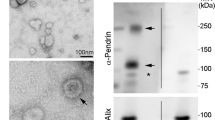
Abstract
Urinary extracellular vesicles (UEV) mainly derive from cells of the urogenital tract and their cargo (proteins, nucleic acids, lipids, etc.) reflects their cells of origin. Na chloride cotransporter (NCC) is expressed at the kidney level in the distal convoluted tubule, is involved in salt reabsorption, and is the target of the diuretic thiazides. NCC protein has been recognized and quantified in UEV in previous studies; however, UEV NCC mRNA has never been studied. This study aimed to identify and analyze NCC mRNA levels in primary aldosteronism (PA). The rationale for this investigation stems from previous observations regarding NCC (protein) as a possible biomarker for the diagnosis of PA. To evaluate modulations in the expression of NCC, we analyzed NCC mRNA levels in UEV in PA and essential hypertensive (EH) patients under different conditions, that is, before and after saline infusion, anti-aldosterone pharmacological treatment, and adrenal surgery. NCC mRNA was measured by RT-qPCR in all the samples and was regulated by volume expansion. Its response to mineralocorticoid receptor antagonist was correlated with renin, and it was increased in PA patients after adrenalectomy. NCC mRNA is evaluable in UEV and it can provide insights into the pathophysiology of distal convolute tubule in different clinical conditions including PA.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
The datasets generated for this study will not be made publicly available due to privacy policy. Requests to access the datasets should be directed to FP, francesca.pizzolo@univr.it.
References
Dominguez JH, Liu Y, Gao H, Dominguez JM 2nd, Xie D, Kelly KJ. Renal Tubular Cell-Derived Extracellular Vesicles Accelerate the Recovery of Established Renal Ischemia Reperfusion Injury. J Am Soc Nephrol. 2017;28:3533–44.
Gildea JJ, Seaton JE, Victor KG, Reyes CM, Bigler Wang D, Pettigrew AC, et al. Exosomal transfer from human renal proximal tubule cells to distal tubule and collecting duct cells. Clin Biochem. 2014;47:89–94.
Erdbrugger U, Blijdorp CJ, Bijnsdorp IV, Borras FE, Burger D, Bussolati B, et al. Urinary extracellular vesicles: A position paper by the Urine Task Force of the International Society for Extracellular Vesicles. J Extracell Vesicles. 2021;10:e12093.
Nawaz M, Camussi G, Valadi H, Nazarenko I, Ekstrom K, Wang X, et al. The emerging role of extracellular vesicles as biomarkers for urogenital cancers. Nat Rev Urol. 2014;11:688–701.
Greening DW, Simpson RJ. Understanding extracellular vesicle diversity - current status. Expert Rev Proteom. 2018;15:887–910.
Carvajal CA, Tapia-Castillo A, Perez JA, Fardella CE. Primary Aldosteronism, Aldosterone, and Extracellular Vesicles. Endocrinology. 2022;163:bqab240.
Qi Y, Wang X, Rose KL, MacDonald WH, Zhang B, Schey KL, et al. Activation of the Endogenous Renin-Angiotensin-Aldosterone System or Aldosterone Administration Increases Urinary Exosomal Sodium Channel Excretion. J Am Soc Nephrol. 2016;27:646–56.
Wolley MJ, Wu A, Xu S, Gordon RD, Fenton RA, Stowasser M. In Primary Aldosteronism, Mineralocorticoids Influence Exosomal Sodium-Chloride Cotransporter Abundance. J Am Soc Nephrol. 2017;28:56–63.
van der Lubbe N, Jansen PM, Salih M, Fenton RA, van den Meiracker AH, Danser AH, et al. The phosphorylated sodium chloride cotransporter in urinary exosomes is superior to prostasin as a marker for aldosteronism. Hypertension. 2012;60:741–8.
Castagna A, Pizzolo F, Chiecchi L, Morandini F, Channavajjhala SK, Guarini P, et al. Circadian exosomal expression of renal thiazide-sensitive NaCl cotransporter (NCC) and prostasin in healthy individuals. Proteom Clin Appl. 2015;9:623–9.
Corbetta S, Raimondo F, Tedeschi S, Syren ML, Rebora P, Savoia A, et al. Urinary exosomes in the diagnosis of Gitelman and Bartter syndromes. Nephrol Dial Transpl. 2015;30:621–30.
O’Shaughnessy KM. Gordon Syndrome: a continuing story. Pediatr Nephrol. 2015;30:1903–8.
Miranda KC, Bond DT, McKee M, Skog J, Paunescu TG, Da Silva N, et al. Nucleic acids within urinary exosomes/microvesicles are potential biomarkers for renal disease. Kidney Int. 2010;78:191–9.
Bazzell BG, Rainey WE, Auchus RJ, Zocco D, Bruttini M, Hummel SL, et al. Human Urinary mRNA as a Biomarker of Cardiovascular Disease A Proof-of-Principle Study of Sodium Loading in Prehypertension. Circ Genom Precis Med. 2018;11:e002213.
Spanu S, van Roeyen CRC, Denecke B, Floege J, Muhlfeld AS. Urinary Exosomes: A Novel Means to Non-Invasively Assess Changes in Renal Gene and Protein Expression. Plos One. 2014;9:e109631.
Thery C, Witwer KW, Aikawa E, Alcaraz MJ, Anderson JD, Andriantsitohaina R, et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): a position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J Extracell Vesicles. 2018;7:1535750.
Ko B, Mistry AC, Hanson L, Mallick R, Wynne BM, Thai TL, et al. Aldosterone acutely stimulates NCC activity via a SPAK-mediated pathway. Am J Physiol Ren Physiol. 2013;305:F645–52.
Feng X, Zhang Y, Shao N, Wang Y, Zhuang Z, Wu P, et al. Aldosterone modulates thiazide-sensitive sodium chloride cotransporter abundance via DUSP6-mediated ERK1/2 signaling pathway.2015;308: F1119-27.
Olivieri O, Ciacciarelli A, Signorelli D, Pizzolo F, Guarini P, Pavan C, et al. Aldosterone to Renin ratio in a primary care setting: the Bussolengo study. J Clin Endocrinol Metab. 2004;89:4221–6.
Funder JW, Carey RM, Mantero F, Murad MH, Reincke M, Shibata H, et al. The Management of Primary Aldosteronism: Case Detection, Diagnosis, and Treatment: An Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metab. 2016;101:1889–916.
Mulatero P, Milan A, Fallo F, Regolisti G, Pizzolo F, Fardella C, et al. Comparison of confirmatory tests for the diagnosis of primary aldosteronism. J Clin Endocrinol Metab. 2006;91:2618–23.
Pizzolo F, Salvagno G, Caruso B, Cocco C, Zorzi F, Zaltron C, et al. Fully automated chemiluminescence vs RIA aldosterone assay in primary aldosteronism work-up. J Hum Hypertens. 2017;31:826–30.
Olivieri O, Castagna A, Guarini P, Chiecchi L, Sabaini G, Pizzolo F, et al. Urinary prostasin: a candidate marker of epithelial sodium channel activation in humans. Hypertension. 2005;46:683–8.
Zhou H, Yuen PS, Pisitkun T, Gonzales PA, Yasuda H, Dear JW, et al. Collection, storage, preservation, and normalization of human urinary exosomes for biomarker discovery. Kidney Int. 2006;69:1471–6.
Wardak H, Tutakhel OAZ, Van Der Wijst J. Role of the alternative splice variant of NCC in blood pressure control. Channels (Austin). 2018;12:346–55.
Bazzell BG, Rainey WE, Auchus RJ, Zocco D, Bruttini M, Hummel SL, et al. Human Urinary mRNA as a Biomarker of Cardiovascular Disease. Circ Genom Precis Med. 2018;11:e002213.
Gunasekaran PM, Luther JM, Byrd JB. For what factors should we normalize urinary extracellular mRNA biomarkers? Biomol Detect Quantif. 2019;17:100090.
McKiernan J, Donovan MJ, O’Neill V, Bentink S, Noerholm M, Belzer S, et al. A Novel Urine Exosome Gene Expression Assay to Predict High-grade Prostate Cancer at Initial Biopsy. JAMA Oncol. 2016;2:882–9.
Gracia T, Wang X, Su Y, Norgett EE, Williams TL, Moreno P, et al. Urinary Exosomes Contain MicroRNAs Capable of Paracrine Modulation of Tubular Transporters in Kidney. Sci Rep. 2017;7:40601.
Hoorn EJ, Gritter M, Cuevas CA, Fenton RA. Regulation of the Renal NaCl Cotransporter and Its Role in Potassium Homeostasis. Physiol Rev. 2020;100:321–56.
Wu A, Wolley MJ, Wu Q, Gordon RD, Fenton RA, Stowasser M. The Cl(-)/HCO3(-) exchanger pendrin is downregulated during oral co-administration of exogenous mineralocorticoid and KCl in patients with primary aldosteronism. J Hum Hypertens. 2021;35:837–48.
Kim GH, Masilamani S, Turner R, Mitchell C, Wade JB, Knepper MA. The thiazide-sensitive Na-Cl cotransporter is an aldosterone-induced protein. Proc Natl Acad Sci . 1998;95:14552–7.
Poulsen SB, Christensen BM. Long-term aldosterone administration increases renal Na(+)-Cl(-) cotransporter abundance in late distal convoluted tubule. Am J Physiol Ren Physiol. 2017;313:F756–F66.
Czogalla J, Vohra T, Penton D, Kirschmann M, Craigie E, Loffing J. The mineralocorticoid receptor (MR) regulates ENaC but not NCC in mice with random MR deletion. Pflug Arch: Eur J Physiol. 2016;468:849–58.
van der Lubbe N, Lim CH, Meima ME, van Veghel R, Rosenbaek LL, Mutig K, et al. Aldosterone does not require angiotensin II to activate NCC through a WNK4-SPAK-dependent pathway. Pflug Arch. 2012;463:853–63.
Wang XY, Masilamani S, Nielsen J, Kwon TH, Brooks HL, Nielsen S, et al. The renal thiazide-sensitive Na-Cl cotransporter as mediator of the aldosterone-escape phenomenon. J Clin Invest. 2001;108:215–22.
Esteva-Font C, Wang X, Ars E, Guillen-Gomez E, Sans L, Gonzalez Saavedra I, et al. Are sodium transporters in urinary exosomes reliable markers of tubular sodium reabsorption in hypertensive patients? Nephron Physiol. 2010;114:p25–34.
Acknowledgements
We would like to acknowledge the special contributions of our beloved friend and colleague, Sarath Kiran Channavajjhala, who left us too early. His intelligence, dedication, and enthusiasm will always shine in our hearts. This study was performed (in part) at the Laboratorio Universitario di Ricerca Medica (LURM) Research Center at the University of Verona. TEM imaging was carried out at the Department of Neurosciences, Biomedicine and Movement Sciences at the University of Verona with the assistance of Dr. Paolo Bernardi.
Funding
This research was funded by the Italian Ministry for University and Research, project “Ricerca di Base” (to FP). The funder had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
OO, AC, and FP contributed to conception and design of the study. LB, FM, NT, and DD performed the experiments. LB, OO, FP, AC, PB, SF and FM interpreted the results. NT, AC and LB prepared the figures. LB, FP, SF and AC drafted the manuscript. LB, DD, GLS, GS, FM analysed data. All authors contributed to manuscript revision, read, and approved the final version for submission.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
This study was carried out in accordance with recommendations of the Ethics Committee of our Institution (Azienda Ospedaliera Universitaria Integrata, Verona, Italy), who approved the protocol. Written and informed consent were obtained from patients in accordance with the Declaration of Helsinki.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pizzolo, F., Bertolone, L., Castagna, A. et al. Urinary extracellular vesicle mRNA analysis of sodium chloride cotransporter in hypertensive patients under different conditions. J Hum Hypertens 37, 524–531 (2023). https://doi.org/10.1038/s41371-022-00744-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41371-022-00744-3