Abstract
Sepsis is an acute inflammatory reaction and a cause of acute respiratory distress syndrome (ARDS). In the present study, we explored the roles and underlying mechanism of the lncRNA Nuclear enriched abundant transcript 1 (NEAT1) in ARDS. The expression levels of genes, proteins and pro-inflammatory cytokines in patients with ARDS, LPS-stimulated cells and septic mouse models were quantified using qPCR, western blotting and ELISA assays, respectively. The molecular targeting relationship was validated by conducting a dual-luciferase reporter assay. Cell proliferation was assessed using the Cell Counting Kit-8 (CCK-8) assay. The cell cycle phase was determined by flow cytometry assay. The expression levels of NEAT1 and pro-inflammatory cytokines were higher in patients with ARDS and septic models than in controls. Knockdown of NEAT1 significantly increased cell proliferation and cycle progression and prolonged mouse survival in vitro and in vivo. Mechanistically, miR-27a was identified as a downstream target of NEAT1 and directly inhibited PTEN expression. Further rescue experiments revealed that inhibition of miR-27a impeded the promoting effects of NEAT1 silence on cell proliferation and cycle progression, whereas inhibition of PTEN markedly weakened the inhibitory effects of NEAT1 overexpression on cell proliferation and cycle progression. Altogether, our study revealed that NEAT1 plays a promoting role in the progression of ARDS via the NEAT1/miR-27a/PTEN regulatory network, providing new insight into the pathologic mechanism behind ARDS.
Similar content being viewed by others
Introduction
Sepsis is a systemic inflammatory syndrome arising from the systemic immunological response of the host to infection [1]. It progresses rapidly and remains by far one of the most common causes of death of hospitalised patients [2, 3]. A study has shown that the lung is one of the organs easily damaged during sepsis, and ~25–50% of patients with sepsis may suffer acute lung injury [4]. Recently, sepsis-induced acute lung injury has been clinically defined as acute respiratory distress syndrome (ARDS), and is characterised by hypoxaemia, pulmonary oedema and inflammatory responses [5]. According to statistics, the incidence of ARDS has been reported to be 190,000 cases per year in the United States [6]. Although much progress has been made in the development of pharmacological interventions, the mortality of ARDS remains high due to complex sepsis-induced responses. Therefore, studies elucidating the physiological and molecular mechanisms of lung damage are necessary to identify effective therapeutic targets for ARDS.
Long noncoding RNAs (lncRNAs) are single-stranded RNAs with a length of 200 nucleotides [7]. lncRNAs frequently function as competing endogenous RNAs (ceRNAs) to regulate the expression levels of targeted genes by sponging microRNAs (miRNAs), and play critical roles in a variety of biological processes, such as cell differentiation, cell proliferation and apoptosis [8, 9]. According to recent evidence, lncRNAs participate in the disease progression of sepsis and inflammatory responses [10, 11]. For example, lncRNA MALAT1 aggravates skeletal muscle cell apoptosis and the inflammatory response caused by sepsis [12]. Nuclear enriched abundant transcript 1 (NEAT1) is an oncogenic lncRNA that facilitates the tumorigenesis of multiple cancers, such as colon cancer, prostate cancer and ovarian cancer [13]. Likewise, NEAT1 also plays an essential role in sepsis and the inflammatory response [14]. The expression level of NEAT1 is positively correlated with the severity of sepsis-induced myocardial injury [15]. Moreover, the expression levels of IL-6 and TNF-α in THP-1 cells are also regulated by NEAT1 [16]. However, the functional roles and mechanism of NEAT1 in sepsis-induced lung injury are still not very clear.
miRNAs are small noncoding RNAs of 18–22 nucleotides that negatively regulate gene expression by sponging the 3′-UTR of target genes, leading to mRNA degradation or translational repression [17, 18]. A member of a highly conserved miRNA family, miR-27, has been reported to be involved in various biological processes, including cell growth, differentiation and inflammation [19, 20]. miR-27a and miR-27b, which differ by only one nucleotide and represent two isoforms of the miR-27 family, have attracted extensive attention from researchers [21]. Recently, the expression level of miR-27a is reported to be twofold lower in RAW264.7 cells after LPS treatment [22]. Another study illustrated that miR-27a is significantly downregulated in liver tissue from septic mice and function as a therapeutic target of paclitaxel in septic mice [23]. According to a bioinformatics analysis, miR-27a may be a potential target of NEAT1. However, researchers have not clearly determined whether miR-27 participates in sepsis-induced ARDS.
In the present study, the roles and underlying mechanism of NEAT1 in sepsis-induced ARDS were investigated. The expression levels of NEAT1 in patients with ARDS, LPS-treated cells and CLP-induced septic mice were upregulated. Then, gain- or loss-of-function experiments of NEAT1 in vitro and in vivo confirmed that NEAT1 suppressed cell cycle progression and facilitated inflammatory responses, thus aggravating sepsis-induced lung injury by regulating the miR-27a/PTEN axis, which provides new insights into the development of ARDS.
Materials and methods
Clinical samples
Clinical blood samples from controls (n = 15) and patients with ARDS (n = 22) were collected from Guizhou Provincial People’s Hospital. Patients with ARDS presenting with sepsis included in this study were diagnosed according to the American College of Chest Physicians/Society of Critical Care Medicine [24]. The pathological information of clinical patients is provided in Supplementary material Table 1. Patients were excluded when they were younger than 18 years, were diagnosed with cancers or haematological malignancies, treated with immunosuppressive medication within 3 months, or were pregnant or lactating. Experimental controls were obtained from normal people who were not diagnosed with lung, cardiac, infectious, or allergic diseases and were not prescribed treatments for chronic diseases. Plasma samples were isolated from blood through centrifugation at 2000× g for 10 min at 4 °C and then stored at −80 °C until further detection. All participants provided informed consent, and all protocols were approved by the Ethics Committee of Guizhou Provincial People’s Hospital and conducted according to the Declaration of Helsinki.
Enzyme-linked immunosorbent assay
Concentrations of IL-1β, IL-6 and TNF-α in the serum and supernatant of cultured cells were measured using commercially specific enzyme-linked immunosorbent assay (ELISA) kits according to the manufacturer’s instructions (R&D, Minneapolis, Minnesota).
Quantitative PCR
Total RNA was extracted from cells, plasma samples and lung tissues obtained from septic mice using TRIzol reagent (Invitrogen, USA). The OD260/280 ratio of RNA samples was measured using a Nanodrop 2000 spectrophotometer (Thermo Scientific, Waltham, MA, USA). RNA samples with OD260/280 values ranging from 1.9 to 2.0 were reverse transcribed into cDNAs. First-strand cDNA synthesis was then performed using a miScript Reverse Transcription kit (Qiagen, Inc.) quantitative PCR (qPCR) was then conducted using the miScript SYBR® Green PCR kit (Qiagen, Inc.) and StepOnePlus Real-Time PCR Systems (7500; Thermo Scientific). The following reaction conditions were used: hot start at 95 °C for 5 min; 40 cycles of 30 s at 95 °C, 30 s at 60 °C and 30 s at 72 °C; and 10 min at 72 °C. The expression levels were quantified using the 2−ΔΔCt method. The following primer sequences were used for qPCR:
NEAT1 forward, 5′-CTTCCTCCCTTTAACTTATCCATTCAC-3′ and
NEAT1 reverse, 5′-CTCTTCCTCCACCATTACCAACAATAC-3
miR-27a forward, 5′- ATGGTTCGTGGGTTCACA -3′ and
miR-27a reverse, R: 5′- GTGGCTAAGTTCCGACG-3′;
PTEN forward, 5′-TTGAAGACCATAACCCACC-3′ and
PTEN reverse: 5′-AGTTCCGCCACTGAACAT-3′;
U6 forward: 5’- CTCGCTTCGGCAGCACA -3′ and
U6 reverse: 5′-AACGCTTCACGAATTTGCGT-3′; and
GAPDH forward, 5′-GCACCGTCAAGGCTGAGAAC-3′ and
GADPH reverse, 5′-TGGTGAAGACGCCAGTGGA-3′.
Cell culture and LPS treatment
BEAS-2B cells and 16HBE cells were purchased from ATCC (Manassas, VA) and cultured in BEGM media (Lonza, Mapleton, IL, USA) or RPMI 1640 medium (Invitrogen, Darmstadt, Germany) according to the manufacturer’s recommendations. Cells (1.0 × 105) were cultured in six-well plates with medium supplemented with 10% foetal bovine serum (FBS) and 1% penicillin-streptomycin (Grand Island, NY, USA) and grown at 37 °C in a humidified incubator with 5% CO2. The medium was changed every 48 h. Cells were grown to ~75% confluence and passaged. Cells were used after passage 3 in subsequent experiments. For LPS stimulation, LPS (1 μg/mL) was added to the medium for 24 h.
Cell transfections
The full NEAT1 sequence was amplified and inserted into the pcDNA-3.1 vector (Thermo Fisher Scientific). The shRNAs targeting NEAT1 and PTEN, miR-27a mimics, miR-27a inhibitor, and their respective negative control plasmids were purchased from GenePharma (Shanghai, China). Transfection was conducted using Lipofectamine 2000 reagent (Invitrogen, USA) according to the manufacturer’s protocol.
Cell cycle assay
Cells were collected by centrifugation (800×g, 6 min), washed three times with PBS and fixed with 70% ethanol at 4 °C for 48 h. After washing with PBS, cells were stained with 1 mL of staining solution (20 mg/mL propidium iodide and 10 U/mL RNase, Sigma-Aldrich) for 30 min at 37 °C. Then, the cell cycle was analysed using a FACScan flow cytometer (Becton-Dickinson, Brea, CA, USA).
Cell Counting kit-8 assay
Proliferation was measured using the CCK-8 kit (Dojindo, Japan) according to the manufacturer’s protocol. Briefly, 1.0 × 104 cells were cultured in 48-well microtiter plates for appropriate periods (0, 24, 48 or 72 h). Cells were collected, 10 μL of CCK-8 reagent were added to each well, and then the optical density values at 490 nm were measured using an Elx800 Reader (Bio-Tek Instruments Inc.).
Luciferase reporter assay
For reporter assays, 1.0 × 104 16HBE cells were seeded into 24-well plates. The 3′-UTR fragments of the lncRNA NEAT1 containing the wild-type or mutant miR-27a binding sites and the 3′UTR fragments of PTEN containing the wild-type or mutant miR-27a binding site were synthesised by GenePharma and inserted into a luciferase reporter gene plasmid (Invitrogen). Then, miR-27a mimics, the miR-27a inhibitor or their respective negative controls and reporter plasmids were co-transfected into cells using Lipofectamine 2000 (Invitrogen). Forty-eight hours after transfection, luciferase activities were measured and analysed using the Dual-Luciferase Reporter Assay System (Promega) according to the manufacturer’s protocols.
Western blot
Antibodies against p21 (1:1000, Abcam #ab7960), CDK2 (1:1000, Cell Signaling #CST2546), Cyclin D1 (1:1000, Abcam #137875), PTEN (1:800, Abcam #ab170941) and GAPDH (1:10000, Abcam #ab181602) were used according to the manufacturers’ protocols. After treatments, total proteins were extracted from 16HBE and BEAS-2B cells using cell lysis buffer containing proteinase inhibitors. The concentration was measured using a BCA kit (#23225, Pierce). Fifty micrograms of protein were electrophoresed on 10% SDS-PAGE gels using standard procedures and then transferred to PVDF membranes (Millipore). After three washes with TTBS, the membranes were blocked with 5% milk for 2 h. Then, membranes were incubated with primary antibodies at 4 °C overnight. After washing with TBST, blots were incubated with the appropriate secondary HRP antibody for 2 h. Finally, the bands were imaged with chemiluminescence reagents using a gel imaging system (#1708370, Life Science), and band intensity was quantified using ImageJ software.
CLP model of sepsis
Wild-type C57BL/6 mice (male, 8 weeks old) were obtained from The Jackson Laboratory (Bar Harbor, ME, USA). The mice were fed and maintained under the following conditions: food and water available ad libitum, temperature 23 ± 2 °C, humidity 60%, 12:12 h light/dark cycle. Mice were acclimated to the new environment for a week. sh-NEAT1 and its negative control (sh-NC) were packaged into the LV10-CMV-RFP-Puro vector (GenePharma, Shanghai, China). Then, the mice were randomly divided into four groups: the control (sham) group, CLP group, CLP + sh-NC group and CLP + sh-NEAT1 group (10 mice for each group). CLP surgery was performed on the 5th day after lentivirus injection as reported previously [25]. Briefly, the mouse caecum was ligated below the ileocecal valve. After ligation, the caecum was punctured once with a 23-gauge needle. Sham-operated mice underwent the same procedure but without ligation and puncture. Mice were euthanized by cervical dislocation after treatments, and lung tissues were immediately dissected for further studies. All procedures were performed in accordance with the national guidelines and approved by the Experimental Animal Ethics Committee of Guizhou Provincial People’s Hospital.
Statistical analysis
The results are presented as the means ± SD and were analysed with SigmaStat10.0 software (SPSS). The differences between groups were assessed using Student’s t test or one-way analysis of variance (ANOVA). P < 0.05 was considered statistically significant.
Results
lncRNA NEAT1 was upregulated of in ARDS
22 ARDS patients and 15 controls were enroled in this study, and the expression level of NEAT1 in plasma samples was examined. As shown in Fig. 1A, NEAT1 expression was increased in patients with ARDS compared to controls, suggesting that NEAT1 may be a regulator related to ARDS occurrence. Next, we detected pro-inflammatory cytokine levels using ELISA assay. The result revealed that the levels of TNF-α, IL-1β and IL-6 in patients with ARDS were much higher than those in the control group (Fig. 1B-1D). Stimulation of 16HBE cells and BEAS-2B cells with 1 µg/mL LPS for 24 h markedly increased the expression of NEAT1 (Fig. 1E). Similarly, the levels of inflammatory cytokines in the LPS group were also markedly increased compared with those in the control group (Fig. 1F), confirming that an in vitro cell model of inflammatory damage was successfully established. Subsequently, the cells at G1, S and G2/M phase were examined using flow cytometry. The results revealed that LPS treatment significantly increased the percentage of cells in G1 phase while reducing the percentage of cells in S phase (Fig. 1G), indicating that LPS treatment blocked G1-S cell cycle transition.
A Plasma samples were harvested from patients with ARDS and controls, and the expression level of NEAT1 was determined using qPCR. B-D Plasma samples were obtained from patients with ARDS and controls, and the levels of TNF-α, IL-6 and IL-1β were detected using ELISA. E-G 16HBE and BEAS-2B cells were untreated or treated with 1 µg/mL LPS for 24 h, and then cells and culture supernatants were collected. Next, the expression of NEAT1 was quantified using qPCR (E). The levels of TNF-α, IL-6 and IL-1β were detected using ELISA (F). The cell cycle phase was evaluated using flow cytometry (G). The results are presented as the means ± SD, and differences were assessed using Student’s t test or one-way analysis of variance. *P < 0.05, **P < 0.01, and ***P < 0.001.
Knockdown of NEAT1 attenuated LPS-induced lung cell injury
BEAS-2B cells and 16HBE cells were infected with 50 nM NEAT1 shRNA or its negative control and then exposed to LPS for 24 h to further clarify the potential roles of NEAT1 in ARDS. The transfection of sh-NEAT1 significantly reduced NEAT1 expression (Fig. 2A). Similarly, sh-NEAT1 markedly reduced the levels of TNF-α, IL-1β and IL-6 stimulated by LPS treatment (Fig. 2B). Furthermore, the CCK-8 assay results showed that inhibition of NEAT1 markedly relieved the suppression on cell proliferation induced by LPS treatment (Fig. 2C). Flow cytometry results revealed that downregulation of NEAT1 resulted in a lower proportion of cells in G1 phase and a higher proportion of cells that progressed to S phase compared to the LPS group (Fig. 2D). Consistent with these findings, western blot assay also showed that NEAT1 knockdown decreased the level of p21 but increased the levels of CDK2 and Cyclin D1 (Fig. 2E). Altogether, knockdown of NEAT1 significantly alleviated the inflammatory response and facilitated the G1-S phase transition in vitro.
16HBE cells and BEAS-2B cells were transfected with shNEAT1 or its negative control and then treated with LPS for 24 h. A NEAT1 expression was measured using qPCR. B The levels of TNF-α, IL-6 and IL-1β in the medium supernatants were measured using ELISA. C CCK8 assay was employed to detect cell proliferation. D The cell cycle phase was measured using flow cytometry. E The protein levels of p21, CDK2 and Cyclin D1 were detected using western blot. The results are shown as the means ± SD, and the differences were assessed using Student’s t test or one-way analysis of variance. *P < 0.05, **P < 0.01, and ***P < 0.001.
miR-27a was a downstream target of NEAT1
The TargetScan database was used to predict the binding sites between miR-27a and NEAT1 (Fig. 3A), and the targeting relationship was validated using the dual-luciferase reporter assay. The luciferase activity was significantly reduced in 16HBE cells co-transfected with WT-NEAT1 plasmids and miR-27a mimics but was markedly increased after the co-transfection of WT-NEAT1 plasmids and the miR-27a inhibitor. However, co-transfection of miR-27a mimics or the miR-27a inhibitor and MUT-NEAT1 plasmids in 16HBE cells resulted in a non-significant alteration of luciferase activity (Fig. 3B). Moreover, the expression level of miR-27a was markedly reduced by NEAT1 overexpression but significantly increased by NEAT1 downregulation (Fig. 3C, D). Next, we examined the expression of miR-27a in patients with ARDS using qPCR. The data showed that miR-27a was downregulated in patients with ARDS and inversely correlated with NEAT1 expression (Fig. 3E, F). All the aforementioned results indicated that NEAT1 inhibited miR-27a expression by directly targeting miR-27a.
A StarBase software was used to predict the potential binding sites between miR-27a and NEAT1. B Luc-WT-NEAT1 or Luc-MUT-NEAT1 plasmids were co-transfected with miR-27a mimics, miR-27a inhibitor or their negative controls in 16HBE cells, and luciferase activity was measured. C, D 16HBE and BEAS-2B cells were transfected with pcDNA-NC, pcDNA-NEAT1, sh-NC or sh-NEAT1 for 24 h, and the expression levels of NEAT1 and miR-27a were detected using qPCR. E Plasma samples were collected from patients with ARDS and controls, and the expression level of miR-27a was detected using qPCR. F Pearson correlation scatter plot showed that miR-27a expression was inversely correlated with NEAT1 expression. Data are presented as the means ± SD and were analysed using Student’s t test or one-way analysis of variance. *P < 0.05, **P < 0.01, and ***P < 0.001.
miR-27a reversed the effects of NEAT1 on the cell cycle and inflammation
We also explored whether miR-27a was involved in the regulation of NEAT1-mediated cell injury. 16HBE and BEAS-2B cells were transfected with sh-NEAT1 alone or co-transfected with sh-NEAT1 + miR-27a inhibitor and then exposed to LPS. As shown in Fig. 4A, miR-27a expression was significantly reduced after LPS treatment, but was restored by the transfection of sh-NEAT1. Moreover, miR-27a expression was further downregulated by the miR-27a inhibitor. Subsequently, the levels of IL-1β, IL-6 and TNF-1α were significantly increased in the sh-NEAT1 + miR-27a inhibitor group compared to the sh-NEAT1 group, suggesting that knockdown of miR-27a reversed the inhibitory effects of sh-NEAT1 on cellular inflammatory responses (Fig. 4B). Moreover, the effects of NEAT1 downregulation on cell proliferation, cell cycle progression and cell cycle-associated proteins were also reversed by the miR-27a inhibitor (Fig. 4C–E). In conclusion, all of the aforementioned data illustrated that miR-27a was a downstream effector of NEAT1 in vitro.
16HBE and BEAS-2B cells were transfected with sh-NEAT1 alone or co-transfected with sh-NEAT1 + miR-27a inhibitor for 24 h before treatment with LPS, and then cells and culture supernatants were collected. A The expression level of miR-27a was detected using qPCR. B The levels of TNF-α, IL-6 and IL-1β were measured ELISA. C CCK8 assay was employed to analyse cell proliferation. D The cell cycle phase was evaluated using flow cytometry. E The protein levels of PTEN, p21, CDK2 and Cyclin D1 were detected using western blot. Data are presented as the means ± SD and were analysed using Student’s t test or one-way analysis of variance. *P < 0.05, **P < 0.01, and ***P < 0.001.
PTEN functioned as an effector of miR-27a
PTEN is a key negative modulator of cell cycle progression. Hence, we first examined the expression level of PTEN in patients with ARDS and found that PTEN was expressed at high level in patients with ARDS compared with controls (Fig. 5A). Subsequently, the Pearson correlation coefficient revealed a negative correlation between PTEN and miR-27a expression at the clinical level (Fig. 5B). The potential binding sites between miR-27a and PTEN are shown in Fig. 5C. The luciferase activity was significantly reduced in 16HBE cells co-transfected with WT-PTEN plasmids and miR-27a mimics but was markedly increased after the co-transfection of WT-PTEN plasmids and miR-27a inhibitor. However, co-transfection with miR-27a mimics or miR-27a inhibitor and MUT-PTEN plasmids in 16HBE cells resulted in a non-significant alteration of luciferase activity (Fig. 5D). qPCR assay described that miR-27a overexpression induced by the transfection of miR-27a mimics markedly suppressed PTEN expression, while miR-27a downregulation induced by the transfection of the miR-27a inhibitor significantly increased PTEN expression (Fig. 5E, F). Then, we researched the effects of PTEN on the roles of miR-27a in regulating the levels of cell cycle-related proteins. The upregulation of miR-27a decreased the levels of PTEN and p21 but increased the expression levels of Cyclin D1 and CDK2, whereas overexpression of PTEN markedly reversed these effects (Fig. 5G). Based on these results, PTEN functioned as a downstream effector of miR-27a in the regulation of the cell cycle.
A Plasma samples were collected from patients with ARDS and controls, and the expression of PTEN was detected using qPCR. B Pearson correlation scatter plot shows that miR-27a expression was inversely correlated with PTEN expression. C The potential binding site between miR-27a and PTEN. D Luc-WT-PTEN or Luc-MUT-PTEN plasmids were co-transfected with miR-27a mimics, miR-27a inhibitor or their negative controls in 16HBE cells, and luciferase activity was measured. E, F 16HBE and BEAS-2B cells were transfected with miR-27a mimics, miR-27a inhibitor or their negative control for 24 h. The expression levels of miR-27a and PTEN were detected using qPCR. G 16HBE and BEAS-2B cells were transfected with miR-27a mimic alone or co-transfected with miR-27a mimics and sh-PTEN. The protein levels of PTEN, p21, CDK2 and Cyclin D1 were detected using western blot. Data are presented as the means ± SD and were analysed using Student’s t test or one-way analysis of variance. *P < 0.05, **P < 0.01, and ***P < 0.001.
PTEN participated in the regulation of NEAT1 during LPS-induced cell injury
The biological association between NEAT1 and PTEN was investigated. qPCR assay suggested that overexpression of NEAT1 upregulated PTEN expression, while the silencing of NEAT1 downregulated PTEN expression (Fig. 6A). Furthermore, the expression level of PTEN was positively correlated with NEAT1 expression in patients with ARDS (Fig. 6B). Collectively, NEAT1 positively regulates PTEN expression. Next, we investigated the effects of PTEN on NEAT1-mediated cell injury. The qPCR results showed that the pcDNA-PTEN vectors were successfully inserted into cells (Fig. 6C). Subsequently, the inhibition on the cell cycle and proliferation caused by NEAT1 upregulation was markedly reversed by silencing PTEN (Fig. 6D, E). A similar trend was observed that the increase of p21 level and the reduction of Cyclin D1 and CDK2 levels caused by NEAT1 overexpression were also blocked by PTEN silence (Fig. 6F). Taken together, upregulation of NEAT1 suppressed cell proliferation and cell cycle progression by modulating PTEN expression.
A 16HBE cells and BEAS-2B cells were transfected with pcDNA-NC, pcDNA-NEAT1, sh-NC or sh-NEAT1, and then the expression level of PTEN was detected using qPCR. B Pearson correlation scatter plot shows that NEAT1 expression was positively correlated with PTEN expression. C, D 16HBE and BEAS-2B cells were transfected with pcDNA-NEAT1 or co-transfected with pcDNA-NEAT1 and sh-PTEN for 24 h and then exposed to LPS. The expression of PTEN and NEAT1 was subsequently detected using qPCR (C). Cell proliferation was analysed using the CCK-8 assay (D). The cell cycle phase was evaluated using flow cytometry (E). The protein levels of p21, CDK2 and Cyclin D1 were detected using western blot (F). Data are presented as the means ± SD and were analysed using Student’s t test or one-way analysis of variance. *P < 0.05, **P < 0.01, and ***P < 0.001.
Knockdown of NEAT1 improved the survival and cell cycle progression and relieved inflammation in CLP mice model
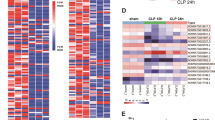
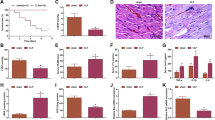
Septic mouse models were established to verify the biological roles of NEAT1 and PTEN in vivo. As disclosed in Fig. 7A–C, compared with the sham group, NEAT1 and PTEN expression were significantly increased in the CLP group, but miR-27a expression was decreased. However, knockdown of NEAT1 by sh-NEAT1 successfully reduced the expression of PTEN and restored the expression of miR-27a in CLP-induced sepsis models. Subsequently, NEAT1 silencing obviously increased the survival rate of mice (Fig. 7D). The levels of TNF-α, IL-6 and IL-1β were significantly increased in lung tissues from with CLP-induced septic mice, while these effects were markedly decreased by NEAT1 knockdown (Fig. 7E). Moreover, western blot assay also revealed that NEAT1 silencing reduced PTEN and p21 protein levels but increased CDK2 and cyclin D1 protein levels in CLP-induced septic mice (Fig. 7F). Thus, NEAT1 knockdown improved the survival rate of septic mice and relieved the inflammatory response and cell cycle arrest.
CLP surgery was performed on the fifth day after lentivirus injection, and lung samples were obtained immediately after the mice were euthanized. A–C The expression levels of NEAT1, miR-27a and PTEN in lung tissues from CLP-induced septic mice were analysed using qPCR. D The effect of NEAT1 knockdown on the survival rate of CLP-induced septic mice. E The levels of TNF-α, IL-6 and IL-1β in lung tissues of CLP-induced septic mice were detected using ELISA. F The protein levels of p21, CDK2 and Cyclin D1 in lung tissues from CLP-induced septic mice were detected using western blot. Data are presented as the means ± SD and were analysed using Student’s t test or one-way analysis of variance. *P < 0.05, **P < 0.01, and ***P < 0.001.
Discussion
Sepsis is a severe form of infection characterised by constant immune dysfunction with inflammatory response syndrome [26]. ARDS is a complication of sepsis that manifests as a disruption of the alveolar/endothelial barrier associated with massive pulmonary infiltration of neutrophils [27,28,29]. Currently, the lack of understanding of the pathophysiology, therapeutic strategy and drugs leads to the high mortality rate of patients with ARDS. Increasing evidences have disclosed that lncRNAs and miRNAs are associated with the occurrence and development of ARDS [30, 31]. In this study, we identified the abnormal expression of NEAT1 and miR-27a in ARDS and explored their roles and mechanism in vitro and in vivo. According to a recent study, NEAT1 is an essential regulator of the immune system [32]. For example, NEAT1 is upregulated in blood samples from patients with tuberculosis and is associated with Mtb infection [16]. Likewise, NEAT1 expression is also upregulated in patients with sepsis and is considered a potential molecular marker for disease diagnosis [33]. Recently, the functions and mechanism of NEAT1 in sepsis-induced diseases were investigated. For example, knockdown of NEAT1 in rat kidney cells inhibited LPS-induced cell damage by upregulating miR-204 [32, 34]. Consistent with these findings, we also certified that knockdown of NEAT1 alleviated lung cell damage by regulating inflammatory responses and the cell cycle in vitro. Moreover, NEAT1 knockdown improved the survival rate of CLP-induced septic mice. These results suggested the potential functions of NEAT1 in ARDS.
miR-27a plays an important role in regulating the immune response and is associated with sepsis progression [35,36,37]. Notably, the expression level and function of miR-27a are controversial in different septic models. On the one hand, a study showed higher expression of miR-27a in the lungs of septic mice, and overexpression of miR-27a promotes pulmonary inflammation and reduces the survival of septic mice [38]. On the other hand, several studies have reported that miR-27a expression is significantly decreased in the lung tissues of LPS-treated mice, and its upregulation plays a protective effect on acute lung injury by inhibiting the MyD88/NF-κB signalling pathway [37, 39]. Furthermore, Xie et al. [36] reported that miR-27a is downregulated in LPS-stimulated macrophages and suppresses TLR2- and TLR4-driven inflammatory responses. We speculated that the controversies may be associated with individual differences and the complexity of the pathological mechanisms of the diseases. In the present study, miR-27a was also downregulated in patients with ARDS and interacted with NEAT1 to negatively modulate the regulatory effects of NEAT1 on cell survival, cell cycle progression and inflammation in vitro. Based on these results, miR-27a may exert protective effects on sepsis-induced ARDS.
PTEN, a phosphatase that targets multiple proteins, functions as an essential regulator of senescence, quiescence, cell death and the cell cycle [40, 41]. PTEN restrains cell proliferation and survival by modulating the cell cycle, such as the G1/S and G2/M transitions [40]. Another study revealed that PTEN participates in the pathogenesis of ARDS by acting as a target of the MALAT1/miR-425 axis to promote cell apoptosis [30]. Additionally, PTEN also activates β-catenin and aggravates the HMGB1-mediated inflammatory response in acute lung injury [42]. Furthermore, PTEN facilitates LPS-induced pulmonary inflammation and injury through the AKT/NF-kappaB signalling pathway and sepsis-induced acute lung injury by inhibiting β-catenin pathway [43, 44]. These observations implied that PTEN may promote the progression of sepsis-induced ARDS. Consistently, our results identified that PTEN was expressed at high levels in patients with ARDS and was regulated by the NEAT1/miR-27a axis to block the G1/S transition of the cell cycle, reduce the levels of Cyclin D1 and CDK2 and increase the level of p21, indicating that PTEN is involved in the mechanism of action of NEAT1 in sepsis-induced ARDS.
In summary, our study clarified that NEAT1 was upregulated in sepsis-induced ARDS. Downregulation of NEAT1 improved inflammatory responses and cell cycle progression by modulating the miR-27a/PTEN axis (Fig. 8). Based on these results, we propose a new underlying mechanism of NEAT1 in ARDS, and our data may provide a theoretical reference for a deeper understanding of the pathological mechanism of ARDS.
Data availability
The datasets used or analysed during the current study are available from the corresponding author on reasonable request.
References
Candel FJ, Borges Sa,M, Belda S, Bou G, Del Pozo JL, Estrada O, et al. Current aspects in sepsis approach. Turning things around. Rev Esp Quimioter. 2018;31:298–315.
Schlichting D, McCollam JS. Recognizing and managing severe sepsis: a common and deadly threat. South Med J. 2007;100:594–600.
Yealy DM, Huang DT, Delaney A, Knight M, Randolph AG, Daniels R, et al. Recognizing and managing sepsis: what needs to be done? BMC Med. 2015;13:98.
Zhao H, Zhao M, Wang Y, Li F, Zhang Z. Glycyrrhizic acid prevents sepsis-induced acute lung injury and mortality in rats. J Histochem Cytochem. 2016;64:125–37.
Blumhagen RZ, Hedin BR, Malcolm KC, Burnham EL, Moss M, Abraham E, et al. Alternative pre-mRNA splicing of Toll-like receptor signaling components in peripheral blood mononuclear cells from patients with ARDS. Am J Physiol Lung Cell Mol Physiol. 2017;313:L930–l939.
Monaghan SF, Chung CS, Chen Y, Lomas-Neira J, Fairbrother WG, Heffernan DS, et al. Soluble programmed cell death receptor-1 (sPD-1): a potential biomarker with anti-inflammatory properties in human and experimental acute respiratory distress syndrome (ARDS). J Transl Med. 2016;14:312.
Liu X, Xiao ZD, Han L, Zhang J, Lee SW, Wang W, et al. LncRNA NBR2 engages a metabolic checkpoint by regulating AMPK under energy stress. Nat Cell Biol. 2016;18:431–42.
Chen Y, Lin Y, Bai Y, Cheng D, Bi Z. A long noncoding RNA (lncRNA)-associated competing endogenous RNA (ceRNA) network identifies eight lncRNA biomarkers in patients with osteoarthritis of the knee. Med Sci Monit. 2019;25:2058–65.
Wang P, Liu G, Xu W, Liu H, Bu Q, Sun D. Long noncoding RNA H19 inhibits cell viability, migration, and invasion via downregulation of IRS-1 in thyroid cancer cells. Technol Cancer Res Treat. 2017;16:1102–12.
Aune TM, Spurlock CF 3rd. Long non-coding RNAs in innate and adaptive immunity. Virus Res. 2016;212:146–60.
He F, Zhang C, Huang Q. Long noncoding RNA nuclear enriched abundant transcript 1/miRNA-124 axis correlates with increased disease risk, elevated inflammation, deteriorative disease condition, and predicts decreased survival of sepsis. Medicine. 2019;98:e16470.
Yong H, Wu G, Chen J, Liu X, Bai Y, Tang N, et al. lncRNA MALAT1 accelerates skeletal muscle cell apoptosis and inflammatory response in sepsis by decreasing BRCA1 expression by recruiting EZH2. Mol Ther Nucleic Acids. 2020;19:97–108.
Klec C, Prinz F, Pichler M. Involvement of the long noncoding RNA NEAT1 in carcinogenesis. Mol Oncol. 2019;13:46–60.
Zhang CC, Niu F. LncRNA NEAT1 promotes inflammatory response in sepsis-induced liver injury via the Let-7a/TLR4 axis. Int Immunopharmacol. 2019;75:105731.
Wang SM, Liu GQ, Xian HB, Si JL, Qi SX, Yu YP. LncRNA NEAT1 alleviates sepsis-induced myocardial injury by regulating the TLR2/NF-kappaB signaling pathway. Eur Rev Med Pharmacol Sci. 2019;23:4898–907.
Huang S, Huang Z, Luo Q, Qing C. The expression of lncRNA NEAT1 in human tuberculosis and its antituberculosis effect. Biomed Res Int. 2018;2018:9529072.
Shen Y, Yu J, Jing Y, Zhang J. MiR-106a aggravates sepsis-induced acute kidney injury by targeting THBS2 in mice model. Acta Cir Bras. 2019;34:e201900602.
Ling L, Lu HT, Wang HF, Shen MJ, Zhang HB. MicroRNA-203 acts as a potent suppressor in septic shock by alleviating lung injury via inhibition of VNN1. Kidney Blood Press Res. 2019;44:565–82.
Lin Q, Gao Z, Alarcon RM, Ye J, Yun Z. A role of miR-27 in the regulation of adipogenesis. FEBS J. 2009;276:2348–58.
Xie W, Li L, Zhang M, Cheng HP, Gong D, Lv YC, et al. MicroRNA-27 prevents atherosclerosis by suppressing lipoprotein lipase-induced lipid accumulation and inflammatory response in apolipoprotein E knockout mice. PLoS ONE. 2016;11:e0157085.
Zhang M, Wu JF, Chen WJ, Tang SL, Mo ZC, Tang YY, et al. MicroRNA-27a/b regulates cellular cholesterol efflux, influx and esterification/hydrolysis in THP-1 macrophages. Atherosclerosis. 2014;234:54–64.
Cheng Y, Kuang W, Hao Y, Zhang D, Lei M, Du L, et al. Downregulation of miR-27a* and miR-532-5p and upregulation of miR-146a and miR-155 in LPS-induced RAW264.7 macrophage cells. Inflammation. 2012;35:1308–13.
Yang Q, Zhang D, Li Y, Li Y, Li Y. Paclitaxel alleviated liver injury of septic mice by alleviating inflammatory response via microRNA-27a/TAB3/NF-kappaB signaling pathway. Biomed Pharmacother. 2018;97:1424–33.
Vasilescu C, Dragomir M, Tanase M, Giza D, Purnichescu-Purtan R, Chen M, et al. Circulating miRNAs in sepsis-A network under attack: an in-silico prediction of the potential existence of miRNA sponges in sepsis. PLoS ONE. 2017;12:e0183334.
Lee S, Nakahira K, Dalli J, Siempos II, Norris PC, Colas RA, et al. NLRP3 inflammasome deficiency protects against microbial sepsis via increased lipoxin B4 synthesis. Am J Respir Crit Care Med. 2017;196:713–26.
Mohnle P, Hirschberger S, Hinske LC, Briegel J, Hubner M, Weis S, et al. MicroRNAs 143 and 150 in whole blood enable detection of T-cell immunoparalysis in sepsis. Mol Med. 2018;24:54.
Neudecker, V, Brodsky, KS, Clambey, ET, Schmidt, EP, Packard, TA, Davenport, B et al. Neutrophil transfer of miR-223 to lung epithelial cells dampens acute lung injury in mice. Sci Transl Med. 9, eaah5360 (2017).
Bao, X, Zhang, Q, Liu, N, Zhuang, S, Li, Z, Meng, Q et al. Characteristics of circular RNA expression of pulmonary macrophages in mice with sepsis-induced acute lung injury. J Cell Mol Med https://doi.org/10.1111/jcmm.14577 (2019).
Liu W, Liu K, Zhang S, Shan L, Tang J. Tetramethylpyrazine showed therapeutic effects on sepsis-induced acute lung injury in rats by inhibiting endoplasmic reticulum stress protein kinase RNA-like endoplasmic reticulum kinase (PERK) signaling-induced apoptosis of pulmonary microvascular endothelial cells. Med Sci Monit. 2018;24:1225–31.
Wang L, Liu J, Xie W, Li G, Yao L, Zhang R, et al. Overexpression of MALAT1 relates to lung injury through sponging miR-425 and promoting cell apoptosis during ARDS. Can Respir J. 2019;2019:1871394.
Wang Y, Fu X, Yu B, Ai F. Long non-coding RNA THRIL predicts increased acute respiratory distress syndrome risk and positively correlates with disease severity, inflammation, and mortality in sepsis patients. J Clin Lab Anal. 2019;33:e22882.
Chen Y, Qiu J, Chen B, Lin Y, Chen Y, Xie G, et al. Long non-coding RNA NEAT1 plays an important role in sepsis-induced acute kidney injury by targeting miR-204 and modulating the NF-kappaB pathway. Int Immunopharmacol. 2018;59:252–60.
Huang S, Qian K, Zhu Y, Huang Z, Luo Q, Qing C. Diagnostic value of the lncRNA NEAT1 in peripheral blood mononuclear cells of patients with sepsis. Dis Markers. 2017;2017:7962836.
Prinz, F, Kapeller, A, Pichler, M, & Klec, C. The implications of the long non-coding RNA NEAT1 in non-cancerous diseases. Int J Mol Sci. 20, 627 (2019).
Li W, Qiu X, Liu J, Han Y, Wei D, Ji G, et al. miR-27a protects against acute lung injury in LPS-treated mice by inhibiting NF-kappaB-mediated inflammatory response. Int J Clin Exp Pathol. 2018;11:2980–9.
Xie N, Cui H, Banerjee S, Tan Z, Salomao R, Fu M, et al. miR-27a regulates inflammatory response of macrophages by targeting IL-10. J Immunol. 2014;193:327–34.
Ju M, Liu B, He H, Gu Z, Liu Y, Su Y, et al. MicroRNA-27a alleviates LPS-induced acute lung injury in mice via inhibiting in fl ammation and apoptosis through modulating TLR4/MyD88/NF-kappaB pathway. Cell Cycle. 2018;17:2001–18.
Wang Z, Ruan Z, Mao Y, Dong W, Zhang Y, Yin N, et al. miR-27a is up regulated and promotes inflammatory response in sepsis. Cell Immunol. 2014;290:190–5.
Wang Y, Zhang X, Tian J, Liu G, Li X, Shen D. Sevoflurane alleviates LPSinduced acute lung injury via the microRNA27a3p/TLR4/MyD88/NFkappaB signaling pathway. Int J Mol Med. 2019;44:479–90.
Brandmaier A, Hou SQ, Shen WH. Cell cycle control by PTEN. J Mol Biol. 2017;429:2265–77.
Sachdeva K, Do DC, Zhang Y, Hu X, Chen J, Gao P. Environmental exposures and asthma development: autophagy, mitophagy, and cellular senescence. Front Immunol. 2019;10:2787.
Zhou M, Fang H, Du M, Li C, Tang R, Liu H, et al. The modulation of regulatory T cells via HMGB1/PTEN/β-catenin axis in LPS induced acute lung injury. Front Immunol. 2019;10:1612.
Wu J, Sun Z, Sun H, Li Y. MicroRNA27a promotes tumorigenesis via targeting AKT in triple negative breast cancer. Mol Med Rep. 2018;17:562–70.
Liu, L, Chen, J, Zhang, X, Cui, X, Qiao, N, Zhang, Y et al. The protective effect of PPARgamma in sepsis-induced acute lung injury via inhibiting PTEN/beta-catenin pathway. Biosci Rep. 40, BSR20192639 (2020).
Funding
This work is supported by special funds for basic scientific research business expenses of the central public welfare research institutes of the Chinese Academy of Medical Sciences (No. 2019PT32000), Doctor Foundation of Guizhou Provincial People’s Hospital (GZSYBS [2019]04).
Author information
Authors and Affiliations
Contributions
XL: study concepts, definition of intellectual content and statistical analysis; XYZ: study concepts, manuscript preparation and editing; YJN: data acquisition and definition of intellectual content; GHL and QZ: literature research and experimental studies; XF: data analysis and statistical analysis; SY: data analysis; QHZ: study design, manuscript preparation and editing and manuscript review; JQL: guarantor of integrity of the entire study, study concepts and design and manuscript review. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
All protocol was approved by the Ethics Committee of Guizhou Provincial People’s Hospital and conducted according to the Declaration of Helsinki.
Informed consent
All patients were provided with informed consent.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Lv, X., Zhang, XY., Zhang, Q. et al. lncRNA NEAT1 aggravates sepsis-induced lung injury by regulating the miR-27a/PTEN axis. Lab Invest 101, 1371–1381 (2021). https://doi.org/10.1038/s41374-021-00620-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41374-021-00620-7
This article is cited by
-
Signaling pathways and potential therapeutic targets in acute respiratory distress syndrome (ARDS)
Respiratory Research (2024)
-
Role of ATF3 triggering M2 macrophage polarization to protect against the inflammatory injury of sepsis through ILF3/NEAT1 axis
Molecular Medicine (2024)
-
Programmed cell death 4 governs NLRP3-mediated pyroptosis in septic lung disorders
Molecular Biology Reports (2024)