Abstract
Objective
We aim to identify potential risk factors associated with longer duration of supplemental oxygen use in preterm infants at risk for bronchopulmonary dysplasia (BPD) to better inform families and weaning protocols.
Study design
This is a retrospective study of infants with a birth gestational age (GA) < 32 0/7 weeks admitted to the neonatal intensive care unit (NICU) between October 2017 and September 2019.
Results
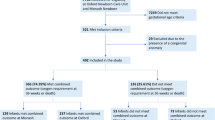
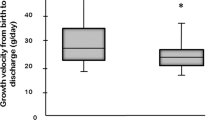
A total of 172 infants met criteria for inclusion and analysis, of which 69 (40.1%) infants required LFNC. Risk factors for longer duration included lower birth GA or birth weight, increased ventilator days, and diagnosis of a patent ductus arteriosus (PDA). BPD was diagnosed in 69.6% who required LFNC, of which 47.8% were discharged on home oxygen.
Conclusion
Younger birth GA, lower birth weight, increased ventilator days, and presence of a PDA were identified as risk factors for longer LFNC duration.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
Data availability
Data are available by request to Lawrence.Rhein@umassmemorial.org.
References
Fawke J, Lum S, Kirkby J, Hennessy E, Marlow N, Rowell V, et al. Lung function and respiratory symptoms at 11 years in children born extremely preterm: the EPICure study. Am J Respir Crit Care Med. 2010;182:237–45.
Davidson LM, Berkelhamer SK. Bronchopulmonary dysplasia: chronic lung disease of infancy and long-term pulmonary outcomes. J Clin Med. 2017;6:4.
Jensen EA, Dysart K, Gantz MG, McDonald S, Bamat NA, Keszler M, et al. The diagnosis of bronchopulmonary dysplasia in very preterm infants. An evidence-based approach. Am J Respir Crit Care Med. 2019;200:751–9.
Walsh MC, Szefler S, Davis J, Allen M, Van Marter L, Abman S, et al. Summary proceedings from the bronchopulmonary dysplasia group. Pediatrics 2006;117:S52–S6.
Stoll BJ, Hansen NI, Bell EF, Shankaran S, Laptook AR, Walsh MC, et al. Neonatal outcomes of extremely preterm infants from the NICHD Neonatal Research Network. Pediatrics 2010;126:443–56.
Horbar JD, Edwards EM, Greenberg LT, Morrow KA, Soll RF, Buus-Frank ME, et al. Variation in performance of neonatal intensive care units in the United States. JAMA Pediatrics 2017;171:e164396-e.
Jensen EA, Wright CJ. Bronchopulmonary dysplasia: the ongoing search for one definition to rule them all. J Pediatrics. 2018;197:8–10.
Beam KS, Aliaga S, Ahlfeld SK, Cohen-Wolkowiez M, Smith PB, Laughon MM. A systematic review of randomized controlled trials for the prevention of bronchopulmonary dysplasia in infants. J Perinatol. 2014;34:705–10.
Wai KC, Kohn MA, Ballard RA, Truog WE, Black DM, Asselin JM, et al. Early cumulative supplemental oxygen predicts bronchopulmonary dysplasia in high risk extremely low gestational age newborns. J Pediatrics. 2016;177:97–102. e2.
Podraza W, Michalczuk B, Jezierska K, Domek H, Kordek A, Łoniewska B, et al. Correlation of retinopathy of prematurity with bronchopulmonary dysplasia. Open Med. 2018;13:67–73.
Network SSGotEKSNNR. Target ranges of oxygen saturation in extremely preterm infants. N Engl J Med. 2010;362:1959–69.
Stop R. Investigators: Supplemental Therapeutic Oxygen for Prethreshold Retinopathy Of Prematurity (STOP-ROP), a randomized, controlled trial. I: primary outcomes. Pediatrics 2000;105:295–310.
Simoes E, Rosenberg AA, King SJ, Groothuis JR. Room air challenge: prediction for successful weaning of oxygen-dependent infants. J Perinatol: Off J Calif Perinat Assoc. 1997;17:125–9.
Roberts T, Campbell A, Larsen P, Elder D. Preterm infants at discharge: nap polysomnography versus 24‐hour oximetry. Acta Paediatrica. 2017;106:1754–9.
Vetter-Laracy S, Osona B, Peña-Zarza JA, Gil JA, Figuerola J. Hypoxia challenge testing in neonates for fitness to fly. Pediatrics. 2016;137:2–3.
Hussain N, Schwenn J, Trzaski J, Pappagallo M. Stress Oximetry: Description of a Test to Determine Readiness for Discontinuing Oxygen Therapy in Infants with Chronic Lung Disease. Int J Pediatrics. 2018;2018:5–6.
Vento G, Vendettuoli V, Aurilia C, Tana M, Tirone C, Lio A, et al. A modified physiologic test for bronchopulmonary dysplasia: a clinical tool for weaning from CPAP and/or oxygen-therapy the premature babies? Ital J Pediatrics. 2019;45:2.
Trzaski JM, Hagadorn JI, Hussain N, Schwenn J, Wittenzellner C. Predictors of successful discontinuation of supplemental oxygen in very low-birth-weight infants with bronchopulmonary dysplasia approaching neonatal intensive care unit discharge. Am J Perinatol. 2012;29:79–86.
Jackson JK, Ford SP, Meinert KA, Leick-Rude MK, Anderson B, Sheehan MB, et al. Standardizing nasal cannula oxygen administration in the neonatal intensive care unit. Pediatrics 2006;118:S187–S96.
Yang C-Y, Yang M-C, Chu S-M, Chiang M-C, Lien R. A randomized pilot study comparing the role of PEEP, O2 flow, and high-flow air for weaning of ventilatory support in very low birth weight infants. Pediatrics Neonatol. 2018;59:198–204.
Lau YY, Tay YY, Shah VA, Chang P, Loh KT. Maintaining optimal oxygen saturation in premature infants. Perm J. 2011;15:e108.
Funding
This study was part of the University of Massachusetts Medical School “Senior Scholars” research program.
Author information
Authors and Affiliations
Contributions
1. Dr LR is responsible for the content of the paper, including the data and analysis. 2. LR and JD conceived and designed the study. LR is the principal investigator who oversaw the execution, analysis, and writing of the study. JD, MH, HW made substantial contributions to the acquisition of data and were involved in the analysis and interpretation of data. AL further analyzed the data and created the figures/graphs. JD wrote the draft of the paper. LR, JD, MG, and HW revised and edited the paper. All authors read and approved the final paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
The study was reviewed and approved by the UMass Chan Medical School Institutional Review Board (IRB). Informed consent was waived for this study.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Daigneault, J., Horgan, M., White, H. et al. Low flow nasal cannula requirement among preterm infants: predictors and description of clinical course. J Perinatol 42, 1680–1685 (2022). https://doi.org/10.1038/s41372-022-01498-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41372-022-01498-z