Abstract
Procollagen-lysine, 2-oxoglutarate 5-dioxygenases (PLODs) play important roles in cancer progression, but their role in ovarian cancer remains elusive. In silico analysis of expression of PLODs in ovarian cancer was performed with reproduction of The Cancer Genome Atlas dataset. PLOD-enriched pathways and related gene(s) were validated by immunohistochemistry (IHC) in 80 ovarian cancer tissue blocks and in vivo xenograft murine models. PLODs (PLOD-1, -2, and -3) were overexpressed in ovarian cancer tissue. Overexpression of individual PLODs showed mutual exclusivity. Each of the three PLODs was differentially expressed between normal and cancer tissue of the ovary. PLOD1 was not prognostic, whereas lower PLOD2 and higher PLOD3 expression were associated with worsened prognosis, respectively. Cases with PLOD overexpression showed enrichment in gap junctions. GJA1 (connexin 43) was significantly overexpressed in cases with PLOD overexpression. IHC in tissue showed the strongest positive correlation between PLOD3 and connexin 43 expression, followed by PLOD2. As per Harmonizome, we selected SKOV3 and CAOV3 cell lines based on constitutive high PLOD1 and PLOD2/PLOD3 expression, respectively for in vitro and in vivo modeling. Only knockdown of PLOD3 was significantly associated with decreased GJA1 expression level in both cell lines. IHC in murine xenograft tumors also showed significantly lower connexin 43 in PLOD3-KD SKOV3 tumors. We conclude that PLODs are generally overexpressed in ovarian cancer and each PLOD may be functionally non-redundant. Association between PLOD3 and gap junctions warrants further investigation.
Similar content being viewed by others
Introduction
Ovarian cancer is a major health threat to women that lacks heralding symptoms and severely compromises prognosis once spreads locally or distantly [1]. The former phenotype often invades pelvic wall, whereas the latter usually involves peritoneum, lymph nodes, lung, and liver. Germline genetic disposition accounts for ~10% of ovarian cancer cases and the most prominent culprit genetics are BRCA1/2 mutations [2]. The most common histological type of ovarian cancer is of epithelial origin that takes up 95% of cases, amid high-grade serous adenocarcinoma (HGSOC) is the most common subtype [3]. Despite multiple treatment modalities, the 5-year survival of ovarian cancer in the United States is ~45% and insightful understanding of the biology is therefore warranted.
Collagen is the major component of the extracellular matrix. The crosslink and precipitation of collagen protein depends on hydroxylation of lysine residue, a process catalyzed by procollagen-lysine, 2-oxoglutarate 5-dioxygenase (PLOD) [4]. Aberrant hydroxylation of lysyl and collagen crosslink are associated with progression of diseases like fibrosis and cancer. Three lysyl hydroxylases (LH1, LH2, and LH3) were identified with encoding genes mapped to PLOD1, PLOD2, and PLOD3. Expression of PLODs are mainly regulated at transcription level by various cytokine, transcription factors, and microRNAs [4]. Dysregulation of PLODs promotes cancer progression and metastasis, suggesting that targeting PLODs is a potential strategy for cancer treatment.
Nonetheless, the relation between PLODs and ovarian cancer remains elusive. In the current study, we aim to evaluate role of PLODs in ovarian cancer via exploration in silico and validation in tissue and in vivo. Our findings may provide novel insight to the biology of ovarian cancer and to potential therapeutic target for the disease.
Materials and methods
In silico analysis
The Cancer Genome Atlas (TCGA) dataset was used to examine expression of PLODs in ovarian cancer. Using the cBioPortal platform, we reproduced HGSOC dataset of TCGA. By setting over-expression cut-off z-score of “Exp ≥ 2”, we plotted cases with overexpressed PLODs in comparison to paired normal tissue using the OncoPrint function of cBioPortal. Differential expression of PLODs was examined using the National Cancer Institute’s Clinical Proteomic Tumor Analysis Consortium dataset, reproduced using the UALCAN platform [5]. Immunohistochemistry (IHC) staining of PLODs was examined and reproduced at Human Protein Atlas dataset, in which the survival plots of TCGA ovarian cancer patients with automatically designated high or low PLODs expression was generated. Enriched genes at expression level in cases with PLOD overexpression were generated at cBioPortal and then submitted to NET-GE analytical platform for functional annotation. Survival analysis and correlation of gene expression for combined PLODs expression in TCGA ovarian cancer cohort were studied using the GEPIA platform. Constitutive expression of PLODs in ovarian cancer cells were profiled at Harmonizome platform.
IHC
A commercially available tissue microarray (TMA) chip (combined chip of ovarian epithelial tumor and ovarian tissue adjacent to tumor, Bioaitech, China) with 80 primary ovarian cancer samples, 10 metastatic ovarian cancer lesions, and 10 adjacent normal ovary tissue sections were used. A standard IHC protocol was followed. Briefly, chip was first sliced, mounted, and subsequently deparaffinized. Sections were dehydrated with alcohol and hydrogen peroxide was applied for blockade. The sodium citrate buffer solution was prepared for antigen retrieval with microwave. The non-fat milk was applied for non-specific antigen blockade. Primary antibodies against PLOD1 (Abcam, ab262947, 1:500), PLOD2 (Abcam, ab90088, 1:200), and PLOD3 (Abcam, ab128698, 1:500) were applied overnight. Secondary antibody and DAB was subsequently applied and the section was counter-stained with hematoxylin. Production intensity and extensity of staining was first calculated. Intensity score ranged from 0 to 3 representing from no staining to brown. Extensity score was as follows: 0 for negative, 1 for 1–25% cells stained, 2 for 26–50%, 3 for 51–75%, and 4 for 76–100%. A final score of 0–4, 5–8, and 9–12 corresponded to IHC score of 0, 1, and 2, respectively. Sections were all de-identified and only information of cancer subtype, TNM stage, and tumor grade were available. Ethical approval was obtained by the chip provider and the whole experiment conformed to Declaration of Helsinki.
Cell line and RNA interference
The SKOV3 and CAOV3 cells were obtained from the National Experimental Cell Resource Sharing Platform of China. Cell lines were cultured with RPMI 1640 medium supplemented with 10% bovine serum. shRNAs targeting PLODs were selected from TCR as follows: TRCN0000293919 for PLOD1, TRCN0000064811 for PLOD2, TRCN0000294013 for PLOD3. A standard non-lipofectamine transfection protocol was followed and puromycin-resistant clone was selected. Green-fluorescent protein was integrated for efficacy determination. Quantitative PCR was performed to validate efficacy and expression of GJA1 with primers listed in Supplementary Table 2.
Xenograft murine model
Female BALB/c nude mice of 6 weeks of age were used to establish murine model. Stably transfected SKOV3 cells with shPLOD3 or shCON were injected subcutaneously to the right flank of mice (N = 10 each group). Tumors were monitor every 3 days and mice were bred for a total of 4 weeks before euthanized. CAOV3 cells were not used in murine models as pilot study showed significantly suppressed proliferation when PLOD3 was knocked down and no measurable tumor could be spotted. Extracted tumors were paraffin-embedded and IHC was performed to detect PLOD3 and GJA1. Antibody against GJA1 (connexin 43, ThermoFisher Cat. 71-0700) was applied at dilution of 1:200.
Results
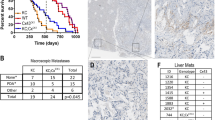
The exploration was initiated by reproduction of TCGA HGSOC dataset, which showed overexpression of PLODs (as compared to paired normal tissue) in 23% of all cases (Fig. 1a). Expression of PLOD1, PLOD2, and PLOD3 showed trend toward mutual exclusivity, indicating functional non-redundancy in part. By individual query, we found that protein levels of PLOD1 and PLOD2 were deferentially expressed between normal and cancer tissue whereas PLOD3 was not (Fig. 1b). Reproduction of HPA for PLOD expression between cancerous and normal ovary tissue however showed substantial higher expression of all PLODs in cancer as compared in normal ovary tissue (Fig. 1c). Survival plot for individual PLOD showed higher expression of PLOD3 and lower expression of PLOD2 was respectively associated with worsened overall survival (Fig. 1d). However, analysis that encompassed all PLODs showed no significant association with survival (Supplementary Fig. 1). To profile function of PLODs, we plotted genes of which the expression was significantly altered in PLODs-overexpressed cases and submitted to NET-GE. Despite a relatively small number of genes (Fig. 1e), gap junction-related genes were significantly enriched (Fig. 1f). Given the importance of connexins in gap junctions, we cross-referenced expression of all connexin genes in the enriched list and found that only GJA1 (connexin 43) was significantly upregulated in PLODs-overexpressed cases (Fig. 1g).
a Reproduced from TCGA ovarian cancer cohort, shown were cases with overexpressed PLOD1, PLOD2, and PLOD2 marked in pink. b Reproduced from CPTAC, shown were differential expression PLODs between normal ovarian tissue and ovarian cancer. Reproduced from the Human Protein Atlas, shown were c representative IHC images for positive PLOD staining in ovarian cancer and negative staining in normal ovary tissue and d plots for overall survival with automatically designated cut-off value of expression level. Reproduced from TCGA ovarian cancer cohort, shown were e volcano plot of significantly enriched genes in cases with PLOD-overexpresssion, f functional annotation of the enriched genes, and g expression of GJA1 in PLOD overexpressed and unaltered cases (*P < 0.05; ****P < 0.0001; NS not significant).
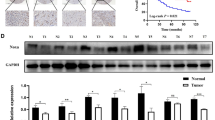
In the validation, we first compared differential expression of PLODs in TMA section. We found that PLOD1 and PLOD3 were differentially expressed in ovarian cancer (Fig. 2a). Subgroup analysis showed that higher PLOD3 expression was associated with advanced tumor stage, whereas PLOD1 or PLOD2 was not (Table 1). Nodal positive cases also showed significantly higher expression of PLOD3 but not of PLOD2, while PLOD1 expression showing marginal significance (Table 1). Of note, expression of PLOD2 and PLOD3 were higher in metastatic lesion compared to in primary tumor (Table 1). Correlation analysis for combined expression of PLODs and GJA1 in TCGA cohort showed significant linear correlation with moderate effect (Table 2). In our validation cohort, we found that expression of PLOD3 showed strong correlation with that of GJA1, whereas expression of PLOD2 showed moderate correlation (Fig. 2b). Both PLOD2 and PLOD3 showed significant linearity with GJA1, respectively (Table 2).
a Representative IHC image showing expression of PLOD and GJA1 in validation tissue microarray chip, scale bar equals 100 micrometers. b Reproduced from TCGA ovarian cancer cohort, shown was correlation between PLOD and GJA1; c expression GJA1 in ovarian cancer cell lines with or without PLOD knockdown; d representative IHC image of PLOD3 expression in SKOV3 xenograft tumor and expression and relation with GJA1 (**P < 0.01; ***P < 0.001).
Choice of cell lines was based on constitutive expression of PLODs (Supplementary Table 1). Only PLOD3-KD induced significant decrease of GJA1 in both SKOV3 and CAOV3 cells (Fig. 2c). Though not the primary scope of this study, we observed significant inhibition of proliferation in PLOD3-KD CAOV3 cells, validating the essential role of PLOD3 in CAOV3 cells (Supplementary Fig. 2). We then examined whether this association extended in vivo and found significant lower connexin 43 level in PLOD3-KD tumors of SKOV3 cells, as CAOV3 cells was not able to form tumors when PLOD3 was knocked down (Fig. 2d).
Discussion
In the current study, we found that PLODs play a role in ovarian cancer. The trend to mutual exclusivity of PLODs and different association with clinicopathological parameters and connexin 43 indicated functional linkage and non-redundancy of PLODs in ovarian cancer. In the maturation of collagen, lysyl residue in the helix undergoes O-linked glycosylation, a process catalyzed by hydroxylysine galactosyltransferase (GT) and galactosylhydroxylysine-glucosyltransferase (GGT) [6, 7]. Enzymatic activity of GT and GGT was only found in PLOD3 but not in PLOD1 or PLOD2 [8], despite those proteins being highly homologous. Unlike PLOD1 and PLOD2, PLOD3 has glycosylation activity, which induces adhesion of mono- and di-saccharide to hydroxylated lysine of collagen [9]. PLODs are mainly regulated at transcription level. For instance, hypoxia-induced factor-1 activates PLOD1 in breast cancer and to a better extent, activates PLOD2 in cancer development [10]. Unlike PLOD1 and PLOD3, regulation of PLOD3 is poorly understood. One report showed that miR-663a targets 3’-UTR of PLOD3 and downregulates its expression that ensues decreased accumulation of extracellular collagen. Given the critical role of PLODs in collagen biosynthesis, unveiling the molecular mechanism therein may substantially broaden our understanding of the role of collagen in cancer progression.
In general, increased collagen precipitation and crosslink promote cancer development and invasion via augmenting migratory ability [11,12,13,14]. PLOD3 is overexpressed in hepatic cell carcinoma (HCC) and is a potential biomarker. Inhibition of PLOD3 suppresses occurrence and growth of HCC in autochthonous murine model [15]. Nicastri et al. used proteomics to identify 54 differential genes that were upregulated in colon cancer, including PLOD2 and PLOD3 [16]. Interestingly, PLOD2 has little impact on primary tumor growth but has substantial impact on metastasis. PLOD2 hydroxylates telopeptidyl lysine residues on collagen, subsequently increasing the level of hydroxylysine aldehyde-derived collagen cross-links and lowering levels of lysine aldehyde-derived cross-links in lung cancer tissue [17]. Former knowledge indicates that PLOD2 protein is located in ER [18], yet a recent study shows that PLOD2 protein can be secreted by lung cancer cells and can induce secretion of collagen [19]. As client protein and function of PLOD1, PLOD2, and PLOD3 are different in collagen formation, it is important to exploit PLOD-specific inhibitors to curb cancer progression.
In the current study, we have for the first time reported the relation between collagen synthesis and connexin 43 in cancer. Connexin is a four-pass transmembrane protein that facilitates assembly of gap junctions of adjacent cells, promoting gap junction intercellular communication. In common, connexin is proposed to mediate formation of hemichannels and GJIC assembly to transport iron and small molecules. Many studies have reported down-expression of connexin or altered cellular localization that leads to GJIC loss. Connexin is in general believed to be cancer-suppressive [20]. However, recent studies show that connexin differs in function by its expression level and membrane localization, showing increased levels of connexin 43 and 26 in metastatic lesion of cancer and both connexins promote cancer cells to migrate to pulmonary endothelium [21]. Our findings unveil a potential novel regulatory mechanism between collagen and gap junctions, two important extracellular microenvironment that play a role in cancer metastasis. We thus postulate that ovarian cancer increases the accumulation of connexin 43 on the cell surface, leading to greater gap junction intercellular communication and enhanced collagen organization [22, 23].
References
Webb PM, Jordan SJ. Epidemiology of epithelial ovarian cancer. Best Pract Res Clin Obstet Gynaecol. 2017;41:3–14.
Toss A, Tomasello C, Razzaboni E, Contu G, Grandi G, Cagnacci A, et al. Hereditary ovarian cancer: not only BRCA 1 and 2 genes. Biomed Res Int. 2015;2015:341723.
Ebell MH, Culp MB, Radke TJ. A systematic review of symptoms for the diagnosis of ovarian cancer. Am J Prev Med. 2016;50:384–94.
Qi Y, Xu R. Roles of PLODs in collagen synthesis and cancer progression. Front Cell Dev Biol. 2018;6:66.
Chandrashekar DS, Bashel B, Balasubramanya SAH, Creighton CJ, Ponce-Rodriguez I, Chakravarthi B, et al. UALCAN: a portal for facilitating tumor subgroup gene expression and survival analyses. Neoplasia. 2017;19:649–58.
Shinkai H, Yonemasu K. Hydroxylysine-linked glycosides of human complement subcomponent C1q and various collagens. Biochem J. 1979;177:847–52.
Sricholpech M, Perdivara I, Nagaoka H, Yokoyama M, Tomer KB, Yamauchi M. Lysyl hydroxylase 3 glucosylates galactosylhydroxylysine residues in type I collagen in osteoblast culture. J Biol Chem. 2011;286:8846–56.
Heikkinen J, Risteli M, Wang C, Latvala J, Rossi M, Valtavaara M, et al. Lysyl hydroxylase 3 is a multifunctional protein possessing collagen glucosyltransferase activity. J Biol Chem. 2000;275:36158–63.
Valtavaara M, Szpirer C, Szpirer J, Myllyla R. Primary structure, tissue distribution, and chromosomal localization of a novel isoform of lysyl hydroxylase (lysyl hydroxylase 3). J Biol Chem. 1998;273:12881–6.
Gilkes DM, Bajpai S, Wong CC, Chaturvedi P, Hubbi ME, Wirtz D, et al. Procollagen lysyl hydroxylase 2 is essential for hypoxia-induced breast cancer metastasis. Mol Cancer Res. 2013;11:456–66.
Provenzano PP, Eliceiri KW, Campbell JM, Inman DR, White JG, Keely PJ. Collagen reorganization at the tumor-stromal interface facilitates local invasion. BMC Med. 2006;4:38.
Provenzano PP, Inman DR, Eliceiri KW, Knittel JG, Yan L, Rueden CT, et al. Collagen density promotes mammary tumor initiation and progression. BMC Med. 2008;6:11.
Levental KR, Yu H, Kass L, Lakins JN, Egeblad M, Erler JT, et al. Matrix crosslinking forces tumor progression by enhancing integrin signaling. Cell. 2009;139:891–906.
Zhu J, Xiong G, Fu H, Evers BM, Zhou BP, Xu R. Chaperone Hsp47 drives malignant growth and invasion by modulating an ECM gene network. Cancer Res. 2015;75:1580–91.
Shen Q, Eun JW, Lee K, Kim HS, Yang HD, Kim SY, et al. Barrier to autointegration factor 1, procollagen-lysine, 2-oxoglutarate 5-dioxygenase 3, and splicing factor 3b subunit 4 as early-stage cancer decision markers and drivers of hepatocellular carcinoma. Hepatology. 2018;67:1360–77.
Nicastri A, Gaspari M, Sacco R, Elia L, Gabriele C, Romano R, et al. N-glycoprotein analysis discovers new up-regulated glycoproteins in colorectal cancer tissue. J Proteome Res. 2014;13:4932–41.
Chen Y, Terajima M, Yang Y, Sun L, Ahn YH, Pankova D, et al. Lysyl hydroxylase 2 induces a collagen cross-link switch in tumor stroma. J Clin Invest. 2015;125:1147–62.
Liefhebber JM, Punt S, Spaan WJ, van Leeuwen HC. The human collagen beta(1-O)galactosyltransferase, GLT25D1, is a soluble endoplasmic reticulum localized protein. BMC Cell Biol. 2010;11:33.
Chen Y, Guo H, Terajima M, Banerjee P, Liu X, Yu J, et al. Lysyl hydroxylase 2 is secreted by tumor cells and can modify collagen in the extracellular space. J Biol Chem. 2016;291:25799–808.
Wu JI, Wang LH. Emerging roles of gap junction proteins connexins in cancer metastasis, chemoresistance and clinical application. J Biomed Sci. 2019;26:8.
Zeng SG, Lin X, Liu JC, Zhou J. Hypoxiainduced internalization of connexin 26 and connexin 43 in pulmonary epithelial cells is involved in the occurrence of nonsmall cell lung cancer via the P53/MDM2 signaling pathway. Int J Oncol. 2019;55:845–59.
Ehrlich HP, Sun B, Saggers GC, Kromath F. Gap junction communications influence upon fibroblast synthesis of Type I collagen and fibronectin. J Cell Biochem. 2006;98:735–43.
Moyer KE, Ehrlich HP. Modulation of human fibroblast gap junction intercellular communication by hyaluronan. J Cell Physiol. 2003;196:165–70.
Funding
National Natural Science Foundation of China, Grant/Award Number: 81602288; National Key R&D Program of China, Grant/Award Number: 2016YFC1303100; Research project of Shanghai Municipal Health and Family Planning Commission, Grant/Award Number: 20174Y0046.
Author information
Authors and Affiliations
Contributions
TG and CX designed the study. TG, CG, and BL conducted experiments. TG performed statistical analysis. TG and CG drafted manuscript. All authors proof-read the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Guo, T., Gu, C., Li, B. et al. PLODs are overexpressed in ovarian cancer and are associated with gap junctions via connexin 43. Lab Invest 101, 564–569 (2021). https://doi.org/10.1038/s41374-021-00533-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41374-021-00533-5
This article is cited by
-
Collagen synthesis and gap junctions: the highway for metastasis of ovarian cancer
Laboratory Investigation (2021)