Abstract
Objective
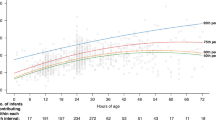
Adherence to guidelines for phototherapy initiation in preterm infants was 39% in our academic NICU (61% of phototherapy was initiated at total bilirubin (TB) levels below recommended thresholds). We hypothesized that adoption of an electronic health record integrated clinical decision support (CDS) tool would improve adherence to phototherapy guidelines.
Study design
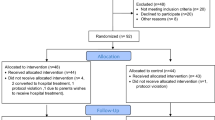
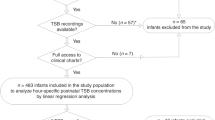
We developed and implemented Premie BiliRecs (PBR), a novel CDS tool for phototherapy initiation in preterm infants from 27 through 34 weeks postmenstrual age. The primary outcome measure was the proportion of phototherapy initiation events consistent with recommended TB thresholds.
Result
Following the implementation of PBR, adherence to guidelines for phototherapy initiation in preterm infants increased to 69.8% (p < 0.001), an improvement of 77%. There was no increase in the incidence of severe hyperbilirubinemia nor exchange transfusions.
Conclusion
The adoption of PBR was associated with improved adherence to phototherapy guidelines in preterm infants without increased adverse events.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Bhutani VK, Wong RJ, Stevenson DK. Hyperbilirubinemia in preterm neonates. Clin Perinatol. 2016;43:215–32.
Ramy N, Ghany EA, Alsharany W, Nada A, Darwish RK, Rabie WA, et al. Jaundice, phototherapy and DNA damage in full-term neonates. J Perinatol. 2016;36:132–6.
Sirota L, Straussberg R, Gurary N, Aloni D, Bessler H. Phototherapy for neonatal hyperbilirubinemia affects cytokine production by peripheral blood mononuclear cells. Eur J Pediatr. 1999;158:910–3.
Wickremasinghe AC, Kuzniewicz MW, Grimes BA, McCulloch CE, Newman TB. Neonatal phototherapy and infantile cancer. Pediatrics. 2016;137:e20151353.
Tyson JE, Pedroza C, Langer J, Green C, Morris B, Stevenson D, et al. Does aggressive phototherapy increase mortality while decreasing profound impairment among the smallest and sickest newborns? J Perinatol. 2012;32:677–84.
Morris BH, Oh W, Tyson JE, Stevenson DK, Phelps DL, O’Shea TM, et al. Aggressive vs. conservative phototherapy for infants with extremely low birth weight. N Engl J Med. 2008;359:1885–96.
American Academy of Pediatrics Subcommittee on Hyperbilirubinemia. Management of hyperbilirubinemia in the newborn infant 35 or more weeks of gestation. Pediatrics. 2004;114:297–316.
Longhurst C, Turner S, Burgos AE. Development of a Web-based decision support tool to increase use of neonatal hyperbilirubinemia guidelines. Jt Comm J Qual Patient Saf. 2009;35:256–62.
Bratlid D, Nakstad B, Hansen TW. National guidelines for treatment of jaundice in the newborn. Acta Paediatr. 2011;100:499–505.
Maisels MJ, Watchko JF, Bhutani VK, Stevenson DK. An approach to the management of hyperbilirubinemia in the preterm infant less than 35 weeks of gestation. J Perinatol. 2012;32:660–4.
Silveira PC, Sumption S, Raja AS, Tajmir S, Khorasani R. Impact of a clinical decision support tool on adherence to the Ottawa Ankle Rules. Am J Emerg Med 2016;34:412–8.
Tierney, WM. Improving clinical decisions and outcomes with information: review. Int J Med Inform, 2001;62:1–9.
Electronic Medical Record Adoption Model. https://www.himssanalytics.org/emram. Published 2017. Accessed April 24, 2019.
Palma JP, Arain YH. Development of a web-based decision support tool to operationalize and optimize management of hyperbilirubinemia in preterm infants. Clin Perinatol. 2016;43:375–83.
Wallenstein MB, Bhutani VK. Jaundice and kernicterus in the moderately preterm infant. Clin Perinatol. 2013;40:679–88.
Bhutani VK, Wong RJ. Bilirubin neurotoxicity in preterm infants: risk and prevention. J Clin Neonatol. 2013;2:61–69.
National Collaborating Centre for Women’s and Children’s Health. Neonatal Jaundice. London, England: Royal College of Obstetricians and Gynaecologists; 2010.
Greene SM, Reid RJ, Larson EB. Implementing the learning health system: from concept to action. Ann Intern Med. 2012;157:207–10.
Acknowledgements
In addition to the Stanford neonatology division and the Stanford Children’s Health Information Services department, the authors wish to acknowledge Gomathi Krishnan, PhD and the Stanford Center for Clinical Informatics for their assistance in extracting the clinical data required to make this project possible.
Author information
Authors and Affiliations
Contributions
YA and JP conceptualized and designed the study, drafted the initial manuscript, and reviewed and revised the manuscript. JB and JF collected data, carried out the initial analyses, and reviewed and revised the manuscript. VB conceptualized and designed the study, supervised data collection, and critically reviewed the manuscript for important intellectual content. All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics
This project was reviewed and approved by the Stanford University School of Medicine Institutional Review Board. No protected health information is transmitted between the EHR and the web-based CDS tool. The CDS tool guidelines were extrapolated from existing expert-consensus guidelines, but we implemented safeguards to decrease the potential for inappropriate use and unintended consequences. Because lower treatment thresholds are required shortly after birth, the PBR homepage displays a reminder that the tool is not intended for use in infants less than 48 h of age, and we prevented the EHR-integrated link to PBR from displaying in patients less than 48 h of age.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Arain, Y., Banda, J.M., Faulkenberry, J. et al. Clinical decision support tool for phototherapy initiation in preterm infants. J Perinatol 40, 1518–1523 (2020). https://doi.org/10.1038/s41372-020-00782-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41372-020-00782-0
This article is cited by
-
Reference intervals for end-tidal carbon monoxide of preterm neonates
Journal of Perinatology (2022)