-induced HaCaT cells. Molecules. 2019;24:3064
Mukherjee S, Dudley JI, Das DK. Dose-dependency of resveratrol in providing health benefits. Dose Response. 2010;8:478–500.
Pastore S, Lulli D, Maurelli R, Dellambra E, De Luca C, Korkina LG. Resveratrol induces long-lasting IL-8 expression and peculiar EGFR activation/distribution in human keratinocytes: mechanisms and implications for skin administration. PLoS ONE. 2013;8:e59632.
Sui H, Wang F, Weng Z, Song H, Fang Y, Tang X, et al. A wheat germ-derived peptide YDWPGGRN facilitates skin wound-healing processes. Biochem Biophys Res Commun. 2020;524:943–50.
Loryman C, Mansbridge J. Inhibition of keratinocyte migration by lipopolysaccharide. Wound Repair Regen. 2008;16:45–51.
Latruffe N, Lançon A, Frazzi R, Aires V, Delmas D, Michaille JJ, et al. Exploring new ways of regulation by resveratrol involving miRNAs, with emphasis on inflammation. Ann N Y Acad Sci. 2015;1348:97–106.
Wang M, Jiang S, Yu F, Zhou L, Wang K. Noncoding RNAs as molecular targets of resveratrol underlying its anticancer effects. J Agric Food Chem. 2019;67:4709–19.
Chen JQ, Ou YL, Huang ZP, Hong YG, Tao YP, Wang ZG, et al. MicroRNA-212-3p inhibits the proliferation and invasion of human hepatocellular carcinoma cells by suppressing CTGF expression. Sci Rep. 2019;9:9820.
Fahs F, Bi X, Yu FS, Zhou L, Mi QS. New insights into microRNAs in skin wound healing. IUBMB Life. 2015;67:889–96.
Sakamaki K, Imai K, Tomii K, Miller DJ. Evolutionary analyses of caspase-8 and its paralogs: deep origins of the apoptotic signaling pathways. Bioessays. 2015;37:767–76.
Schonfelder U, Radestock A, Elsner P, Hipler UC. Cyclodextrin-induced apoptosis in human keratinocytes is caspase-8 dependent and accompanied by mitochondrial cytochrome c release. Exp Dermatol. 2006;15:883–90.
Hayes TF, Benaich N, Goldie SJ, Sipilä K, Ames-Draycott A, Cai W, et al. Integrative genomic and functional analysis of human oral squamous cell carcinoma cell lines reveals synergistic effects of FAT1 and CASP8 inactivation. Cancer Lett. 2016;383:106–14.
Acknowledgements
We would like to give our sincere gratitude to the reviewers for their constructive comments.
Funding
This work was supported by Natural Science Fund Project of Inner Mongolia autonomous region (2018BS08009); Inner Mongolia Medical University Science and Technology Million Project (YKD2017KJBW016), and Natural Science Foundation of Hunan Province (2020JJ5861).
Author information
Authors and Affiliations
Contributions
Conception and study design: Y. L.; Data acquisition: W. X.; Data analysis: C.-W. W., J.-P. S.; Paper drafting: Z.-Q. S.; Paper revising: J.-D. Z.
Corresponding author
Ethics declarations
Ethical approval
The animal experiments were approved via the Animal Ethical Committee of The Third Xiangya Hospital and conducted in line with the National Institutes of Health.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Liu, Y., Xiong, W., Wang, CW. et al. Resveratrol promotes skin wound healing by regulating the miR-212/CASP8 axis. Lab Invest 101, 1363–1370 (2021). https://doi.org/10.1038/s41374-021-00621-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41374-021-00621-6
This article is cited by
-
Hydroxysafflor Yellow A Promotes HaCaT Cell Proliferation and Migration by Regulating HBEGF/EGFR and PI3K/AKT Pathways and Circ_0084443
Chinese Journal of Integrative Medicine (2024)
-
Mechanism of Resveratrol Improving Ischemia–Reperfusion Injury by Regulating Microglial Function Through microRNA-450b-5p/KEAP1/Nrf2 Pathway
Molecular Biotechnology (2023)