Abstract
A diabetic foot ulcer (DFU) is one of the most devastating complications of diabetes. It has been reported that lncRNA GAS5 plays a vital role in wound healing in DFUs. However, the specific mechanism remains unclear. In this research, we aimed to investigate the role of GAS5 in wound healing in DFUs as well as the underlying mechanism. qPCR or western blotting was performed to measure the expression levels of GAS5, HIF1A, VEGF and TAF15. CCK-8 or EdU assays, flow cytometry, wound healing assays and tube formation assays were carried out to assess the proliferation, apoptosis, wound healing and in vitro angiogenesis of HUVECs, respectively. RNA pull-down and RIP assays were performed to verify the interaction between GAS5 and TAF15. ChIP and luciferase assays were conducted to verify the binding of TAF15 to the HIF1A promoter. In the DFU mouse model, H&E and Masson staining were used to determine epidermal and dermal thickness and collagen formation. GAS5 and HIF1A were downregulated in the skin tissues of DFU patients, and GAS5 overexpression promoted cell proliferation, wound healing and tubule formation in HG-treated HUVECs. In addition, GAS5 facilitated HIF1A expression by interacting with TAF15. Rescue assays demonstrated that the suppression of HIF1A/VEGF pathway activation partially reversed the functional roles of GAS5 in HUVECs. Furthermore, GAS5 accelerated wound healing by activating the HIF1A/VEGF pathway in mice with DFUs. GAS5 activates the HIF1A/VEGF pathway by binding to TAF15, resulting in accelerated wound healing in DFUs. Our findings may provide a theoretical basis for the clinical treatment of DFUs.
Similar content being viewed by others
Introduction
Diabetic foot ulcers (DFUs) are a major cause of amputation in diabetic patients; DFUs affect over 170 million people worldwide, and 10–15% of people with diabetes develop foot ulcers [1]. In addition, each year, ~20% of DFU patients require amputation surgery to prevent the disease from worsening [2]. DFUs seriously affect human health and reduce people’s quality of life. Moreover, studies have shown that DFU treatment is difficult due to the difficulty of wound repair [3]. In addition, angiogenesis is reported to be an important factor for wound healing [4]. Therefore, understanding the mechanism underlying wound healing in DFUs and seeking effective ways to promote angiogenesis will help to identify effective methods for DFU treatment.
Long noncoding RNAs (lncRNAs) are vital intracellular regulatory molecules that play functional roles in various physiological processes [5]. Importantly, lncRNAs have been proven to be regulators of wound healing [6]. LncRNA H19 in MSC-derived exosomes upregulates PTEN by regulating miR-152-3p, resulting in accelerated wound healing in DFUs [7]. Liu et al. reported that lncRNA MALAT1 accelerates wound healing in diabetic mice by regulating the hypoxia-inducible factor (HIF)-1A pathway [8]. LncRNA growth arrest-specific 5 (GAS5) is a non-protein-coding RNA that is encoded by the GAS5 gene. Recently, GAS5 was demonstrated to be related to the prevalence of diabetes [9] and serves as a target for therapeutic intervention in type 2 diabetes management [10]. It is worth noting that GAS5 is also involved in regulating DFU wound healing [11]. However, the role of GAS5 in wound healing in DFUs, as well as the underlying mechanism, remains unclear.
TATA box-binding protein associated factor 15 (TAF15) is a member of the FET family and plays an important role in regulating mRNA transcription, RNA splicing and protein translation [12]. Moreover, TAF15 was reported to promote cell proliferation and modulate cell cycle-related gene expression [13, 14]. It is generally known that lncRNAs can interact with RNA-binding proteins (RBPs) to regulate their target genes. Chen et al. reported that upregulated LINC01048 promotes tumour cell proliferation by interacting with TAF15 to activate YAP1 [15]. However, limited information is available regarding the function of TAF15 in DFUs and its interaction with GAS5.
HIF proteins are composed of HIF1A, 2A, 3A and the constitutively expressed subunit 1B. As one of the most widely studied HIF proteins, HIF1A is known to activate the expression of many hypoxia-inducible genes, which may be essential for regulating various cell functions [16]. In addition, it has been reported that HIF1A plays a pivotal role in endothelial cell tube formation, and the specific mechanism may be related to the upregulation of vascular endothelial growth factor (VEGF) [17].
In the present study, we hypothesize that GAS5 may activate the HIF1A/VEGF pathway by binding to TAF15 to accelerate wound healing in DFUs in vitro and in vivo. Our research may shed new light on the clinical treatment of DFUs.
Materials and methods
Human skin tissue specimens
Skin tissues from the edges of wounds were obtained from DFU patients and healthy controls (characteristics are listed in Table 1) in our hospital between August 2018 and March 2019. DFU patients were included if they were diagnosed with type 2 diabetes according to the World Health Organization (WHO) diagnostic criteria for DM (WHO 1999), signed written informed consent forms, had no haematological diseases, had no malignant tumours and had normal liver and kidney function. Patients were excluded if they had acute complications, such as diabetic ketoacidosis or hypertonic coma, concurrent primary glomerular disease, liver or kidney dysfunction, cardiac insufficiency or other serious diseases. The experimental protocols were approved by the Ethics Committee of Yiyang Central Hospital, and the experimental procedures were conducted according to the principles outlined in the Declaration of Helsinki.
Cell culture and treatments
Human umbilical vein endothelial cells (HUVECs) were obtained from ScienCell Research Laboratories and were cultured in endothelial cell medium supplemented with 1% penicillin–streptomycin solution and 5% foetal bovine serum in a humidified incubator containing 5% CO2 at 37 °C. For the treatments, HUVECs were incubated with normal glucose (5.5 mM), high glucose (HG; 25 mM) or a high concentration of mannose (OS; 5.5 mM glucose and 19.5 mM mannose) for 48 h. Mannose and glucose were purchased from Sigma-Aldrich (St. Louis, MO, USA).
Cell transfection
For the overexpression of GAS5 and TAF15, GAS5 or TAF15 cDNA was cloned into the pcDNA3.1 vector (Invitrogen, Carlsbad, CA, USA). HIF1A shRNA (sh-HIF1A) and the negative control were purchased from RiboBio (Guangzhou, China). Cells were seeded into plates, and Lipofectamine™ 3000 Transfection Reagent (Invitrogen) was used for cell transfection. The HUVECs were subjected to subsequent experiments 48 h after transfection.
CCK-8 assay
Cell viability was analysed by Cell Counting Kit-8 (Beyotime, Nanjing, China) according to the instructions. After treatment, 10 μL CCK-8 solution was added to each well and incubated for 4 h. Then, a microplate reader was used to detect the absorbance of each well at 450 nm, and the absorbance values indicated cell viability.
5-Ethynyl-2′-deoxyuridine (EdU) assay
An EdU assay was performed to assess cell proliferation by using an EdU Imaging Kit (RiboBio). First, the transfected HUVECs were incubated with prepared EdU medium for 2 h. Subsequently, the cells were fixed with 4% paraformaldehyde for 30 min, and other reagents were added and incubated according to the instructions. After washing, the cells were stained with Hoechst 33342 solution for 30 min at 37 °C in the dark. Finally, images were obtained with a fluorescence microscope, and ImageJ software (ImageJ Software, Inc.) was used for statistical analysis. The experiments were repeated three times.
Flow cytometry
An Annexin V-FITC/PI Apoptosis Detection Kit (Beyotime) was used for apoptosis analysis. In detail, the cells were resuspended in 500 μL binding buffer, and then, 5 μL FITC-conjugated antibodies and 5 μL propidium iodide were added to the cell suspension and incubated for 15 min in the dark. Finally, cell apoptosis was assessed by using a flow cytometer (FACSort; Becton Dickinson). The data were analysed using ModFit software (Verity Software House, Top-sham, USA).
Wound healing assay
HUVECs (2 × 105) were added to 12-well cell culture plates and treated with high concentrations of glucose or mannose and the indicated plasmids. After treatment, the cell monolayer was scratched with a 200 μL pipette tip, forming an artificial wound. Then, the cells in the cell culture plates were photographed (from 0 to 24 h) under an inverted microscope to assess the wound healing of the HUVECs.
Tube formation assay
After treatment, 5 × 104 cells per well were seeded in Matrigel-pretreated 24-well plates and incubated for 48 h. Then, the formation of tubules was observed under an optical microscope. Finally, CellSens Standard software (Bio-Rad Laboratories, Hercules, CA, USA) was used to measure the numbers of tubes per field in each group.
Quantitative real-time PCR (qPCR)
TRIzol reagent (Takara) was used to extract the total RNA from HUVECs or tissues. Then, the RNA concentrations were measured, and the RNA samples were reverse transcribed into cDNA. qPCR was performed using SYBR GreenMix (Takara) on a real-time fluorescent qPCR instrument as previously described [18]. The relative RNA expression was calculated with the 2−ΔΔCt method and normalized to the GAPDH RNA expression. The primers used to this study are listed in Table 2.
Western blot
Total proteins were isolated from HUVECs or tissues by using cell lysis buffer (Beyotime). Western blotting was performed as previously described [19]. All the antibodies used in this study were obtained from Abcam (Cambridge, UK; 1:1000). A goat anti-rabbit IgG secondary antibody served as the secondary antibody (Santa Cruz, San Francisco, USA, 1:2000). The optical densities of the protein bands were quantified by ImageJ software.
Fluorescence in situ hybridization (FISH) assay
First, the prepared HUVECs were incubated with acidic sodium sulfite for 30 min and then incubated with pepsin for 20 min. The slides were soaked in 50, 80 and 96% ethanol solutions for 3 min before being dried. Subsequently, 10 μL of hybridization buffer and 1 μL of GAS5 probe (Thermo Fisher Scientific, Waltham, MA, USA) were added to the slide hybridization zone and incubated for 90 min at 4 °C. Furthermore, the slides were incubated in NP40 solution, washed with 70% ethanol for 4 min, and dried in the dark. Ten microlitres of DAPI dye (Sigma-Aldrich) was added to the slides and incubated in the dark for 5 min. Finally, the results were observed under a fluorescence microscope.
RNA pull-down assay
Biotin-RNA Labeling Mix was used for RNA biotinylation and transcription. Then, biotin-labelled RNAs were incubated with RNase-free TURBO DNase I (Invitrogen) and Sephadex G-50 Quick Spin Columns (Sigma-Aldrich) and then with cell lysate supernatants for 2 h at 4 °C. Subsequently, the supernatants were mixed with Dynabeads™ MyOne™ Streptavidin T1 (Invitrogen) and incubated for 1 h. After washing, the proteins bound to GAS5 were detected by western blot.
RNA immunoprecipitation (RIP) assay
The physical interaction between GAS5 and TAF15 was assessed using an EZMagna RIP Kit (Millipore, Billerica, MA, USA) according to the manufacturer’s instructions. The detailed procedure was similar to a previously described procedure [20].
Luciferase assay and chromatin immunoprecipitation (ChIP) assay
pcDNA3.1-NC or pcDNA3.1-TAF15 was cloned into the psi-CHECK2 reporter vector. Lipofectamine 3000 (Invitrogen) was used for cotransfection of the HIF1A promoter (>hg38_knownGene_ENST00000337138.9 range = chr14: 61694513-61695512). Forty-eight hours after transfection, the luciferase activity was measured with the Dual-Luciferase Reporter Assay System (Promega, Madison, WI, USA). To verify the binding of TAF15 to the promoter region of HIF1A, a ChIP assay was performed using an EZMagna ChIP kit (Millipore) according to the manufacturer’s instructions. Finally, the precipitated DNA was assessed via qPCR.
DFU model
The mice (8–10 weeks old) used in this study were purchased from Hunan SLAC Jingda Laboratory Animal Co., Ltd. (Changsha, China). The animal experiments described in this paper were carried out in accordance with the Guidelines for the Care and Use of Laboratory Animals issued by the National Institutes of Health.
In this study, diabetes was induced by the intraperitoneal administration of streptozotocin (STZ, 45 mg/kg body weight in 0.1 M citrate buffer, pH 4.5) for five consecutive days to 24 healthy mice as previously described [21]. The serum glucose level was analysed 5 days after the last STZ treatment and was then closely monitored for 2 weeks. Animals with fasting blood glucose levels higher than 280 mg/dL were considered diabetic, and wounds were then induced. To this end, the diabetic mice were randomly divided into three groups, each with eight mice, and a rectangular wound (3 mm × 4 mm) was made on the dorsal surface of the foot of each mouse by using ophthalmic scissors. The groups were treated as follows: NC group: pcDNA3.1-NC (500 μL of 1 nM plasmid in PBS) was intradermally injected at the wound edges; GAS5 group: pcDNA3.1-GAS5 (500 μL of 1 nM plasmid in PBS) was injected; GAS5 + sh-HIF1A group: pcDNA3.1-GAS5 and sh-HIF1A plasmids (500 μL of 1 nM plasmid in PBS) were injected. Images of the wounds were captured on days 0, 8 and 16, and the wound healing rates and times were calculated with ImageJ software. On the 16th day after injury, wound samples were collected for histological analysis, gene and protein expression measurement and collagen deposition assessment.
Mouse crawling assessment
On day 8 after the operation, the crawling abilities of the mice with DFUs were assessed via the flexibility score. In detail, the mice were placed on a wooden board with a width of 1 m and allowed to crawl and move freely for 5 min. The movement of the wounded foot was scored. The flexibility scores were defined as follows: 0 = unable to crawl; 1 = serious difficulty in crawling; 2 = moderate difficulty in crawling; 3 = slight difficulty in crawling and 4 = normal crawling. The mice were scored by a researcher who was blinded to the treatment conditions.
Histological analysis of the skin
The epidermal and dermal thicknesses of mouse skin tissues were assessed by haematoxylin and eosin (H&E) staining. In short, embedded skin tissues were stained with haematoxylin for 10 min and then with eosin for 5 min. Masson staining was performed for collagen quantification with a Masson assay kit (Bogoo Biotechnology, Shanghai, China). In detail, the quantitative analysis of the Masson staining-positive areas was performed using ImageJ, and the average percentage of the stained areas was calculated.
Capillary density in the wounds was observed by immunohistochemistry using primary CD31 antibodies (Abcam; 1:1000) followed by staining with DAB (Beyotime). The slices were imaged by an optical microscope, and the images were analysed via ImageJ software.
Statistical analysis
All the data are presented as the mean ± SD of three independent experiments and were statistically analysed by GraphPad Prism version 8.0 software (GraphPad Software, Inc.). Student’s t test (two groups) or one-way ANOVA with Tukey’s post hoc test (multiple groups) was performed to evaluate significant differences between groups. A P value < 0.05 was considered to be significantly different.
Results
GAS5 and HIF1A are downregulated in the skin tissues of DFU patients
It has been reported that GAS5 expression is tissue and cell type specific and related to the development of DFUs [11]. To investigate GAS5 expression in the skin tissues of DFU patients, qPCR and western blotting were performed to assess the levels of GAS5 and HIF1A. The results indicated that GAS5 and HIF1A were significantly downregulated in the skin tissues of DFU patients compared with the skin tissues of control subjects (Figs. 1A, B). Furthermore, the correlation analysis results showed that HIF1A expression was positively correlated with GAS5 expression in the skin tissues of DFU patients (Fig. 1C). Collectively, GAS5 and HIF1A were expressed at low levels in DFUs, providing evidence that GAS5 may play a key role in DFU progression by modulating the HIF1A signalling pathway.
HG inhibits HUVEC proliferation, wound healing and tubule formation
Next, HUVECs were treated with HG to establish an in vitro DFU model. First, qPCR was used to assess GAS5 expression in the HG-induced environment. The findings showed that compared with the control, OS (5.5 mM glucose and 19.5 mM mannose) had no effect on the GAS5 levels in HUVECs, while HG (25 mM) significantly inhibited GAS5 expression (Fig. 2A). Furthermore, compared with the control or OS, HG markedly inhibited the cell viability, wound healing and tubule formation of HUVECs (Figs. 2B, D, E). Conversely, flow cytometry analysis showed that compared with the control or OS, HG significantly accelerated HUVEC apoptosis (Fig. 2C). Taken together, these results suggest that HG treatment suppressed HUVEC proliferation, wound healing and tubule formation.
A GAS5 expression was measured via qPCR assay. B A CCK-8 assay was carried out to assess cell viability. C Flow cytometry was conducted to evaluate cell apoptosis. D A wound healing assay was performed to assess wound healing. E A tube formation assay was carried out to evaluate the angiogenic capacity of HUVECs. **P < 0.01, ***P < 0.001 vs. the control group.
GAS5 overexpression promotes HG-induced HUVEC proliferation, wound healing and tubule formation
To further investigate the effect of GAS5 on HUVEC bioactivity, pcDNA3.1-GAS5 was transfected into HUVECs to overexpress GAS5. pcDNA3.1-NC served as the negative control. The overexpression efficiency of GAS5 was assessed by qPCR (Fig. 3A). Furthermore, we found that GAS5 overexpression facilitated cell proliferation, wound healing and tube formation in HG-induced HUVECs (Figs. 3B, D, E). However, the HUVEC apoptosis induced by HG was inhibited by GAS5 overexpression (Fig. 3C). These findings indicated that GAS5 overexpression reduced the effects of HG on HUVEC bioactivity. In addition, GAS5 overexpression clearly increased the levels of HIF1A and VEGF in HG-treated HUVECs (Fig. 3F), indicating that GAS5 may induce the expression of HIF1A and VEGF to accelerate wound healing. Based on the above results, we showed that GAS5 overexpression promotes cell proliferation, wound healing and tubule formation in HG-treated HUVECs.
A qPCR was performed to detect the levels of GAS5. B Cell proliferation was assessed by EdU staining. C Flow cytometry was conducted to evaluate cell apoptosis. D A wound healing assay was performed to assess wound healing. E A tube formation assay was carried out to evaluate the angiogenic capacity of HUVECs. F qPCR and western blotting were performed to measure the levels of HIF1A and VEGF. **P < 0.01, ***P < 0.001 vs. the control group.
GAS5 interacts with TAF15 and promotes TAF15 expression
Then, we examined whether lncRNA GAS5 regulated wound healing by modulating target genes via interaction with RBPs. First, a FISH assay was conducted to detect the location of GAS5 in HUVECs. The results showed that GAS5 was distributed in both the nucleus and cytoplasm (Fig. 4A). Subsequently, we synthesized biotin-labelled sense and antisense GAS5 RNAs via in vitro transcription (Fig. 4B). The interaction between GAS5 and TAF15 was confirmed by RNA pull-down assay (Fig. 4C). Considering that TAF15 is a transcription factor and RBP, we performed an RIP assay to further demonstrate the specific interaction between GAS5 and TAF15 (Fig. 4D). Moreover, both the RNA and protein levels of TAF15 were upregulated by GAS5 overexpression (Fig. 4E). Together, these results suggest that GAS5 could interact with TAF15 and promote its expression in vitro.
A FISH assay was performed to observe the distribution of GAS5 in HUVECs. B Biotin-labelled sense and antisense GAS5 RNAs were synthesized by in vitro transcription. C, D RNA pull-down and RIP assays were used to verify the interaction between GAS5 and TAF15, and SNRNP70 served as the positive control. E qPCR and western blotting were conducted to measure TAF15 expression. **P < 0.01, ***P < 0.001 vs. the control group.
GAS5 accelerates HIF1A expression by interacting with TAF15
Studies have shown that hypoxia is related to angiogenesis, fibrosis and cell proliferation via the regulation of HIF1A [22]. To further elucidate the interaction among GAS5, HIF1A and TAF15, qPCR was performed to determine the levels of TAF15 in the skin tissues of DFU patients. The results showed that TAF15 was significantly downregulated in the skin tissues of DFU patients compared with the skin tissues of control subjects (Fig. 5A). In addition, the levels of TAF15 and HIF1A were significantly downregulated in HG-treated HUVECs compared with the control- or OS-treated HUVECs (Fig. 5B). Furthermore, TAF15 positively regulated the expression level of HIF1A in HUVECs (Fig. 5C). Mechanistically, ChIP and luciferase analyses indicated that TAF15 could bind to the HIF1A promoter (Figs. 5D, E). These in vitro assays suggested that GAS5 may activate HIF1A transcription by recruiting TAF15 to the HIF1A promoter.
A The levels of TAF15 in the skin tissues of DFU patients. B Measurement of the TAF15 and HIF1A expression levels in HG-treated HUVECs. C The effect of TAF15 on the expression level of HIF1A. D, E ChIP and luciferase assays were conducted to verify the interaction between TAF15 and the HIF1A promoter. Ago2 served as the negative control. **P < 0.01, ***P < 0.001 vs. the control group.
GAS5 promotes cell proliferation, wound healing and tubule formation via the HIF1A/VEGF pathway in HUVECs treated with HG
VEGF acts as an inducer of angiogenesis, stimulating the formation of new blood vessels. To investigate the specific mechanism by which VEGF promotes the tubule formation of HUVECs, pcDNA3.1-GAS5 or sh-HIF1A was transfected into HUVECs to overexpress GAS5 or knock down HIF1A. qPCR and western blotting were performed to assess the levels of HIF1A and VEGF, and the results showed that knockdown of HIF1A significantly inhibited VEGF expression, while overexpression of GAS5 markedly increased HIF1A and VEGF expression, and this effect was partially reversed by HIF1A inhibition (Fig. 6A). In addition, HIF1A suppression markedly inhibited the proliferation, wound healing and tubule formation of HUVECs, whereas GAS5 overexpression significantly promoted these cellular functions, and this effect was partly reversed by HIF1A knockdown (Figs. 6B, D, E). In contrast, flow cytometry analysis showed that HIF1A downregulation significantly promoted HUVEC apoptosis, and GAS5 overexpression inhibited HUVEC apoptosis, and this effect was reduced by HIF1A suppression (Fig. 6C). Collectively, GAS5 may promote HUVEC proliferation, wound healing and tubule formation by activating the HIF1A/VEGF pathway.
A Expression levels of HIF1A and VEGF were measured via qPCR and western blotting. B EdU assay was used to assess cell proliferation. C Flow cytometry was carried out to evaluate cell apoptosis. D A wound healing assay was conducted to assess the effect of GAS5/HIF1A on wound healing. E A tube formation assay was performed to evaluate the angiogenic capacity of HUVECs. *P < 0.05, **P < 0.01, ***P < 0.001 vs. the control group.
GAS5 accelerates DFU wound healing by activating the HIF1A/VEGF pathway
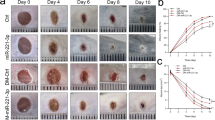
To further verify the above conclusions in vivo, we injected the relevant plasmids into the edges of the wounds of mice with DFUs. First, the expression of GAS5 and HIF1A was assessed, and the results showed that GAS5 overexpression notably increased the GAS5 levels, while sh-HIF1A markedly decreased the HIF1A levels (Fig. 7A). Then, the crawling abilities of the mice were observed on day 8 after the operation. GAS5 improved the crawling abilities of the mice, which was partially reversed by the suppression of HIF1A (Fig. 7B). qPCR and western blotting were used to measure the levels of HIF1A and VEGF in the skin tissues of the mice with DFUs, and we found that GAS5 overexpression significantly upregulated HIF1A and VEGF expression, but this effect was inhibited by HIF1A suppression (Fig. 7C). Subsequently, statistical analysis of wound healing in the mice with DFUs showed that GAS5 overexpression accelerated wound healing, and this effect was inhibited by HIF1A silencing (Figs. 7D, E). In addition, the capillary density in the GAS5-treated wounds was higher than that in the NC-treated wounds, and silencing of HIF1A blocked the effect of GAS5 on newly formed vessels (Fig. 7F). Moreover, the H&E staining results showed that GAS5 overexpression significantly enhanced epithelial migration and epidermal and dermal thickness, but these effects were inhibited by HIF1A knockdown. Masson staining analysis indicated that GAS5 overexpression markedly promoted collagen fibre formation, but this effect was inhibited by HIF1A silencing (Fig. 7G). Taken together, these results suggest that GAS5 may accelerate wound healing in DFUs by activating the HIF1A/VEGF pathway.
A The expression of GAS5 and HIF1A was assessed via qPCR. B The crawling abilities of mice were observed on day 8 after the operation. C qPCR and western blotting were performed to measure HIF1A and VEGF expression in the skin wounds of mice. D, E Statistical analysis of skin wound healing degree and time in mice with DFUs. F Capillary density in wounds was detected by immunohistochemistry staining. G H&E and Masson staining were conducted to evaluate epithelial cell migration, dermis and epidermis thickness and collagen fibre formation. *P < 0.05, **P < 0.01, ***P < 0.001 vs. the control group.
Discussion
DFUs are a serious chronic disease caused by diabetic complications, and ~15% of patients with diabetes suffer from foot ulcers [1]. The difficulty in wound healing is the major problem in DFU treatment [23]. Wound healing is a complex and long-term process that involves cell proliferation, collagen fibre formation and epidermal tissue formation [24]. It was reported that GAS5 and TAF15 are involved in the regulation of cell proliferation, and the HIF1A/VEGF pathway is related to angiogenesis in the process of wound healing [25, 26]. Our findings indicated that GAS5 could activate the HIF1A/VEGF pathway by binding to TAF15, resulting in accelerated skin wound healing in DFUs; this finding provides a theoretical basis for the clinical treatment of DFUs.
Diabetes is an autoimmune disease that is characterized by the autoimmune destruction of β-cells, leading to insulin deficiency; the continuous inflammatory response in diabetes further leads to DFUs [27]. In the whole process of DFU development, the dysregulation of some functional genes promotes disease progression [28]. Our results indicated that the levels of GAS5 and HIF1A were downregulated in the skin tissues of DFU patients. In addition, the slow growth of skin cells may also be one of the reasons why wounds are difficult to heal in patients with DFUs [29]. Functional assays showed that HG inhibited the proliferation, wound healing and tubule formation of HUVECs, whereas apoptosis was accelerated by HG, indicating that HG may prevent wound healing by inhibiting cell bioactivity. Moreover, studies have demonstrated that GAS5 participates in the regulation of cell bioactivity via various mechanisms. In HG-treated mesangial cells, GAS5 can inhibit cell proliferation and extracellular matrix accumulation through the NF-κB pathway [30]. Xie et al. reported that GAS5 is involved in HG-induced cell inflammation, oxidative stress and pyroptosis by sponging miR-452-5p [31]. In this study, our findings showed that GAS5 was mainly distributed in the nuclei of HUVECs and that GAS5 expression was downregulated by HG. In addition, GAS5 overexpression accelerated the proliferation, wound healing and tubule formation of HG-treated HUVECs, but GAS5 overexpression inhibited HUVEC apoptosis. These findings indicated that GAS5 may participate in the bioactivity of HG-treated HUVECs by regulating target genes.
TAF15, a member of the FET family, plays vital roles in regulating gene transcription, RNA splicing and protein translation and exhibits a high translocation rate in certain diseases, including tumours and diabetes [32]. In this study, TAF15 was downregulated in the skin tissues of DFU patients, and its expression was downregulated by HG. Previous studies have shown that lncRNAs can regulate gene expression by interacting with RBPs. Furthermore, RNA pull-down and RIP assays indicated that there was an interaction between GAS5 and TAF15, and GAS5 overexpression significantly increased TAF15 expression in HUVECs. These results indicated that GAS5 may regulate the bioactivity of HG-treated HUVECs by interacting with TAF15.
As an important transcription factor related to angiogenesis, HIF1A is involved in the progression of various diseases. Li et al. reported that upregulated HIF1A promotes HUVEC angiogenesis under hypoxic conditions [17]. In addition, HIF1A serves as a target gene that is regulated by various lncRNAs. LncRNA MIR31HG targets HIF1A to promote cervical cancer cell proliferation and accelerate tumour progression [33]. In the present study, HIF1A was expressed at low levels under HG conditions but was significantly upregulated by TAF15 overexpression. ChIP and luciferase assays indicated that TAF15 bound to the HIF1A promoter. HIF1A silencing inhibited VEGF expression, which may affect HUVEC tube formation. Moreover, GAS5 overexpression increased the expression of the HIF1A and VEGF genes, which was reduced by HIF1A knockdown. Taken together, these results suggest that GAS5 may participate in the regulation of HIF1A and VEGF expression by interacting with TAF15. Further functional studies indicated that GAS5 overexpression promoted the HG-induced proliferation, wound healing and tubule formation of HUVECs via the HIF1A/VEGF pathway, which was confirmed in a DFU mouse model. In addition, the specificity of HIF1A in regulating VEGF is well known, but it is unclear whether the effects of GAS5 in the model are dependent on TAF15-mediated transcription or whether other mechanisms may also occur. This may be important for designing therapies to promote or inhibit metabolic pathways in the future. Using the starBase online prediction tool, we found several microRNAs (miRNAs) that potentially bind to GAS5 and HIF1A (data not shown), suggesting that GAS5 may act as a competing endogenous RNA by sponging miRNAs to upregulate HIF1A expression and subsequently modulate DFU progression. The functional mechanisms of GAS5 in DFUs are worth further exploring in the next stage of our research.
In summary, our findings showed that the GAS5 and HIF1A levels were downregulated in the skin tissues of DFU patients and that GAS5 overexpression promoted cell proliferation, wound healing and tubule formation in HG-treated HUVECs. In addition, GAS5 increased HIF1A expression by interacting with TAF15. GAS5 accelerated wound healing in DFUs by activating the HIF1A/VEGF pathway in vitro and in vivo. In conclusion, GAS5 activates the HIF1A/VEGF pathway by binding to TAF15, resulting in accelerated wound healing in DFUs. Our findings may provide a promising therapeutic target for the clinical treatment of DFUs.
Data availability
All data generated or analyzed during this study are included in this published article.
Change history
05 May 2021
A Correction to this paper has been published: https://doi.org/10.1038/s41374-021-00603-8
References
Tao F, Tang X, Tao H, Luo Y, Cao H, Xiang W, et al. Surgical treatment of diabetic foot ulcers during the COVID-19 pandemic in China. J Diabetes Complicat. 2020;34:107622.
Samad R, Yusuf S, Andriani A, Erfina E. Nurses’ perspectives on diabetic foot ulcer’s odor: a qualitative study. Enferm Clin. 2020;30:300–33.
Caruso P, Longo M, Gicchino M, Scappaticcio L, Caputo M, Maiorino MI, et al. Long-term diabetic complications as predictors of foot ulcers healing failure: a retrospective study in a tertiary-care center. Diabetes Res Clin Pract. 2020;163:108147.
Seraphim PM, Leal EC, Moura J, Gonçalves P, Gonçalves JP, Carvalho E. Lack of lymphocytes impairs macrophage polarization and angiogenesis in diabetic wound healing. Life Sci. 2020;254:117813.
Tao H, Shi P, Zhao XD, Xuan HY, Ding XS. MeCP2 inactivation of LncRNA GAS5 triggers cardiac fibroblasts activation in cardiac fibrosis. Cell Signal. 2020;74:109705.
Kumar H, Srikanth K, Park W, Lee SH, Choi BH, Kim H, et al. Transcriptome analysis to identify long non coding RNA (lncRNA) and characterize their functional role in back fat tissue of pig. Gene. 2019;703:71–82.
Li B, Luan S, Chen J, Zhou Y, Wang T, Li Z, et al. The MSC-derived exosomal lncRNA H19 promotes wound healing in diabetic foot ulcers by upregulating PTEN via microRNA-152-3p. Mol Ther Nucleic Acids. 2020;19:814–26.
Liu XQ, Duan LS, Chen YQ, Jin XJ, Zhu NN, Zhou X, et al. lncRNA MALAT1 accelerates wound healing of diabetic mice transfused with modified autologous blood via the HIF-1α signaling pathway. Mol Ther Nucleic Acids. 2019;17:504–15.
Carter G, Miladinovic B, Patel AA, Deland L, Mastorides S, Patel NA. Circulating long noncoding RNA GAS5 levels are correlated to prevalence of type 2 diabetes mellitus. BBA Clin. 2015;4:102–7.
Shi Y, Parag S, Patel R, Lui A, Murr M, Cai J, et al. Stabilization of lncRNA GAS5 by a small molecule and its implications in diabetic adipocytes. Cell Chem Biol. 2019;26:319–330.e6.
Sawaya AP, Pastar I, Stojadinovic O, Lazovic S, Davis SC, Gil J, et al. Topical mevastatin promotes wound healing by inhibiting the transcription factor c-Myc via the glucocorticoid receptor and the long non-coding RNA Gas5. J Biol Chem. 2018;293:1439–49.
Ruan X, Zheng J, Liu X, Liu Y, Liu L, Ma J, et al. lncRNA LINC00665 stabilized by TAF15 impeded the malignant biological behaviors of glioma cells via STAU1-mediated mRNA degradation. Mol Ther Nucleic Acids. 2020;20:823–40.
Ballarino M, Jobert L, Dembele D, de la Grange P, Auboeuf D, Tora L. TAF15 is important for cellular proliferation and regulates the expression of a subset of cell cycle genes through miRNAs. Oncogene. 2013;32:4646–55.
Pan L, Li Y, Jin L, Li J, Xu A. TRPM2-AS promotes cancer cell proliferation through control of TAF15. Int J Biochem Cell Biol. 2020;120:105683.
Chen L, Chen Q, Kuang S, Zhao C, Yang L, Zhang Y, et al. USF1-induced upregulation of LINC01048 promotes cell proliferation and apoptosis in cutaneous squamous cell carcinoma by binding to TAF15 to transcriptionally activate YAP1. Cell Death Dis. 2019;10:296.
Jiang YZ, Li Y, Wang K, Dai CF, Huang SA, Chen DB, et al. Distinct roles of HIF1A in endothelial adaptations to physiological and ambient oxygen. Mol Cell Endocrinol. 2014;391:60–67.
Li L, Wang M, Mei Z, Cao W, Yang Y, Wang Y, et al. lncRNAs HIF1A-AS2 facilitates the up-regulation of HIF-1α by sponging to miR-153-3p, whereby promoting angiogenesis in HUVECs in hypoxia. Biomed Pharmacother. 2017;96:165–72.
Dong SM, Cui JH, Zhang W, Zhang XW, Kou TC, Cai QC, et al. Inhibition of translation initiation factor eIF4A is required for apoptosis mediated by Microplitis bicoloratus bracovirus. Arch Insect Biochem Physiol. 2017;96:e21423.
Cui JH, Dong SM, Chen CX, Xiao W, Cai QC, Zhang LD, et al. Microplitis bicoloratus bracovirus modulates innate immune suppression through the eIF4E–eIF4A axis in the insect Spodoptera litura. Dev Comp Immunol. 2019;95:101–7.
Liu XH, Sun M, Nie FQ, Ge YB, Zhang EB, Yin DD, et al. Lnc RNA HOTAIR functions as a competing endogenous RNA to regulate HER2 expression by sponging miR-331-3p in gastric cancer. Mol Cancer. 2014;13:92.
He ZY, Wei TH, Zhang PH, Zhou J, Huang XY. Long noncoding RNA-antisense noncoding RNA in the INK4 locus accelerates wound healing in diabetes by promoting lymphangiogenesis via regulating miR-181a/Prox1 axis. J Cell Physiol. 2019;234:4627–40.
Li J, Li SX, Gao XH, Zhao LF, Du J, Wang TY, et al. HIF1A and VEGF regulate each other by competing endogenous RNA mechanism and involve in the pathogenesis of peritoneal fibrosis. Pathol Res Pract. 2019;215:644–52.
Hung SY, Tsai JS, Yeh JT, Chen KH, Lin CN, Yang HM, et al. Amino acids and wound healing in people with limb-threatening diabetic foot ulcers. J Diabetes Complicat. 2019;33:107403.
Kiya K, Kubo T. Neurovascular interactions in skin wound healing. Neurochem Int. 2019;125:144–50.
Zou J, Liu KC, Wang WP, Xu Y. Circular RNA COL1A2 promotes angiogenesis via regulating miR-29b/VEGF axis in diabetic retinopathy. Life Sci. 2020;256:117888.
Zeng B, Li Y, Jiang F, Wei C, Chen G, Zhang W, et al. LncRNA GAS5 suppresses proliferation, migration, invasion, and epithelial-mesenchymal transition in oral squamous cell carcinoma by regulating the miR-21/PTEN axis. Exp Cell Res. 2019;374:365–73.
Apaya MK, Kuo TF, Yang MT, Yang G, Hsiao CL, Chang SB, et al. Phytochemicals as modulators of β-cells and immunity for the therapy of type 1 diabetes: Recent discoveries in pharmacological mechanisms and clinical potential. Pharmacol Res. 2020;156:104754.
Liu GM, Zeng HD, Zhang CY, Xu JW. Key genes associated with diabetes mellitus and hepatocellular carcinoma. Pathol Res Pract. 2019;215:152510.
Fadini GP, Menegazzo L, Rigato M, Scattolini V, Poncina N, Bruttocao A, et al. NETosis Delays diabetic wound healing in mice and humans. Diabetes. 2016;65:1061–71.
Zhang R, Han X, Huang T, Wang X. Danggui buxue tang inhibited mesangial cell proliferation and extracellular matrix accumulation through GAS5/NF-kappaB pathway. Biosci Rep. 2019;39:BSR20181740.
Xie C, Wu W, Tang A, Luo N, Tan Y. lncRNA GAS5/miR-452-5p reduces oxidative stress and pyroptosis of high-glucose-stimulated renal tubular cells. Diabetes Metab Syndr Obes. 2019;12:2609–17.
Ruan X, Zheng J, Liu X, Liu Y, Liu L, Ma J, et al. lncRNA LINC00665 stabilized by TAF15 impeded the malignant biological behaviors of glioma cells via STAU1-mediated mRNA degradation. Mol Ther Nucleic Acids. 2020;20:823–40.
Wang R, Ma Z, Feng L, Yang Y, Tan C, Shi Q, et al. LncRNA MIR31HG targets HIF1A and P21 to facilitate head and neck cancer cell proliferation and tumorigenesis by promoting cell-cycle progression. Mol Cancer. 2018;17:162–7.
Funding
This work was supported by General Program of National Natural Science Foundation of China (81870573).
Author information
Authors and Affiliations
Contributions
Guarantor of integrity of the entire study: G-DX. Study concepts and design: G-DX and W-XP. Definition of intellectual content: P-XH and TZ. Literature research: TZ and Y-QZ. Clinical studies: W-XP, TZ, Y-QZ, KP, LX and J-JY. Experimental studies and manuscript editing: W-XP and TZ. Data acquisition: W-XP and Y-QZ. Data analysis: L-JL and KP. Statistical analysis: LX and J-JY. Manuscript preparation: W-XP and P-XH. Manuscript review: P-XH and G-DX.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethics approval
Experiment protocols were approved by the Ethics Committee of Yiyang Central Hospital and experimental procedures were conducted according to the Declaration of Helsinki Principles. The animal experiments involved in this paper were carried out in accordance with the Guidelines for the Care and Use of Laboratory Animals issued by the National Institutes of Health.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised: Due to a typeset error the Summary was omitted.
Rights and permissions
About this article
Cite this article
Peng, WX., He, PX., Liu, LJ. et al. LncRNA GAS5 activates the HIF1A/VEGF pathway by binding to TAF15 to promote wound healing in diabetic foot ulcers. Lab Invest 101, 1071–1083 (2021). https://doi.org/10.1038/s41374-021-00598-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41374-021-00598-2
This article is cited by
-
TAF15 promotes cell proliferation, migration and invasion of gastric cancer via activation of the RAF1/MEK/ERK signalling pathway
Scientific Reports (2023)
-
SNHG12/NFYC-AS1 Acted as the Sponge for hsa-miR-199a-5p to Promote the Expression of S100A8/S100A7/XDH and was Involved in the Progression of Diabetic Foot Ulcers
Molecular Biotechnology (2023)
-
MicroRNA miR-145-5p Inhibits Cutaneous Wound Healing by Targeting PDGFD in Diabetic Foot Ulcer
Biochemical Genetics (2023)
-
Exosomes Derived from Human Amniotic Mesenchymal Stem Cells Facilitate Diabetic Wound Healing by Angiogenesis and Enrich Multiple lncRNAs
Tissue Engineering and Regenerative Medicine (2023)