Abstract
Objective
To identify gestational age (GA) specific risk factors for severe ROP (sROP).
Study design
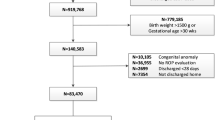
Single-center cohort stratified by GA into <24 weeks, 24–26 weeks and ≥27 weeks.
Results
132/1106 (11.9%) developed sROP. Time to full feeds was the only risk factor [HR 1.003 (1.001–1.006), p = 0.04] for infants<24 weeks GA. For infants 24–26 weeks GA, a higher GA was protective [HR 0.66 (0.51–0.85), p < 0.01], whereas steroids for bronchopulmonary dysplasia (BPD) [HR 2.21 (1.28–3.26), p < 0.01], patent ductus arteriosus (PDA) ligation [HR 1.99 (1.25–3.11), p < 0.01] and use of nitric oxide [HR 1.96 (1.11–3.30), p = 0.01] increased the hazard of sROP. Increasing birthweight was protective [HR 0.70 (0.54–0.89), p < 0.01] in infants ≥27 weeks GA. Cumulative hazard of sROP reached 1.0 by fifteen weeks for <24 weeks GA, 0.4 by twenty weeks for 24–26 weeks GA, and 0.05 by twenty weeks after birth for ≥27 weeks GA.
Conclusions
Risk factors, cumulative hazard, and time to sROP vary by GA.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
Data availability
Sharing of local data generated for this study is subject to rules per institutional review board.
References
Tsai AS, Chou HD, Ling XC, Al-Khaled T, Valikodath N, Cole E, et al. Assessment and management of retinopathy of prematurity in the era of anti-vascular endothelial growth factor (VEGF). Prog Retin Eye Res. 2021;88:101018.
Hartnett ME. Advances in understanding and management of retinopathy of prematurity. Surv Ophthalmol. 2017;62:257–76.
Ludwig CA, Chen TA, Hernandez-Boussard T, Moshfeghi AA, Moshfeghi DM. The Epidemiology of retinopathy of prematurity in the United States. Ophthalmic Surg Lasers Imaging Retin. 2017;48:553–62.
Blencowe H, Lawn JE, Vazquez T, Fielder A, Gilbert C. Preterm-associated visual impairment and estimates of retinopathy of prematurity at regional and global levels for 2010. Pediatr Res. 2013;74(Suppl 1):35–49.
Bahmani T, Karimi A, Rezaei N, Daliri S. Retinopathy prematurity: a systematic review and meta-analysis study based on neonatal and maternal risk factors. J Matern Fetal Neonatal Med. 2021;35:1–19.
Bowe T, Nyamai L, Ademola-Popoola D, Amphornphruet A, Anzures R, Cernichiaro-Espinosa LA, et al. The current state of retinopathy of prematurity in India, Kenya, Mexico, Nigeria, Philippines, Romania, Thailand, and Venezuela. Digit J Ophthalmol. 2019;25:49–58.
Darlow BA, Gilbert C. Retinopathy of prematurity—A world update. Semin Perinatol. 2019;43:315–6.
Slidsborg C, Jensen A, Forman JL, Rasmussen S, Bangsgaard R, Fledelius HC, et al. Neonatal risk factors for treatment-demanding retinopathy of prematurity: a Danish National Study. Ophthalmology 2016;123:796–803.
Seiberth V, Linderkamp O. Risk factors in retinopathy of prematurity. a multivariate statistical analysis. Ophthalmologica 2000;214:131–5.
Kim SJ, Port AD, Swan R, Campbell JP, Chan RVP, Chiang MF. Retinopathy of prematurity: a review of risk factors and their clinical significance. Surv Ophthalmol. 2018;63:618–37.
Hagadorn JI, Richardson DK, Schmid CH, Cole CH. Cumulative illness severity and progression from moderate to severe retinopathy of prematurity. J Perinatol. 2007;27:502–9.
Thomas K, Shah PS, Canning R, Harrison A, Lee SK, Dow KE. Retinopathy of prematurity: Risk factors and variability in Canadian neonatal intensive care units. J Neonatal Perinat Med. 2015;8:207–14.
Enomoto H, Miki A, Matsumiya W, Honda S. Evaluation of oxygen supplementation status as a risk factor associated with the development of severe retinopathy of prematurity. Ophthalmologica 2015;234:135–8.
Yau GS, Lee JW, Tam VT, Liu CC, Yip S, Cheng E, et al. Incidence and risk factors of retinopathy of prematurity from 2 neonatal intensive care units in a Hong Kong Chinese population. Asia Pac J Ophthalmol (Philos). 2016;5:185–91.
Lad EM, Hernandez-Boussard T, Morton JM, Moshfeghi DM. Incidence of retinopathy of prematurity in the United States: 1997 through 2005. Am J Ophthalmol. 2009;148:451–8.
Fierson WM, Chiang MF, Good W, Phelps D, Reynolds J, Robbins SL, et al. Screening examination of premature infants for retinopathy of prematurity. Pediatrics 2018;142:e20183061.
Early Treatment For Retinopathy Of Prematurity Cooperative Group. Revised indications for the treatment of retinopathy of prematurity: results of the early treatment for retinopathy of prematurity randomized trial. Arch Ophthalmol. 2003;121:1684–94.
Pivodic A, Hard AL, Lofqvist C, Smith LEH, Wu C, Brunder MC, et al. Individual risk prediction for sight-threatening retinopathy of prematurity using birth characteristics. JAMA Ophthalmol. 2020;138:21–9.
Schaffer DB, Palmer EA, Plotsky DF, Metz HS, Flynn JT, Tung B, et al. Prognostic factors in the natural course of retinopathy of prematurity. The Cryotherapy for Retinopathy of Prematurity Cooperative Group. Ophthalmology 1993;100:230–7.
Ying GS, Quinn GE, Wade KC, Repka MX, Baumritter A, Daniel E, et al. Predictors for the development of referral-warranted retinopathy of prematurity in the telemedicine approaches to evaluating acute-phase retinopathy of prematurity (e-ROP) study. JAMA Ophthalmol. 2015;133:304–11.
Walsh MC, Yao Q, Horbar JD, Carpenter JH, Lee SK, Ohlsson A. Changes in the use of postnatal steroids for bronchopulmonary dysplasia in 3 large neonatal networks. Pediatrics 2006;118:e1328–35.
Watterberg KL. American Academy of Pediatrics. Committee on F, Newborn. Policy statement-postnatal corticosteroids to prevent or treat bronchopulmonary dysplasia. Pediatrics 2010;126:800–8.
Movsas TZ, Spitzer AR, Gewolb IH. Postnatal corticosteroids and risk of retinopathy of prematurity. J AAPOS. 2016;20:348–52.
Rao R, Mashburn CB, Mao J, Wadhwa N, Smith GM, Desai NS. Brain-derived neurotrophic factor in infants <32 weeks gestational age: correlation with antenatal factors and postnatal outcomes. Pediatr Res. 2009;65:548–52.
Wickramasinghe LC, Lau M, Deliyanti D, Gottschalk TA, van Wijngaarden P, Talia D, et al. Lung and eye disease develop concurrently in supplemental oxygen-exposed neonatal mice. Am J Pathol. 2020;190:1801–12.
Wickramasinghe LC, van Wijngaarden P, Johnson C, Tsantikos E, Hibbs ML. An experimental model of bronchopulmonary dysplasia features long-term retinal and pulmonary defects but not sustained lung inflammation. Front Pediatr. 2021;9:689699.
Gorenflo M, Vogel M, Obladen M. Pulmonary vascular changes in bronchopulmonary dysplasia: a clinicopathologic correlation in short- and long-term survivors. Pediatr Pathol. 1991;11:851–66.
Bhatt AJ, Pryhuber GS, Huyck H, Watkins RH, Metlay LA, Maniscalco WM. Disrupted pulmonary vasculature and decreased vascular endothelial growth factor, Flt-1, and TIE-2 in human infants dying with bronchopulmonary dysplasia. Am J Respir Crit Care Med. 2001;164:1971–80.
Kabra NS, Schmidt B, Roberts RS, Doyle LW, Papile L, Fanaroff A, et al. Neurosensory impairment after surgical closure of patent ductus arteriosus in extremely low birth weight infants: results from the Trial of Indomethacin Prophylaxis in Preterms. J Pediatr. 2007;150:229–34. 34 e1.
Sathanandam S, Gutfinger D, Morray B, Berman D, Gillespie M, Forbes T, et al. Consensus Guidelines for the Prevention and Management of Periprocedural Complications of Transcatheter Patent Ductus Arteriosus Closure with the Amplatzer Piccolo Occluder in Extremely Low Birth Weight Infants. Pediatr Cardiol. 2021;42:1258–74.
Yang Y, Feng Y, Zhou XG, Pan JJ, Zhou XY. Inhaled nitric oxide in preterm infants: An updated meta-analysis. J Res Med Sci. 2016;21:41.
Goldstein IM, Ostwald P, Roth S. Nitric oxide: a review of its role in retinal function and disease. Vis Res. 1996;36:2979–94.
Roberts JD Jr, Fineman JR, Morin FC 3rd, Shaul PW, Rimar S, Schreiber MD, et al. Inhaled nitric oxide and persistent pulmonary hypertension of the newborn. The Inhaled Nitric Oxide Study Group. N. Engl J Med. 1997;336:605–10.
Thoene M, Anderson-Berry A. Early enteral feeding in preterm infants: a narrative review of the nutritional, metabolic, and developmental benefits. Nutrients 2021;13:2289.
Kennedy KA, Tyson JE, Chamnanvanikij S. Early versus delayed initiation of progressive enteral feedings for parenterally fed low birth weight or preterm infants. Cochrane Database Syst Rev. 2000;2:CD001970.
Konnikova Y, Zaman MM, Makda M, D’Onofrio D, Freedman SD, Martin CR. Late enteral feedings are associated with intestinal inflammation and adverse neonatal outcomes. PLoS One. 2015;10:e0132924.
Sood BG, Madan A, Saha S, Schendel D, Thorsen P, Skogstrand K, et al. Perinatal systemic inflammatory response syndrome and retinopathy of prematurity. Pediatr Res. 2010;67:394–400.
Wallace DK, Kylstra JA, Phillips SJ, Hall JG. Poor postnatal weight gain: a risk factor for severe retinopathy of prematurity. J AAPOS. 2000;4:343–7.
Lofqvist C, Andersson E, Sigurdsson J, Engstrom E, Hard AL, Niklasson A, et al. Longitudinal postnatal weight and insulin-like growth factor I measurements in the prediction of retinopathy of prematurity. Arch Ophthalmol. 2006;124:1711–8.
Hellstrom A, Engstrom E, Hard AL, Albertsson-Wikland K, Carlsson B, Niklasson A, et al. Postnatal serum insulin-like growth factor I deficiency is associated with retinopathy of prematurity and other complications of premature birth. Pediatrics 2003;112:1016–20.
Hellstrom A, Perruzzi C, Ju M, Engstrom E, Hard AL, Liu JL, et al. Low IGF-I suppresses VEGF-survival signaling in retinal endothelial cells: direct correlation with clinical retinopathy of prematurity. Proc Natl Acad Sci USA. 2001;98:5804–8.
Fu Z, Yan W, Chen CT, Nilsson AK, Bull E, Allen W, et al. Omega-3/Omega-6 long-chain fatty acid imbalance in Phase I Retinopathy of prematurity. Nutrients 2022;14:1333.
Hellstrom A, Pivodic A, Granse L, Lundgren P, Sjobom U, Nilsson AK, et al. Association of Docosahexaenoic Acid and Arachidonic Acid serum levels with retinopathy of prematurity in preterm infants. JAMA Netw Open. 2021;4:e2128771.
Hellstrom A, Nilsson AK, Wackernagel D, Pivodic A, Vanpee M, Sjobom U, et al. Effect of enteral lipid supplement on severe retinopathy of prematurity: a randomized clinical trial. JAMA Pediatr. 2021;175:359–67.
Berrocal AM, Fan KC, Al-Khersan H, Negron CI, Murray T. Retinopathy of prematurity: advances in the screening and treatment of retinopathy of prematurity using a single center approach. Am J Ophthalmol. 2022;233:189–215.
Bas AY, Demirel N, Koc E, Ulubas Isik D, Hirfanoglu IM, Tunc T, et al. Incidence, risk factors and severity of retinopathy of prematurity in Turkey (TR-ROP study): a prospective, multicentre study in 69 neonatal intensive care units. Br J Ophthalmol. 2018;102:1711–6.
McCourt EA, Ying GS, Lynch AM, Palestine AG, Wagner BD, Wymore E, et al. Validation of the colorado retinopathy of prematurity screening model. JAMA Ophthalmol. 2018;136:409–16.
Binenbaum G, Bell EF, Donohue P, Quinn G, Shaffer J, Tomlinson LA, et al. Development of modified screening criteria for retinopathy of prematurity: primary results from the postnatal growth and retinopathy of prematurity study. JAMA Ophthalmol. 2018;136:1034–40.
Quinn GE, Ying GS, Bell EF, Donohue PK, Morrison D, Tomlinson LA, et al. Incidence and early course of retinopathy of prematurity: secondary analysis of the postnatal Growth and Retinopathy of Prematurity (G-ROP) Study. JAMA Ophthalmol. 2018;136:1383–9.
Acknowledgements
We would like to thank data coordinators Amy Distler, RN and Gina Myers, RN for their help in data collection, and Dr Neeta Vachharajani for statistical support.
Funding
None This research was supported by an unrestricted grant from Research to Prevent Blindness (MR).
Author information
Authors and Affiliations
Contributions
Drs. Reynolds, Rao and Ms. GU had full access to all data in the study and take full responsibility for the integrity of the data and the accuracy of the analyses. Concept and design: TW, RR, HG, MR, AL Data acquisition, analyses and interpretation; TW, HG, RR, MR Drafting of manuscript: HW, MR, RR Critical revision of manuscript: HW, MR, RR, AL, HG Statistical analyses: HG, RR, MR Supervision: MR, RR All authors were involved in reviewing, revising and approving the final manuscript for submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wu, T., Rao, R., Gu, H. et al. Retinopathy of prematurity: risk stratification by gestational age. J Perinatol 43, 694–701 (2023). https://doi.org/10.1038/s41372-023-01604-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41372-023-01604-9