Abstract
Objective
To evaluate the utility of the 1 Tesla (1 T) Embrace (Aspect Imaging) neonatal magnetic resonance imaging (MRI) scanner in a level III NICU.
Study design
Embrace brain MRI findings for 207 infants were reviewed, including 32 scans directly compared within 5 days with imaging on a 3 T Siemens Trio. Clinical MRI scan abnormalities were also compared to cranial ultrasound findings.
Result
Of the 207 Embrace brain MRIs, 146 (70.5%) were obtained for clinical indications and 61 (29.5%) were research cases. Abnormal findings were found in 80 scans, most commonly hemorrhage and white matter injury. Notable findings included a stroke, medullary brainstem tumor, and polymicrogyria. In the 1 T versus 3 T comparison cohort, results were discordant in only one infant with punctate foci of susceptibility noted only on the 3 T scan.
Conclusion
The Embrace MRI scans detected clinically relevant brain abnormalities and in a subset were clinically comparable to 3 T scans.
Similar content being viewed by others
Introduction
Magnetic resonance imaging (MRI) in the neonatal period provides invaluable information regarding brain injury and development [1, 2]. MRI helps identify infants at risk for suboptimal neurodevelopmental outcomes and can provide insight into structural abnormalities prior to the emergence of their functional consequences [3].
Both access to and application of MRI techniques for the neonatal population continue to evolve [4]. Notable advancements include the development of MRI compatible incubators [5,6,7], neonatal head coils [8], strategies for obtaining neonatal MRIs without sedation [9], and most recently, a specialized neonatal intensive care unit (NICU) MRI scanner dedicated to the neonatal population [10, 11].
Despite these advances, not all institutions acquire brain MRI scans in their NICU population for several reasons including limited scanner accessibility in radiology departments and perceived lack of clinically actionable findings. MRI in the neonatal intensive care unit is principally applied for two clinical indications - the first is to determine the nature and extent of brain injury in the term born infant with neonatal encephalopathy, most who have undergone therapeutic hypothermia (TH). The second common indication is in defining the type and extent of brain injury and altered brain development in the preterm infant.
In relation to the term born infant who has undergone TH for hypoxic ischemic encephalopathy (HIE), MRI is regarded as a standard of care. MRI in this population defines the nature of brain injury and its potential timing [12]. In fact, MRI findings of injury have been shown to be highly predictive of neurodevelopmental outcomes in the term born infants with HIE, both with and without hypothermia treatment [13,14,15].
In the preterm infant, controversy remains over the type and timing of neuroimaging needed [16, 17]. While many preterm infants receive cranial ultrasound (cUS), MRI is more sensitive in defining the nature and extent of abnormalities, including cerebellar hemorrhage and cerebral white matter injury [18,19,20,21,22]. White matter injury (WMI) is seen on MRI in over one third of preterm infants born between 24–32 weeks gestation [23]. From a recent meta-analysis review comparing the diagnostic utility of cUS with that of MRI, the latter was determined to be more optimal for examining the nature of brain injury and predicting later neurodevelopmental outcomes in the preterm infant [17]. Cerebellar hemorrhage is one of the more common forms of brain injury in preterm infants, seen in up to 20% of preterm infants [24, 25]. MRI has been demonstrated to have higher sensitivity for the detection of cerebellar hemorrhages compared to cUS, which detects only larger hemorrhages [26, 27].
In both populations, MRI may play an important role in detecting and evaluating brain injuries and abnormalities that may impact short- and long-term neurodevelopmental outcomes [28,29,30,31,32]. MRI is particularly well-suited for identifying infants who could benefit from neurorehabilitation during a period of plasticity in the developing nervous system [33]. It is notable that for adults or children who may be at risk for brain injury and/or have spent significant time in an intensive care unit, the use of neuroimaging with MRI to define the presence of brain injury or abnormalities is often the standard of care [34,35,36,37]. Unfortunately, for the preterm infant who may spend many months in the NICU, this is not the case.
Brigham and Women’s Hospital (BWH) is uniquely equipped with an FDA approved in-NICU MRI scanner, the Embrace Neonatal MRI System (Aspect Imaging). The in-NICU MRI scanner offers numerous benefits. It is specifically designed to accommodate the sickest most unstable NICU infants supported by many devices, such as nitric oxide administration, that are difficult to transport out of the NICU and cannot enter the scanner room of a traditional 1.5 or 3 Tesla (3 T) MRI. Having an MRI system on the NICU floor may also lower patient risk during intra-hospital transport off unit by reducing exposure risk to infection, eliminating change in intravenous lines, decreasing time away from bedside, and providing easy access to lifesaving equipment and personnel.
The objective of this paper is to summarize our clinical experience with the 1 T in-NICU Embrace Neonatal MRI scanner in a level III NICU at BWH over approximately 2 years, including the diagnostic capability with general use and a subset of comparative 3 T MRI scans.
Subjects and methods
From January 2019 until March 2021, 207 preterm (infant born <37 weeks gestation) and term infants were admitted to the level III NICU at BWH. They received either clinically indicated MRIs (per BWH clinical practice guidelines) or were recruited for research MRIs (with consent obtained for the study from the Institutional Review Board). Clinical MRIs are reviewed and reported by one of a team of pediatric neuroradiologists at Boston Children’s Hospital. The standard of practice at Boston Children’s is to review all neonatal research MRIs in the clinical workflow and therefore all research studies were reviewed and reported in the same format as clinical studies.
MRI reports for all 207 infants who received MRIs in the in-NICU Embrace scanner were reviewed. As a comparison to the MRI, cUS reports for the abnormal clinical scans were also examined. Standard clinical cUS is obtained within the first 12 h of life for term HIE infants and for preterm infants, standard clinical cUS is obtained within the first 3 days of life, day 7–10, day 28, and at term equivalent age (TEA).
A subset of 32 infants participated in a study directly comparing the Embrace 1 T MRI system (Aspect Imaging) with a 3 T MRI system (Siemens Trio). For this study, both imaging sets were read by a pediatric neuroradiologist and scans were acquired from the same infant within 5 days of each other. From March 2020 onwards, due to COVID-19, all neonatal clinical MRIs were performed in the Embrace system with few exceptions.
There were some notable incidental findings, unrelated to the infant’s NICU course, that were first captured on the Embrace scanner. Lastly, we highlight a series of three standout compassionate care cases where the Embrace scanner had an impact on continuation or redirection of care.
Magnetic resonance imaging procedures
To prepare for the scan, infants are fed 30–40 min prior. In the Embrace scanner suite, earplugs and Natus MiniMuffs® Noise Attenuators are placed in and over the ears with a hat to secure. An MRI compatible pulse oximeter probe and MRI compatible quatrode chest leads are placed on the infant. Infants are then swaddled firmly in a blanket to secure their arms and body and limit motion before placing in the pre-heated temperature-controlled MRI bed. During the scan, infant vitals are monitored and recorded every 10 min by a nurse. The infant’s face remains visible on a video monitor while in the scanner.
Sequences
The imaging protocols on the Embrace MRI scanner consist of multiplanar pulse sequences designed by Aspect Imaging for assessment of brain structure and function. These sequences were improved over time with the most recent acquisition parameters including but not limited to the following:
-
1.
T1-weighted Fast Spin Echo scan parameters: slice thickness = 3–4 mm, TR = 600, TE = 12, FOV = 140 mm, acquisition matrix = 200 × 200, pixel bandwidth = 200 Hz, number of averages = 1–2.
-
2.
T1-weighted 3D Gradient Echo scan parameters: TR = 20, TE = 3.5, slice thickness = 1 mm, acquisition matrix = 140 × 140 × 100(slices), flip angle = 15, pixel bandwidth = 286 Hz, number of averages = 1.
-
3.
T2-weighted Fast Spin Echo scan parameters: TR = 10–13 s, TE = 130–150, slice thickness = 3–4 mm, FOV = 140 mm, acquisition matrix = 200 × 200, pixel bandwidth = 250 Hz, number of averages = 2, Echo train length (ETL) = 20, echo spacing = 12 ms.
-
4.
Diffusion Weighted “Turbo Spin Echo” Imaging parameters: TR = 14 s, TE = 125, slice thickness = 3–4, FOV = 140 × 110 mm, acquisition matrix = 92 × 72 (using partial Fourier acceleration), diffusion b-value = 1000, ETL = 43, pixel bandwidth = 435 Hz, number averages = 4, echo spacing = 9 ms with calculated Exponential Images and Apparent Diffusion Coefficient maps.
Analysis
MRI reports were classified as either normal or abnormal. Normal were categorized using keywords: “no gross abnormalities”, “no significant abnormality”, “unremarkable”, or “normal” as written in the impression. Any minor parturitional findings, such as minor subdural hemorrhage, were also classified as normal. “Abnormal” signified any MRI with a report that could not be classified as normal such as findings of volume loss, prominence of subarachnoid space, WMI, hypoxic ischemic injury, tumor, ischemic injury, any form of hemorrhage, malformations, or used language such as “punctate” or “foci”.
Results
Of the 207 Aspect MRIs, 146 (70.5%) scans were clinically indicated cases and 61 (29.5%) were performed for research purposes. In total, there were 94 scans in term born infants, 85 scans in preterm infants imaged at TEA, and 28 scans in preterm infants imaged prior to term. The clinical scans in the Embrace MRI included the following neonatal populations: critically ill preterm infants imaged for injury severity (n = 14), high risk preterm infants routinely imaged at TEA (n = 60), term born infants who have undergone TH (n = 52), and term born infants imaged for other clinical concern (n = 20). Of the 146 infants scanned for clinical indications, Embrace images were interpreted to have abnormal findings on 59 scans (40.4%).
For the 61 infants scanned for research purposes, these were obtained in serially imaged preterm infants (n = 13), preterm at TEA and typical term born infants in the comparison subset (n = 32) (one imaged prior to term), and term born infants who were evaluated for TH and did not receive treatment (n = 16). Of these research MRIs, there were a total of 21 abnormal scans (34.4%).
For all scans, the most common abnormalities were hemorrhage (most commonly intraventricular or germinal matrix hemorrhage) and WMI (Table 1). Of these findings, most were seen in the preterm population (n = 52) with term abnormalities being less common (n = 28). In total, there were only nine cases (9/207, 4.3%) where expert neuroradiologists requested a repeat MRI using the 3 T scanner following the 1 T scan for magnetic resonance angiography, magnetic resonance venography, or magnetic resonance spectroscopy or to obtain higher resolution imaging for diagnostic purposes.
Comparison of 1T and 3T MRI scanners
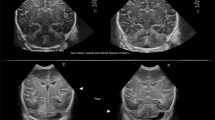
In the 32 infants (n = 1 preterm, n = 24 TEA, and n = 7 term born) with both Embrace 1 T and 3 T scans, the 1 T report correlated with the 3 T report for the presence of injury in all but one case. In this one case, there were very small scattered punctate susceptibility foci in the cerebellum seen on the 3 T and not on the Embrace 1 T. Reduced sensitivity for hemorrhage due to the absence of susceptibility weighted imaging on the 1 T scanner was a likely explanation for increased sensitivity for hemorrhage with the 3 T. Overall, there was no difference in the severity of brain injury noted between the 1 T and 3 T magnets. In addition to the injury, there was a notable case where the initial 3 T MRI was reported as normal while the 1 T Embrace scan reported right perisylvian polymicrogyria (Fig. 1a).
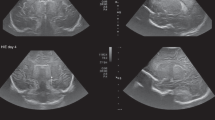
a Siemens Trio: sagittal T2 weighted imaging shows perisylvian polymicrogyria (arrows). b Siemens Prisma: axial diffusion weighted imaging shows left occipital PCA infarct (arrow). PCA: posterior cerebral artery. c Siemens Verio: sagittal T2 weighted images show left medullary brainstem tumor (arrow).
Comparison of MRI and cUS
Of the 59 abnormal clinical scans (n = 8 preterm, n = 32 TEA, and n = 19 term born), there were 15 cases in which the complementary cUS were reported as normal. Of the 17 clinical MRIs with reported WMI, there were 10 where MRI detected WMI and the cUS was reported as either normal or showed abnormalities not involving the white matter (Table 2, cases 1–10). Two MRIs detected injury patterns consistent with HIE, which were undetected on the cUS (Table 2, cases 11 and 12). There were 25 cases of hemorrhage detected or suspected on cUS while only 20 on MRI.
Incidental findings first discovered on the embrace
In the preterm subjects receiving biweekly research MRIs until TEA, one infant had moderately severe WMI. Based on these findings, more intensive physical and occupational therapy services were implemented while in the NICU.
In another research population of term infants evaluated for TH treatment but who did not meet treatment criteria, MRIs were obtained. Of the 15 scans obtained on the Embrace scanner, four were classified as abnormal. One MRI detected a stroke in a term baby imaged on day of life three with decreased diffusion and increased T2 signal involving the left occipital lobe consistent with an infarct in the posterior cerebral artery territory. Additional small foci of decreased diffusion were seen along the medial left posterior frontal and parietal cortex, likely representing small foci of additional embolic events in the posterior cerebral artery/middle cerebral artery watershed territories (Fig. 1b). Another scan in this population incidentally detected an expansile T2 hyperintense ventral medulla lesion in a term baby also imaged on day of life three, which was concerning for a low-grade glioma (Fig. 1c). This finding warranted consultation and close follow-up with neurology, neurosurgery, and neuro-oncology.
Compassionate care cases
The advantages of the in-NICU MRI were recognized in a time of great need when it was utilized for NICU infants with severe clinical instability in a series of compassionate care cases. Under these circumstances, critically ill infants were unable to leave the unit, which led to the scanner’s use as a clinical tool prior to becoming the primary diagnostic scanner for NICU infants per the approval by the Departments of Neonatology and Neuroradiology.
In one case (Fig. 2a), a very preterm infant with severe posthemorrhagic ventricular dilation had cUS with recommendations to obtain an MRI to better assess injury and aid in prognosis. Given the prematurity, weight, and ventilator dependence, compassionate care access to the NICU scanner was obtained. MRI findings showed severe ventriculomegaly and bilateral grade III intraventricular hemorrhage with simplified gyral pattern for age but no other lesions. The parents elected to continue intensive care; presently, this infant has significant developmental delay.
a Very preterm infant. Axial T1 weighted image (left) and T2 weighted image (right) show ventriculomegaly and intraventricular hemorrhage. b Extremely preterm infant with respiratory failure and gram-negative E. Coli sepsis. Axial T2 weighted image left shows intraventricular hemorrhage with regions of medullary vein thromboses (left). Axial DWI (middle) and ADC map (right) show severe diffuse white matter and thalamic decreased diffusion consistent with severe diffuse injury. c Term infant scanned on day 4 of life. Axial T2 weighted image (left), axial Diffusion Weighted Image (middle), and axial ADC map (right) in a neonate with hypoxic ischemic encephalopathy shows severe diffuse injury on DWI.
In a second case (Fig. 2b) of an extremely preterm infant who was critically ill on a high frequency jet ventilator and inotrope infusions with sepsis and meningoencephalitis, there was concern over the extent of brain injury. cUS showed a small amount of intraventricular hemorrhage but amplitude integrated electroencephalography showed more severe background abnormality and frequent seizures suggestive of greater abnormality. MRI detected severe WMI and parenchymal destruction with a poorer prognosis than previously anticipated. The information provided by the MRI led to the family’s decision to provide supportive care only and the infant passed away peacefully.
The final compassionate care case (Fig. 2c) involved a term infant, with severe encephalopathy and multiorgan dysfunction. The infant’s electroencephalogram confirmed moderate to severe encephalopathy. The baby was comatose on a high frequency ventilator with nitric oxide, eight infusions, and extremely unstable. MRI findings revealed limited anterior white matter and cortical injury, not diffuse severe injury as expected. These findings led to a continuation of care. This infant has typical developmental outcomes at 12 months of age.
Discussion
The Embrace system, optimized for in-NICU MRI of infants, has the potential to be a valuable clinical tool in providing information for care and treatment. In the small cohort with 3 T comparison, the Embrace provided images with a similar diagnostic yield and the cohort with cUS comparison demonstrated superiority in the detection of brain injury. In clinical and research use, the Embrace scanner demonstrated findings of clinical significance.
With the unexpected COVID-19 pandemic, we were forced to re-evaluate our previous clinical standard of acquiring neonatal MRIs in an adult off-unit 3 T MRI. Having access to an in-unit MRI proved to be extremely practical and useful during a time when leaving the unit was highly undesirable. Following this change in practice, we undertook a much larger number of both clinical and research scans on the in-NICU MRI system allowing us to gain confidence in its clinical utility. As a result, the Embrace has become the primary clinical scanner for our neonatal population at BWH.
By directly comparing the image quality and diagnoses made between our 1 T and 3 T magnets, we were able to demonstrate the potential diagnostic utility of a lower Tesla magnet in a clinical setting. However, the 1 T magnet has its limitations. It lacks the ability to obtain magnetic resonance spectroscopy, magnetic resonance angiography, and magnetic resonance venography. Although it provides diffusion weighted and apparent diffusion coefficient maps, it cannot generate diffusion tensor maps for tractography. If needed, this requires the use of the 3 T scanner. Sensitivity to subtle structural abnormalities relative to a 3 T MRI is also unknown. However, based on expert review, less than 10% of our babies scanned required additional magnetic resonance scans on the 3 T MRI system due to these limitations.
One of the most valuable facets of the Embrace MRI is being able to scan the sickest infants at greatest risk for brain injury to aid in prognosis and guide parental decision making, as evidenced by our compassionate care cases. In one case, information led to the redirection of care while in the two other cases, it led to continued care. One of these two surviving infants has favorable short-term outcomes. Thus, MRI can provide reassuring information in addition to defining more severe and extensive injury.
An in-NICU neonatal MRI provides this vulnerable population equal access to a tool that was previously only designed with the adult patient in mind. The Embrace scanner is enhancing access to accurate neuroimaging of the neonatal brain with resultant knowledge to drive improved patient care.
Data availability
The datasets generated during and/or analysed during the current study are not publicly available due to this being clinical data that is part of the clinical record and not available for data sharing but are available from the corresponding author on reasonable request.
References
Dudink J, Kerr JL, Paterson K, Counsell SJ. Connecting the developing preterm brain. Early Hum Dev. 2008;84:777–782.
Ment LR, Hirtz D, Hüppi PS. Imaging biomarkers of outcome in the developing preterm brain. Lancet Neurol. 2009;8:1042–1055.
Dubois J, Benders M, Borradori-Tolsa C, Cachia A, Lazeyras F, Ha-Vinh Leuchter R, et al. Primary cortical folding in the human newborn: an early marker of later functional development. Brain. 2008;131:2028–2041.
Panigrahy A, Borzage M, Blüml S. Basic principles and concepts underlying recent advances in magnetic resonance imaging of the developing brain. Semin Perinatol. 2010;34:3–19.
Lane A, Chuk LM, Colditz PB, Coulthard A. The MRI-compatible neonatal incubator in practice. J Paediatr Child Health. 2013;49:E377–E380.
Rona Z, Klebermass K, Cardona F, Czaba CD, Brugger PC, Weninger M, et al. Comparison of neonatal MRI examinations with and without an MR-compatible incubator: advantages in examination feasibility and clinical decision-making. Eur J Paediatr Neurol. 2010;14:410–417.
Dumoulin CL, Rohling KW, Piel JE, Rossi CJ, Giaquinto RO, Watkins RD, et al. Magnetic resonance imaging compatible neonate incubator. Concepts Magn Reson Part A Bridg. Educ Res. 2002;15:117–128.
Bekiesińska-Figatowska M, Szkudlińska-Pawlak S, Romaniuk-Doroszewska A, Duczkowski M, Iwanowska B, Duczkowska A, et al. First experience with neonatal examinations with the use of MR-compatible incubator. Pol J Radio. 2014;79:268–274.
Mathur AM, Neil JJ, McKinstry RC, Inder TE. Transport, monitoring, and successful brain MR imaging in unsedated neonates. Pediatr Radio. 2008;38:260–264.
Tkach JA, Hillman NH, Jobe AH, Loew W, Pratt RG, Daniels BR, et al. An MRI system for imaging neonates in the NICU: initial feasibility study. Pediatr Radio. 2012;42:1347–1356.
Tkach JA, Merhar SL, Kline-Fath BM, Pratt RG, Loew WM, Daniels BR, et al. MRI in the neonatal ICU: initial experience using a small-footprint 1.5-T system. AJR Am J Roentgenol. 2014;202:W95–W105.
Sorokan ST, Jefferies AL, Miller SP. Imaging the term neonatal brain. Paediatr Child Health. 2018;23:322–328.
Twomey E, Twomey A, Ryan S, Murphy J, Donoghue VB. MR imaging of term infants with hypoxic-ischaemic encephalopathy as a predictor of neurodevelopmental outcome and late MRI appearances. Pediatr Radio. 2010;40:1526–1535.
Chang PD, Chow DS, Alber A, Lin YK, Youn YA. Predictive values of location and volumetric MRI injury patterns for neurodevelopmental outcomes in hypoxic-ischemic encephalopathy neonates. Brain Sci. 2020;10:991.
Cheong JL, Coleman L, Hunt RW, Lee KJ, Doyle LW, Inder TE, et al. Prognostic utility of magnetic resonance imaging in neonatal hypoxic-ischemic encephalopathy: substudy of a randomized trial. Arch Pediatr Adolesc Med. 2012;166:634–640.
Hand IL, Shellhaas RA, Milla SS. Committee on fetus and newborn, section on neurology, section on radiology. routine neuroimaging of the preterm brain. Pediatrics. 2020;146:e2020029082.
Inder TE, de Vries LS, Ferriero DM, Grant PE, Ment LR, Miller SP, et al. Neuroimaging of the preterm brain: review and recommendations. J Pediatr 2021;237:276–87.e4.
Back SA, Miller SP. Brain injury in premature neonates: A primary cerebral dysmaturation disorder? Ann Neurol. 2014;75:469–486.
Hamrick SE, Miller SP, Leonard C, Glidden DV, Goldstein R, Ramaswamy V, et al. Trends in severe brain injury and neurodevelopmental outcome in premature newborn infants: the role of cystic periventricular leukomalacia. J Pediatr. 2004;145:593–99.
Inder TE, Wells SJ, Mogridge NB, Spencer C, Volpe JJ. Defining the nature of the cerebral abnormalities in the premature infant: a qualitative magnetic resonance imaging study. J Pediatr. 2003;143:171–79.
Agut T, Alarcon A, Cabañas F, Bartocci M, Martinez-Biarge M, Horsch S, et al. Preterm white matter injury: ultrasound diagnosis and classification. Pediatr Res. 2020;87:37–49.
Maalouf EF, Duggan PJ, Counsell SJ, Rutherford MA, Cowan F, Azzopardi D, et al. Comparison of findings on cranial ultrasound and magnetic resonance imaging in preterm infants. Pediatrics. 2001;107:719–727.
Miller SP, Ferriero DM, Leonard C, Piecuch R, Glidden DV, Partridge JC, et al. Early brain injury in premature newborns detected with magnetic resonance imaging is associated with adverse early neurodevelopmental outcome. J Pediatr. 2005;147:609–616.
Steggerda SJ, Leijser LM. Wiggers-de Bruïne FT, van der Grond J, Walther FJ, van Wezel-Meijler G. Cerebellar injury in preterm infants: incidence and findings on US and MR images. Radiology. 2009;252:190–9.
Tam EW, Rosenbluth G, Rogers EE, Ferriero DM, Glidden D, Goldstein RB, et al. Cerebellar hemorrhage on magnetic resonance imaging in preterm newborns associated with abnormal neurologic outcome. J Pediatr. 2011;158:245–50.
Benders MJ, Kersbergen KJ, de Vries LS. Neuroimaging of white matter injury, intraventricular and cerebellar hemorrhage. Clin Perinatol. 2014;41:69–82.
Plaisier A, Raets MM, Ecury-Goossen GM, Govaert P, Feijen-Roon M, Reiss IK, et al. Serial cranial ultrasonography or early MRI for detecting preterm brain injury? Arch Dis Child Fetal Neonatal Ed. 2015;100:F293–300.
Ramenghi LA, Rutherford M, Fumagalli M, Bassi L, Messner H, Counsell S, et al. Neonatal neuroimaging: going beyond the pictures. Early Hum Dev. 2009;85:S75–S77.
Rutherford MA, Supramaniam V, Ederies A, Chew A, Bassi L, Groppo M, et al. Magnetic resonance imaging of white matter diseases of prematurity. Neuroradiology. 2010;52:505–521.
Counsell SJ, Rutherford MA, Cowan FM, Edwards AD. Magnetic resonance imaging of preterm brain injury. Arch Dis Child Fetal Neonatal Ed. 2003;88:F269–F274.
Anderson PJ, Cheong JL, Thompson DK. The predictive validity of neonatal MRI for neurodevelopmental outcome in very preterm children. Semin Perinatol. 2015;39:147–158.
Plaisier A, Govaert P, Lequin MH, Dudink J. Optimal timing of cerebral MRI in preterm infants to predict long-term neurodevelopmental outcome: a systematic review. Am J Neuroradiol. 2014;35:841–47.
Mathur A, Inder T. Magnetic resonance imaging-insights into brain injury and outcomes in premature infants. J Commun Disord. 2009;42:248–255.
Khalaf A, Iv M, Fullerton H, Wintermark M. Pediatric stroke imaging. Pediatr Neurol. 2018;86:5–18.
Donahue MJ, Dlamini N, Bhatia A, Jordan LC. Neuroimaging advances in pediatric stroke. Stroke. 2019;50:240–48.
Nadel J, McNally JS, DiGiorgio A, Grandhi R. Emerging utility of applied magnetic resonance imaging in the management of traumatic brain injury. Med Sci. 2021;9:10.
Wintermark M, Sanelli PC, Albers GW, Bello JA, Derdeyn CP, Hetts SW, et al. Imaging recommendations for acute stroke and transient ischemic attack patients: a joint statement by the American Society of Neuroradiology, the American College of Radiology and the Society of NeuroInterventional Surgery. J Am Coll Radio. 2013;10:828–832.
Acknowledgements
We thank all the families who provided consent for their infants to participate in our research MRI studies. We also thank all our research nurses, Tina Steele, Deborah Cuddyer, and Yvonne Sheldon, and staff who helped obtain scans in our research population.
Author information
Authors and Affiliations
Contributions
KRT and ES participated in research data acquisition, drafted the initial manuscript, and reviewed and revised the manuscript. SM helped to establish the program, draft the manuscript, conceive the work, and assist with data acquisition. PEG and EY reviewed and revised the manuscript and performed MRI reads for research studies. MED designed the studies and reviewed and revised the manuscript. TEI conceptualized and designed the studies, helped to draft the initial manuscript, and reviewed and revised the manuscript. All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Competing interests
TEI and PEG are members of the Scientific Advisory Board for Aspect Imaging.
Ethics approval and consent to participate
For all research studies, parents of all subjects were consented following institutional ethics committee approval of the project by the Mass General Brigham Institutional Review Board.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Thiim, K.R., Singh, E., Mukundan, S. et al. Clinical experience with an in-NICU magnetic resonance imaging system. J Perinatol 42, 873–879 (2022). https://doi.org/10.1038/s41372-022-01387-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41372-022-01387-5
This article is cited by
-
Do we need a brain MRI once or twice in infants treated with hypothermia?
Pediatric Research (2023)
-
Body composition measurement for the preterm neonate: using a clinical utility framework to translate research tools into clinical care
Journal of Perinatology (2022)
-
Graphitic carbon nitride–manganese oxide nanoflowers as promising T1 magnetic resonance imaging contrast material
Applied Physics A (2022)